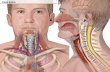
Diagnosis and management of central airway obstruction Authors Armin Ernst, MD, FCCP Felix JF Herth, MD, PhD Heinrich D Becker, MD, FCCP Section Editor Praveen N Mathur, MB, BS Deputy Editor Kevin C Wilson, MD Disclosures All topics are updated as new evidence becomes available and our peer review process is complete. Literature review current through: feb 2012. | This topic last updated: mar 30, 2010. INTRODUCTION — Central airway obstruction can occur secondary to a number of malignant and benign processes (table 1 )[1 ]. Patients may develop symptoms suddenly (eg, obstructing foreign body) or more gradually (eg, most malignant obstructions). In many cases, patients are incorrectly diagnosed as having asthma or chronic obstructive pulmonary disease, which contributes to delayed recognition and therapy. Interventional options for central airway obstruction are subject to the availability of experienced personnel and equipment. In addition, the stability of the patient, the nature of the underlying problem, and the patient's overall prognosis and quality of life impact the choice of intervention [1-5 ]. The broadest range of management options are available at centers where a multidisciplinary team specializes in the evaluation and management of the impaired airway. The diagnosis and general approach to central airway obstruction will be reviewed here. Specific management modalities are discussed in detail separately. DIAGNOSIS — The hallmark of the severely compromised airway is impairment of oxygenation and ventilation. Patients with

Diagnosis and Management of Central Airway Obstruction
Oct 22, 2014
Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript
Diagnosis and management of central airway obstruction Authors Armin Ernst, MD, FCCP Felix JF Herth, MD, PhD Heinrich D Becker, MD, FCCP Section Editor Praveen N Mathur, MB, BS Deputy Editor Kevin C Wilson, MD Disclosures All topics are updated as new evidence becomes available and our peer review process is complete. Literature review current through: feb 2012. | This topic last updated: mar 30, 2010. INTRODUCTION Central airway obstruction can occur secondary to a number of malignant and benign processes (table 1) [1]. Patients may develop symptoms suddenly (eg, obstructing foreign body) or more gradually (eg, most malignant obstructions). In many cases, patients are incorrectly diagnosed as having asthma or chronic obstructive pulmonary disease, which contributes to delayed recognition and therapy. Interventional options for central airway obstruction are subject to the availability of experienced personnel and equipment. In addition, the stability of the patient, the nature of the underlying problem, and the patient's overall prognosis and quality of life impact the choice of intervention [1-5]. The broadest range of management options are available at centers where a multidisciplinary team specializes in the evaluation and management of the impaired airway. The diagnosis and general approach to central airway obstruction will be reviewed here. Specific management modalities are discussed in detail separately. DIAGNOSIS The hallmark of the severely compromised airway is impairment of oxygenation and ventilation. Patients with minor obstruction are often asymptomatic, since airflow limitation is mild. However, rapid deterioration may occur if swelling or secretions increase the degree of luminal impingement during a respiratory tract infection. It is not uncommon for patients with subcritical lesions to be misdiagnosed as suffering from an exacerbation of asthma or chronic obstructive pulmonary disease (COPD) while the true etiology is anatomic airway obstruction. Patients with airway obstruction also frequently present with pneumonia; if symptoms and/or radiographic infiltrates do not resolve within four to six weeks, bronchoscopy should be considered. (See "Nonresolving pneumonia".) Symptoms and signs develop when airflow impairment reaches a critical threshold. Patients complain of shortness of breath, which is often constant and unresponsive to bronchodilators. Monophonic wheezing may be present, and can be unilateral if the lesion is distal to the carina. Stridor is a sign of severe subglottic or tracheal obstruction. Breathing becomes labored in advanced phases and heralds impending respiratory failure.
The onset and progression of symptoms depend upon the nature of the problem (acute with foreign bodies, slowly progressive with an expansile goiter) and the location of the lesion (tracheal versus bronchial). Patients with long-term artificial airways are at increased risk for subglottic or tracheal stenosis and tracheomalacia. Removal of the airway or capping of a tracheostomy tube can result in shortness of breath or stridor, which should prompt a thorough airway evaluation. (See "Endotracheal tube management and complications" and "Overview of tracheostomy".) A number of studies are employed to confirm the presence of central airway obstruction and estimate its magnitude:
Plain chest radiographs are rarely diagnostic. Chest computed tomography (CT) may detect airway compromise, but the test as generally performed is not sensitive. If an airway lesion is suspected and time permits, a high resolution CT with three-dimensional airway reconstruction can prove helpful [6]. These new imaging protocols may assist in both the initial assessment of the lesion and in objective grading of therapeutic success and followup (figure 1). (See "Radiology of the trachea".) Flow volume loops can show the characteristic changes of airway obstruction, frequently before abnormalities in the spirometric volumes are noted (figure 2A-E). (See "Flow-volume loops".) Direct bronchoscopic visualization is the gold standard for confirming the presence of airway obstruction and also aids in discerning its underlying etiology. Often the differentiation of endobronchial or extrinsic lesions can be accomplished only at bronchoscopy (figure 3). (See "Flexible bronchoscopy: Indications and contraindications".)
MANAGEMENT Management of central airway obstruction is divided into two phases: initial stabilization and airway interventions. Initial stabilization In a stable patient, imaging studies and pulmonary function tests should be obtained as outlined above. A patient with severe tracheal or mainstem obstruction and marginal lung function requires initial stabilization to secure ventilation and oxygenation.
Endotracheal intubation is preferred. In the presence of a compromised trachea, this is most safely performed with an awake or mildly sedated patient who is still actively breathing. The use of paralytic agents may be hazardous, since intubation may be difficult or impossible. (See "Endotracheal tube management and complications".) In cases of severe tracheal obstruction, use of the open ventilating rigid bronchoscope is the preferred method of airway control. (See "Rigid bronchoscopy: Intubation techniques".) The rigid bronchoscope not only provides a secure airway during visualization, but is also a therapeutic tool [7]. In emergent cases, the airway can be dilated with the barrel of the scope (figure 4A-B).
Bronchoscopy should be performed after the airway has been secured and appropriate gas exchange documented. During the bronchoscopic examination, the airway is inspected, lesions are assessed, distal secretions are suctioned, and diagnostic tissue is obtained if needed. This information is used to plan further interventions aimed at opening an airway and maintaining patency. If no dedicated airway team is available, patient transfer to a specialized center should be considered after the patient has been stabilized. An appropriate course of antibiotics should be administered after a patent airway has been reestablished in individuals with postobstructive infections. The usefulness of empiric antibiotic therapy after interventions in the absence of evidence of infection is unproven. Similarly, there is no evidence that corticosteroids are effective in reducing complications in this setting. It is advisable for all patients with a history of airway obstruction to carry a card or bracelet identifying them as patients with complicated airways or indwelling airway stents. Airway interventions Further interventions are planned following the initial assessment [1]. The number and scope of therapeutic options has increased dramatically, and a given intervention must be chosen carefully in the context of an individual patient's situation [8]. Multimodality approaches featuring a combination of several interventions are preferred for their mucosal sparing effects and long term success over dilation alone (figure 5 and table 2) [1,3]. The rigid bronchoscope is the preferred instrument for unstable patients and when significant bleeding is expected. Its nonflammable nature also makes it the preferred tool for many laser bronchoscopies. For most other interventions, the flexible bronchoscope with use of conscious sedation provides a therapeutic alternative. (See "Rigid bronchoscopy: Instrumentation" and "Flexible bronchoscopy: Equipment, procedure, and complications".) Foreign body extraction Foreign body aspiration is more common in children than adults, and can lead to sudden, catastrophic, central airway obstruction. Both flexible and rigid bronchoscopy can be used for foreign body extraction; the selection of procedure and technique of removal are discussed elsewhere. (See "Airway foreign bodies in adults".) Rigid or balloon dilation In emergent cases, the airway may be dilated with the rigid bronchoscope. During this procedure (called bronchoplasty), the patient is intubated with the instrument under general anesthesia. The optical telescope is advanced through the stenotic airway opening and the barrel then pushed through the obstruction in a rotating motion. Bleeding is usually minimal due to compression of the lesion by the rigid instrument (figure 4A-B). In less urgent cases, sequential balloon or rigid dilators may be used, particularly when the stenosis occurs after transplantation or long term intubation. For sequential rigid dilation, the patient must be preoxygenated carefully, which may not be feasible in severe airway stenosis. Balloon dilation can safely be performed with the flexible bronchoscope (figure 6) [9]. The major advantage of sequential rigid versus balloon dilation is that there generally is less mucosal trauma. (See "Flexible fiberoptic bronchoscopy balloon dilation".)
Dilation is immediately effective for intrinsic and extrinsic lesions, but the results are usually not sustained. Mucosal disruption from these techniques may in fact produce granulation tissue and accelerate recurrent stenosis [10]. For this reason, dilation is frequently followed by laser and/or stenting procedures, as described below [11]. Laser therapy Nd:YAG laser therapy is frequently performed utilizing the rigid bronchoscope, but can be safely performed with the flexible bronchoscope by experienced endoscopists [12]. The tissue-light interaction leads to thermal tissue damage and destruction of obstructing lesions [13]. (See "Basic principles of medical lasers" and "Bronchoscopic laser resection".) Laser therapy is indicated for short endobronchial central airway lesions with a visible distal lumen. The technique is most commonly applied in cases of malignant intrinsic airway obstruction or in postintubation tracheal stenosis. The effects upon airway lumen size are usually immediate and accompanied by excellent control of bleeding, but as is true with dilation, they are not long-lasting. Large series have demonstrated the safety of laser therapy in experienced hands [14]. Complications include combustion of the endotracheal tube or fiberoptic bronchoscope, hypoxemia, respiratory failure, and destruction of bronchial wall components. Electrocautery and argon plasma coagulation These therapies also rely on thermal tissue destruction. With electrocautery, a high-frequency current is applied to the lesion with bipolar probes. When the current is directly applied to the tissue, heat develops and leads to tissue necrosis. (See "Endobronchial electrocautery".) Argon plasma coagulation is a related therapeutic intervention [15]. Argon gas is emitted through a Teflon tube that can be passed through a flexible bronchoscope. This gas is ionized because of exposure to high-frequency current and an electrical arc is formed which allows for desiccation and tissue destruction without direct contact. The penetration depth is reliably 2 to 3 mm, which makes the argon plasma coagulator a valuable tool in treating superficial bleeding and debulking granulation tissue and tumors such as papillomas (figure 7) [1]. Photodynamic therapy Photodynamic therapy is approved for malignant intrinsic airway obstruction due to lung cancer that is unresponsive or unsuitable for laser therapy. After injection of a photosensitizing agent and a suitable time interval, tumor tissue that has retained the photosensitizing agent is exposed to a laser light of 630 nm wavelength. The laser is delivered through a fiber introduced through the flexible bronchoscope. A nonthermal phototoxic reaction leads to delayed cell death [1,16,17]. (See "Photodynamic therapy of lung cancer".) This therapeutic approach is also suitable for completely obstructed airways due to the predictable penetration depth of 5 to 10 mm. Follow-up bronchoscopy is necessary to remove debris and secretions. The main adverse effect of this approach is associated skin photosensitivity, which can last up to six weeks.
Cryotherapy As opposed to the thermal effects of laser therapy, cryotherapy relies on repeated freeze/thaw cycles for tissue destruction. Cryotherapy may be performed through the flexible bronchoscope and spares cartilaginous structures due to their poor vascularity. The intervention is more time-consuming than laser therapy because of the need for repeat cycles, and a repeat bronchoscopy for clearance of debris and secretions is usually necessary [18,19]. (See "Bronchoscopic cryosurgery: Principles and technique" and "Bronchoscopic cryosurgery: Indications, contraindications, and outcomes".) Cryotherapy can be performed safely in complete mainstem obstruction, but its delayed effects do not make it a first choice in acute situations or severe tracheal stenosis. As with any tissue-destroying interventions, long-term stabilization of the airway is often necessary. Stenting or radiation in the case of malignant lesions may achieve this. External beam radiation and brachytherapy Radiation therapy is a variably effective treatment for malignant airway obstruction, and therapeutic effects may be quite delayed. External beam radiation also may produce unwanted effects on thoracic structures outside the airway, further compromising gas exchange. Endobronchial brachytherapy is a treatment modality that has fewer of these drawbacks and is particularly useful in patients who have received previous maximal doses of external beam radiation [20]. (See "Endobronchial brachytherapy".) After a patent airway has been established by laser resection, dilation, or other methods, a hollow catheter is introduced through the flexible bronchoscope and positioned under direct vision. After the catheter is secured, it is loaded with a radioactive source [21,22]. Depending upon the results, airway stenting should be considered. Adverse effects of brachytherapy are usually minimal (tracheobronchitis, cough), but severe hemoptysis and fistula formation have been reported [22]. Airway stents Stenting should be considered to prevent reocclusion after patency has been restored to occluded or severely stenotic airways. Stents are the intervention of choice for external obstruction and are also highly effective for persistent proximal bronchopleural fistulas and tracheoesophageal fistulas [23,24]. The first dedicated tracheobronchial stent was introduced in 1990; numerous designs with various advantages and disadvantages are now available [25-27]. (See "Airway stents".) Silicone stents generally require introduction with a rigid bronchoscope, but are comparatively inexpensive. Most metal stents can be introduced with the flexible bronchoscope; their greater expense is partially offset by the fact that neither an operating room nor general anesthesia is needed [28,29]. New stent designs combining different materials are currently being evaluated [30-32]. Stents are generally well tolerated, but patients require periodic follow-up. Patients should carry a card detailing type and size of the indwelling stent. Stents do not contraindicate subsequent intubation, but intubation should preferably be performed under fiberoptic guidance if the stent is in the tracheal position.
Meticulous follow-up is indicated to identify potential problems at an early stage. These include recurrence of obstruction, growth of granulation tissue, as well as stent occlusion and migration. If identified early, these complications can be addressed in an elective manner. At our institution, a first follow-up bronchoscopy after stent placement is performed after six to eight weeks, which allows for repositioning of migrated metallic stents. Thereafter, we perform interval bronchoscopies every three to six months and as problems arise. Surgical resection Surgical intervention for airway obstruction is usually reserved for severe, benign, relatively short tracheal lesions. Patient selection is crucial, as the operative morbidity and mortality may be unacceptable in patients with limited cardiopulmonary reserve. Patients should be referred to a center with a large cumulative experience if surgical resection is a consideration. Techniques that are commonly employed are primary end-toend anastomosis and tracheal sleeve resection [33-35]. Anastomotic complications that result in recurrent stenosis may necessitate multiple dilations, reoperation, or permanent tracheostomy. These complications were noted in 9 percent of patients in one large single center series of over 900 procedures [36]. Risk factors for complications following tracheal resection in this study included diabetes, prior tracheal resection, and stenotic lesions longer than 4 cm [36]. Patients undergoing laryngotracheal resection were also at increased risk of anastomotic complications in this report. In the future, biological tissue engineering techniques may be used to create tracheal bioprostheses covered with mucosal tissue [37-44]. Tracheal transplantation, which has been difficult to achieve because of the limited blood supply to the airway, may also be possible as muscle flap reconstruction techniques continue to improve [45,46]. RECOMMENDATIONS Central airway obstruction may be extrinsic or intrinsic and may cause a variety of symptoms, from shortness of breath to respiratory failure and death. In the decompensated patient, immediate restoration of ventilation and oxygenation is of vital importance. Subsequent interventions are based upon the nature of the obstruction and refined according to issues involving quality of life and the duration of expected survival. Frequently, the best therapeutic approach employs a combination of several treatment modalities, and should be chosen at any given time in such a way that leaves open options for further therapy (figure 5). Close follow-up is necessary in order to recognize complications early and intervene accordingly. The most comprehensive assessment and therapy can be provided by centers with a multidisciplinary airway team specializing in compromised airways. Use of UpToDate is subject to the Subscription and License Agreement.
REFERENCES
1. Ernst A, Feller-Kopman D, Becker HD, Mehta AC. Central airway obstruction. Am J Respir Crit Care Med 2004; 169:1278. 2. Ernst A, Silvestri GA, Johnstone D, American College of Chest Physicians. Interventional pulmonary procedures: Guidelines from the American College of Chest Physicians. Chest 2003; 123:1693. 3. Bolliger CT, Mathur PN, Beamis JF, et al. ERS/ATS statement on interventional pulmonology. European Respiratory Society/American Thoracic Society. Eur Respir J 2002; 19:356. 4. Stephens KE Jr, Wood DE. Bronchoscopic management of central airway obstruction. J Thorac Cardiovasc Surg 2000; 119:289. 5. Seijo LM, Sterman DH. Interventional pulmonology. N Engl J Med 2001; 344:740. 6. LoCicero J 3rd, Costello P, Campos CT, et al. Spiral CT with multiplanar and threedimensional reconstructions accurately predicts tracheobronchial pathology. Ann Thorac Surg 1996; 62:811. 7. Colt HG, Harrell JH. Therapeutic rigid bronchoscopy allows level of care changes in patients with acute respiratory failure from central airways obstruction. Chest 1997; 112:202. 8. Beamis JF Jr. Interventional pulmonology techniques for treating malignant large airway obstruction: an update. Curr Opin Pulm Med 2005; 11:292. 9. Hautmann H, Gamarra F, Pfeifer KJ, Huber RM. Fiberoptic bronchoscopic balloon dilatation in malignant tracheobronchial disease: indications and results. Chest 2001; 120:43. 10. Mehta AC, Lee FY, Cordasco EM, et al. Concentric tracheal and subglottic stenosis. Management using the Nd-YAG laser for mucosal sparing followed by gentle dilatation. Chest 1993; 104:673. 11. Noppen M, Schlesser M, Meysman M, et al. Bronchoscopic balloon dilatation in the combined management of postintubation stenosis of the trachea in adults. Chest 1997; 112:1136. 12. Duhamel DR, Harrell JH 2nd. Laser bronchoscopy. Chest Surg Clin N Am 2001; 11:769. 13. Ramser ER, Beamis JF Jr. Laser bronchoscopy. Clin Chest Med 1995; 16:415. 14. Cavaliere S, Venuta F, Foccoli P, et al. Endoscopic treatment of malignant airway obstructions in 2,008 patients. Chest 1996; 110:1536. 15. Morice RC, Ece T, Ece F, Keus L. Endobronchial argon plasma coagulation for treatment of hemoptysis and neoplastic airway obstruction. Chest 2001; 119:781. 16. Edell ES, Cortese DA. Photodynamic therapy. Its use in the management of bronchogenic carcinoma. Clin Chest Med 1995; 16:455. 17. Cortese DA, Edell ES, Kinsey JH. Photodynamic therapy for early stage squamous cell carcinoma of the lung. Mayo Clin Proc 1997; 72:595. 18. Hetzel M, Hetzel J, Schumann C, et al. Cryorecanalization: a new approach for the immediate management of acute airway obstruction. J Thorac Cardiovasc Surg 2004; 127:1427. 19. Mathur PN, Wolf KM, Busk MF, et al. Fiberoptic bronchoscopic cryotherapy in the management of tracheobronchial obstruction. Chest 1996; 110:718. 20. Chella A, Ambrogi MC, Ribechini A, et al. Combined Nd-YAG laser/HDR brachytherapy versus Nd-YAG laser only in malignant central airway involvement: a prospective randomized study. Lung Cancer 2000; 27:169.
21. Nori D, Allison R, Kaplan B, et al. High dose-rate intraluminal irradiation in bronchogenic carcinoma. Technique and results. Chest 1993; 104:1006. 22. Suh JH, Dass KK, Pagliaccio L, et al. Endobronchial radiation therapy with or without neodymium yttrium aluminum garnet laser resection for managing malignant airway obstruction. Cancer 1994; 73:2583. 23. Freitag L, Tekolf E, Steveling H, et al. Management of malignant esophagotracheal fistulas with airway stenting and double stenting. Chest 1996; 110:1155. 24. Wood D. Airway stenting. Chest Surg Clin N Am 2003; 13:211. 25. Dumon JF. A dedicated tracheobronchial stent. Chest 1990; 97:328. 26. Becker, HD. Stenting of the central airways. J Bronchol 1995; 2:98. 27. Wood DE, Liu YH, Vallires E, et al. Airway stenting for malignant and benign tracheobronchial stenosis. Ann Thorac Surg 2003; 76:167. 28. Dasgupta A, Dolmatch BL, Abi-Saleh WJ, et al. Self-expandable metallic airway stent insertion employing flexible bronchoscopy: preliminary results. Chest 1998; 114:106. 29. Saad CP, Murthy S, Krizmanich G, Mehta AC. Self-expandable metallic airway stents and flexible bronchoscopy: long-term outcomes analysis. Chest 2003; 124:1993. 30. Bolliger CT, Breitenbuecher A, Brutsche M, et al. Use of studded Polyflex stents in patients with neoplastic obstructions of the central airways. Respiration 2004; 71:83. 31. Noppen M, Meysman M, Claes I, et al. Screw-thread vs Dumon endoprosthesis in the management of tracheal stenosis. Chest 1999; 115:532. 32. Bolliger CT, Wyser C, Wu X, et al. Evaluation of a new self-expandable silicone stent in an experimental tracheal stenosis. Chest 1999; 115:496. 33. Grillo HC. Development of tracheal surgery: a historical review. Part 1: Techniques of tracheal surgery. Ann Thorac Surg 2003; 75:610. 34. Grillo HC. Development of tracheal surgery: a historical review. Part 2: Treatment of tracheal diseases. Ann Thorac Surg 2003; 75:1039. 35. Bisson A, Bonnette P, el Kadi NB, et al. Tracheal sleeve resection for iatrogenic stenoses (subglottic laryngeal and tracheal). J Thorac Cardiovasc Surg 1992; 104:882. 36. Wright CD, Grillo HC, Wain JC, et al. Anastomotic complications after tracheal resection: prognostic factors and management. J Thorac Cardiovasc Surg 2004; 128:731. 37. Saito Y, Minami K, Kaneda H, et al. New tubular bioabsorbable knitted airway stent: feasibility assessment for delivery and deployment in a dog model. Ann Thorac Surg 2004; 78:1438. 38. Kim J, Suh SW, Shin JY, et al. Replacement of a tracheal defect with a tissueengineered prosthesis: early results from animal experiments. J Thorac Cardiovasc Surg 2004; 128:124. 39. Kojima K, Ignotz RA, Kushibiki T, et al. Tissue-engineered trachea from sheep marrow stromal cells with transforming growth factor beta2 released from biodegradable microspheres in a nude rat recipient. J Thorac Cardiovasc Surg 2004; 128:147. 40. Bugmann P, Rimensberger PC, Kalangos A, et al. Extratracheal biodegradable splint to treat life-threatening tracheomalacia. Ann Thorac Surg 2004; 78:1446.
41. Jaquet Y, Pilloud R, Lang FJ, Monnier P. Prefabrication of composite grafts for long-segment tracheal reconstruction. Arch Otolaryngol Head Neck Surg 2004; 130:1185. 42. Grimmer JF, Gunnlaugsson CB, Alsberg E, et al. Tracheal reconstruction using tissue-engineered cartilage. Arch Otolaryngol Head Neck Surg 2004; 130:1191. 43. Kamil SH, Eavey RD, Vacanti MP, et al. Tissue-engineered cartilage as a graft source for laryngotracheal reconstruction: a pig model. Arch Otolaryngol Head Neck Surg 2004; 130:1048. 44. Martinod E, Seguin A, Holder-Espinasse M, et al. Tracheal regeneration following tracheal replacement with an allogenic aorta. Ann Thorac Surg 2005; 79:942. 45. Cibantos Filho JS, de Mello Filho FV, Campos AD, Ellinguer F. Viability of a 12ring complete tracheal segment transferred in the form of a compound flap: an experimental study in dogs. Laryngoscope 2004; 114:1949. 46. Olias J, Milln G, da Costa D. Circumferential tracheal reconstruction for the functional treatment of airway compromise. Laryngoscope 2005; 115:159. Topic 4384 Version 3.0 2012 UpToDate, Inc. All rights reserved. | Subscription and License Agreement |Release: 20.3 - C20.4 Licensed to: UTPL |Support Tag: [ecapp1005p.utd.com-200.0.29.70-8DA7D5FDCB50339.14]
TOPIC OUTLINE
INTRODUCTION DIAGNOSIS MANAGEMENT Initial stabilization Airway interventions - Foreign body extraction - Rigid or balloon dilation - Laser therapy - Electrocautery and argon plasma coagulation - Photodynamic therapy - Cryotherapy - External beam radiation and brachytherapy - Airway stents - Surgical resection RECOMMENDATIONS REFERENCES
GRAPHICSView All
FIGURES CT reconstruction of trachea Upper airway obstruction Flow volume loops airway obstr Mainstem bronchial obstr Partial mainstem bronchial obs End inspiratory tail Central obstruction types Rigid bronchoscope Dilation with rigid scope Management central obstruction Balloon dilation Juvenile papillomatosis bronch TABLES Causes central airway obstruct Interventions cent obstruction
RELATED TOPICS
Airway foreign bodies in adults Airway stents Basic principles of medical lasers Bronchoscopic cryosurgery: Indications, contraindications, and outcomes Bronchoscopic cryosurgery: Principles and technique Bronchoscopic laser resection Endobronchial brachytherapy Endobronchial electrocautery Endotracheal tube management and complications Flexible bronchoscopy: Equipment, procedure, and complications Flexible bronchoscopy: Indications and contraindications Flexible fiberoptic bronchoscopy balloon dilation Flow-volume loops Nonresolving pneumonia Overview of tracheostomy Photodynamic therapy of lung cancer Radiology of the trachea Rigid bronchoscopy: Instrumentation Rigid bronchoscopy: Intubation techniques
Help improve UpToDate. Did UpToDate answer yo The failed airway in adults Authors Ron M Walls, MD, FRCPC, FAAEM Michael F Murphy, MD, FRCPC
Section Editor John A Marx, MD Deputy Editor Jonathan Grayzel, MD, FAAEM Disclosures All topics are updated as new evidence becomes available and our peer review process is complete. Literature review current through: feb 2012. | This topic last updated: jun 14, 2010. INTRODUCTION A failed airway exists at any time during an attempt at endotracheal intubation when there is an inability to intubate the patient (even with a single attempt) and an inability to ventilate the patient adequately with a bag and mask to maintain oxyhemoglobin saturations above 90 percent. This is the "can't intubate, can't ventilate" type of failed airway. A second form of failed airway has been defined for emergency intubation, and exists when there have been three failed attempts to intubate by an experienced operator, even when bag and mask ventilation is capable of maintaining adequate oxyhemoglobin saturation [1]. This is the "can't intubate, can ventilate" type of failed airway. When either of these two situations arises during emergency airway management, the clinician must take effective action immediately to avoid oxygen desaturation with resultant cerebral hypoxia. A failed airway can arise during a rapid sequence intubation, during management of a difficult airway, or during management of a crash airway. Regardless of the circumstances leading to the airway failure, a deliberate approach must be used to ensure that oxygenation is preserved, and that the airway is ultimately secured. This topic review will discuss management of the failed airway. Discussions of other aspects of airway management are found elsewhere. (See "Rapid sequence intubation in adults" and "The difficult airway in adults" and "The difficult pediatric airway" and "Emergent endotracheal intubation in children".) INCIDENCE OF THE FAILED AIRWAY The incidence of the failed airway, as defined above, is not known for emergency department (ED) airway management, nor for patients undergoing anesthesia in the operating room. Analysis of nearly 9000 ED intubations in the National Emergency Airway Registry (NEAR) found that rapid sequence intubation is successful in approximately 97 percent of cases for which it is the first method chosen, and that surgical cricothyroidotomy (also called cricothyrotomy) is required in approximately 0.8 percent of all emergency intubations and 1.7 percent of intubations of trauma patients [2,3]. An earlier, single center study of 610 emergency intubations noted a cricothyroidotomy rate of 1.1 percent [4]. (See "Emergent surgical cricothyrotomy (cricothyroidotomy)".) It is likely that the incidence of failed airway, particularly of the can't intubate, can ventilate type, is much higher than this. Regardless of the incidence, the failed airway represents a potential catastrophe if not managed effectively. The failed airway scenario is best avoided
by a systematic preintubation evaluation of patients for difficult airway attributes. (See "The difficult airway in adults".) THE FAILED AIRWAY ALGORITHM When a failed airway occurs, the failed airway algorithm provides a series of actions to guide management (algorithm 1) [1,5]. The critical question is whether adequate oxygenation (ie, oxyhemoglobin saturation [SpO2] above 90 percent or stable in the high 80s) can be maintained. If oxygenation is adequate, there is time to plan a series of actions to manage the airway; if the patient's oxygenation cannot be maintained, immediate rescue by (usually) cricothyrotomy is necessary. If time permits, there are a number of alternative or rescue devices that can be used when direct laryngoscopy has failed. In some cases, the rescue airway may provide a definitive airway, with a cuffed endotracheal tube (ETT) in the trachea. If the airway is secured by a cuffed ETT, the airway is considered to be managed, and general resuscitation continues. If adequate oxygenation and ventilation are achieved, but the airway is not protected by a cuffed ETT in the trachea, resuscitation can continue, but arrangements must be made to establish a definitive airway at the earliest appropriate opportunity. As with the difficult airway algorithm, the response to the failed airway can be thought of as a series of discrete steps. Each of these steps is described in detail below. Assistance in the form of personnel, equipment, or airway devices, should be obtained as needed at the moment the failed airway is recognized. Is there time? At the outset, the key determination is whether the patient is being adequately oxygenated (ie, oxyhemoglobin saturation [SpO2] above 90 percent or stable in the high 80s). If so, there is time to create and execute a deliberate rescue plan, perhaps customized to the patient's particular circumstances. This is the "can't intubate, can ventilate" scenario. If the patient cannot be oxygenated adequately with a bag and mask, despite use of optimal technique (ie, can't intubate, can't oxygenate), immediate cricothyrotomy is indicated. Although an alternative airway device might rescue the patient without cricothyrotomy, the extremely brief time before cerebral hypoxia ensues argues for immediate surgical intervention. Attempts to establish an alternative airway, if unsuccessful, may delay the initiation of cricothyrotomy leading to hypoxic brain injury. Thus, we equate "can't intubate, can't ventilate" with cricothyrotomy in the emergency setting. There is one modification to this approach. Placement of a single "best" alternative device, usually an extraglottic airway (eg, Laryngeal Mask Airway (LMA) or King LT Airway) can be attempted in parallel with preparations for the surgical airway. If the operator is able to insert an extraglottic airway and attempt ventilation, while a second clinician simultaneously prepares for a cricothyrotomy, then valuable time is not lost if ventilation using the extraglottic device is unsuccessful. Similarly, a single operator might make one attempt with an extraglottic device, then proceed directly to cricothyrotomy if ventilation is not promptly achieved. This approach
involves a single attempt using a single "go to" device, and cannot delay significantly the initiation of cricothyrotomy if ventilation is not achieved immediately. Whether this single, parallel rescue maneuver is attempted but unsuccessful or not attempted, the primary rescue technique is cricothyrotomy. The performance of cricothyrotomy is discussed separately. (See "Emergent surgical cricothyrotomy (cricothyroidotomy)".) ALTERNATIVE AIRWAY DEVICES The initial goal for failed airway management is to provide adequate oxygenation sufficient to support the patient until a definitive airway can be achieved. So long as the patient is adequately oxygenated using a bag and mask, the clinician may use any of several alternatives to direct laryngoscopy to rescue the failed airway. The devices briefly described below are discussed in detail separately. (See "Devices for difficult emergency airway management in adults".) List of devices
Fiberoptic or video stylet - There are several rigid or semi-rigid fiberoptic and video stylets available (eg, Shikani Optical Stylet [SOS] or Storz Bonfils laryngoscope). These stylets have fiberoptic or video viewers at their distal end and are inserted through the endotracheal tube (ETT). The image generated can be used to guide the ETT between the vocal cords without need of a conventional laryngoscope. Video laryngoscopes - Video laryngoscopes (eg, Glidescope or Storz C-MAC) contain video cameras on the blade that generally provide an excellent view of the glottis, without the need to align the airway axes to achieve a direct view from outside the patient's mouth. These devices function well with the patient in the neutral position, and obstacles to conventional laryngoscopy, such as limited mouth opening or a large tongue, generally do not present a problem. Optical devices Various optical devices are available for intubation of the failed airway or as an alternative to direct laryngoscopy. The best studied of these is the AirTraq, a periscope-like device that uses prisms and mirrors to provide an indirect view of the glottis. The AirTraq incorporates a channel for the ETT, which is advanced when the glottis is properly sighted. Extraglottic airway - Various laryngeal mask airways (LMAs) are available, some specifically designed to facilitate subsequent intubation, some intended strictly as ventilatory devices in their own right. A second type of extraglottic airway is inserted into the esophageal inlet and has two balloons that are inflated to occlude both the esophagus and the pharynx thereby permitting sidestream ventilation of the trachea. Available devices include the Combitube and the King LT airways. Flexible bronchoscope - The flexible bronchoscope, usually an intubating bronchoscope, provides access to the glottis without having to correct for the various angles of the oropharynx, as is required for direct laryngoscopy. Intubation can be achieved nasally or orally, but by either route, attempts can be timeconsuming, making this device more appropriate for a planned approach to certain difficult airways, rather than for the rescue of a failed airway. Attempts using the fiberoptic bronchoscope may have to be abbreviated or abandoned because of the difficulty maintaining oxygenation during fiberoptic airway procedures. Flexible
bronchoscopes require training and practice, but have high success rates when time permits [6]. Was a cuffed endotracheal tube placed in the trachea? Many of the alternative airway devices listed above result in a cuffed ETT in the trachea, in which case, the airway is secured. Others, such as the extraglottic airways, provide for ventilation and oxygenation, but do not protect the airway. If one of the nonprotective devices has been used, resuscitation can proceed, but a plan must be initiated to secure the airway at the earliest opportunity. If at any time oxygenation fails, and the patient reverts to a can't intubate, can't ventilate situation, cricothyrotomy remains the rescue technique of first resort. SUMMARY AND RECOMMENDATIONS
A failed airway exists at any time during an attempt at endotracheal intubation when there is an inability to intubate the patient (even with a single attempt) and an inability to ventilate the patient adequately using a bag and mask or an extraglottic device (ie, maintain oxyhemoglobin saturations [SpO2] above 90 percent or stable in the high 80s). This is the "can't intubate, can't ventilate" type of failed airway. A failed airway also exists when there have been three failed attempts to intubate by an experienced operator, even when ventilation with a bag and mask or an extraglottic device maintains adequate SpO2. This is the "can't intubate, can ventilate" type of failed airway. (See 'Introduction' above.) We recommend that cricothyrotomy be used as the primary rescue maneuver for the "can't intubate, can't ventilate" failed airway (Grade 1C). Cricothyrotomy has a high success rate and relatively low complication rate, and can be performed using open surgical technique or a Seldinger method. (See 'The failed airway algorithm' above.) The critical question in failed airway management is whether adequate oxygenation (ie, SpO2 above 90 percent or stable in the high 80s) can be maintained. If oxygenation is adequate there is time to plan a series of actions to manage the airway; if the patient's oxygenation cannot be maintained, immediate rescue by (usually) cricothyrotomy is necessary. (See 'The failed airway algorithm' above and "Emergent surgical cricothyrotomy (cricothyroidotomy)".) For the can't intubate, can ventilate patient, there are several possible rescue devices. (See 'Alternative airway devices' above.) For ventilation only: Extraglottic airway (eg, Combitube, King LT, Laryngeal mask airway)
For intubation:
Intubating LMA Rigid fiberoptic or video stylet Video laryngoscope Optical device (eg, AirTraq) Flexible fiberoptic scope
Use of UpToDate is subject to the Subscription and License Agreement.
REFERENCES1. Walls, RM. The Emergency Airway algorithms. In: Manual of Emergency Airway Management, Walls, RM, Murphy, MF, Luten, RF, et al (Eds). Philadelphia, Lippincott, Williams and Wilkins 2004. p 8. 2. Sagarin MJ, Barton ED, Chng YM, et al. Airway management by US and Canadian emergency medicine residents: a multicenter analysis of more than 6,000 endotracheal intubation attempts. Ann Emerg Med 2005; 46:328. 3. Walls, RM, et al. Emergency Airway Management: A Multi-center Report of 8937 Emergency Department Intubations. J Emerg Med 2010; [Epub ahead of print]. 4. Sakles JC, Laurin EG, Rantapaa AA, Panacek EA. Airway management in the emergency department: a one-year study of 610 tracheal intubations. Ann Emerg Med 1998; 31:325. 5. The airway management algorithms cited in this review are reproduced with permission from: The Difficult Airway Course: Emergency, and Walls, RM, Murphy, MF. Manual of Emergency Airway Management, 3rd ed, Lippincott Williams & Wilkins, Philadelphia 2008. 6. Dunn S, Connelly NR, Robbins L. Resident training in advanced airway management. J Clin Anesth 2004; 16:472. Topic 274 Version 3.0 The difficult pediatric airway Author Nathan W Mick, MD Section Editor Susan B Torrey, MD Deputy Editor James F Wiley, II, MD, MPH Disclosures All topics are updated as new evidence becomes available and our peer review process is complete. Literature review current through: feb 2012. | This topic last updated: oct 12, 2011. INTRODUCTION Effective airway management includes anticipating and planning for problems. Difficulties frequently occur as the result of patient characteristics that interfere with spontaneous breathing, bag mask ventilation, laryngoscopy, and/or intubation of the trachea. Identifying characteristics of the difficult airway and developing a plan for managing problems are essential principles of anesthesia practice [1]. These principles have been modified and effectively used to evaluate adults in the emergency department [2]. Children infrequently require aggressive airway management and difficulties do not occur often [3]. As a result, evidence specific for children regarding identification and management of difficult airways is limited [4]. Nevertheless, a reasonable, systematic approach for children can be developed from experience with adult patients in the operating room and emergency department.
This topic will review conditions in children that can make airway management difficult, anatomic characteristics that may identify those conditions, and management strategies. General principles of airway management in children, including rapid sequence intubation, and the adult with a difficult airway are discussed separately. (See "Basic airway management in children" and "Emergent endotracheal intubation in children" and "Rapid sequence intubation in children" and "The difficult airway in adults".) DEFINITION A difficult airway is generally defined as a situation in which a clinician experiences difficulty with face mask ventilation, laryngoscopy, or intubation [1,2]. In an emergency setting, this also includes difficulty performing an emergency surgical airway, such as needle cricothyroidotomy. These difficulties may arise whenever any of the following maneuvers cannot be successfully performed:
Positioning to optimally align the pharyngeal and tracheal axes (picture 1). Achieving sufficient positive pressure with a resuscitation bag to inflate the lungs. Opening the mouth and controlling oral structures with the laryngoscope blade. Visualizing the larynx and intubating the trachea. Identifying landmarks in the neck for performing a surgical airway.
CAUSES OF THE DIFFICULT PEDIATRIC AIRWAY Normal pediatric airway Predictable differences in the pediatric airway (as compared with adult airway anatomy) may make management difficult. These differences, most evident in children less than two to three years of age, include the following (see "Emergency airway management in children: Unique pediatric considerations"):
A large occiput affects positioning. A large tongue and small mouth may make laryngoscopy difficult. The larynx may be harder to locate with the laryngoscope because it is higher and more anterior than in an adult. The epiglottis is large and floppy and may difficult to control.
Anatomic features of the normal pediatric airway are reviewed elsewhere. (See "Basic airway management in children", section on 'Anatomic considerations' and "Emergent endotracheal intubation in children".) Congenital abnormalities Numerous congenital conditions have features that may make airway management difficult (table 1). In addition, children with underlying airway abnormalities who acquire an acute condition (such as croup or an upper respiratory tract infection) may quickly develop respiratory compromise. Congenital features that may interfere with airway management include the following:
Misshapen head Positioning of the head to optimally align the pharyngeal and tracheal axes may be difficult if the head is misshapen, as can occur with
craniosynostosis (such as Apert's or Crouzon's syndrome) or hydrocephalus. (See "Craniosynostosis syndromes" and "Hydrocephalus".) Facial abnormalities Facial asymmetry or underdevelopment may make it difficult to achieve a good seal between the face and a mask, creating difficulties with bag mask ventilation (picture 2). As examples, maxillary hypoplasia is a feature of Apert's syndrome, while Goldenhar syndrome includes unilateral hypoplasia of the mandible. (See "Syndromes with craniofacial abnormalities".) Abnormal neck mobility Limited neck mobility (as occurs with Klippel- Feil syndrome) or cervical spine instability (which can occur with Down syndrome and the mucopolysaccharidoses) may interfere with positioning of the head. (See "Clinical features and diagnosis of Down syndrome", section on 'Atlantoaxial instability' and "Complications and management of the mucopolysaccharidoses", section on 'Anesthesia'.) Small oral aperture Opening the mouth for laryngoscopy may be difficult in children with microstomia, which is a feature of Freeman-Sheldon and HallermannStrieff syndromes. Small oral cavity Children with small mandibles or palatal abnormalities (such as high arched or cleft palates) have a smaller oral cavity. This may make laryngoscopy and control of oral structures difficult. For instance, mandibular hypoplasia is a feature of the Robin sequence and Treacher Collins' syndrome (picture 3). Large tongue A large tongue may obstruct the airway during bag mask ventilation or be difficult to control during laryngoscopy. Macroglossia occurs in several conditions (such as hypothyroidism, Beckwith-Wiedeman syndrome, and Down syndrome). It is also a feature of infiltrative diseases such as the mucopolysaccharidoses. Masses Masses in the neck (such as cystic hygromas) may interfere with positioning. Masses within the airway (such as teratomas or hemangiomas) may obstruct the airway and interfere with visualization of the larynx. Mediastinal masses may make tube placement difficult and interfere with ventilation after successful intubation [5]. (See "Congenital anomalies of the jaw, mouth, oral cavity, and pharynx" and "Epidemiology; pathogenesis; clinical features; and complications of infantile hemangiomas", section on 'Airway hemangiomas'.) Laryngeal and subglottic abnormalities Abnormalities of the larynx or subglottic trachea may interfere with intubation (picture 4).
Acquired conditions Acquired conditions that can cause difficulties with airway management may develop as the result of infection, allergic reactions, trauma, or aspiration of a foreign body. Infection The specific difficulties in airway management that arise because of Infection depend upon where the infection is located within the airway.
Retropharyngeal and peritonsillar abscesses may interfere with laryngoscopy and visualization of the larynx. These conditions do not typically require emergency airway management. (See "Retropharyngeal infections in children".)
Epiglottitis is characterized by rapidly progressive inflammation and edema of the supraglottic structures. Airway management is difficult because laryngeal anatomy is distorted and the glottic opening may be small and difficult to identify. Since the introduction of conjugate Haemophilus influenza type B vaccines, the incidence of epiglottitis in children has declined dramatically. (See "Epiglottitis (supraglottitis): Clinical features and diagnosis".) Croup and tracheitis cause subglottic airway obstruction. As a result, it may be difficult to deliver effective bag mask ventilation or to pass an endotracheal tube through the subglottic trachea. Fortunately, both conditions generally respond to medical management. (See "Clinical features, evaluation, and diagnosis of croup", section on 'Pathogenesis' and "Approach to the management of croup".)
Anaphylaxis Edema involving the tongue, retropharynx, and/or larynx, that can interfere with laryngoscopy and visualization of the larynx, may develop as the result of anaphylaxis. Symptoms typically respond to aggressive medical management (table 2). Trauma Injury to the face or airway (as the result of blunt or penetrating trauma, thermal burns, or caustic ingestions) may complicate airway management. Facial burns may make it difficult to deliver bag mask ventilation because an adequate seal between the mask and face cannot be achieved. An expanding hematoma in the pharynx can interfere with laryngoscopy. Injury to the larynx or subglottic trachea may be exacerbated by intubation. Spinal immobilization with a rigid cervical collar, regardless of the presence of injury can make direct laryngoscopy difficult because the clinician is unable to optimally position the patient in the sniffing position and adequately align the visual axis. (See 'Management' below.) Foreign body A foreign body in the airway may cause significant obstruction and require immediate treatment. Identification and removal of the foreign body during laryngoscopy can be challenging. In addition, normal anatomic landmarks may be distorted by the foreign body. (See "Emergent evaluation of acute upper airway obstruction in children", section on 'Causes'.) Piercings around the mouth and tongue may interfere with or become dislodged during laryngoscopy [6]. Other causes Other acquired conditions in children that may make airway management difficult include tumors, previous surgery, or radiation treatment. IDENTIFICATION OF THE DIFFICULT PEDIATRIC AIRWAY The initial evaluation of any critically ill or injured child should include a brief, systematic assessment of the airway to identify characteristics that may complicate management. These characteristics must be taken into consideration when developing an airway management plan. (See "Emergent endotracheal intubation in children" and "Rapid sequence intubation in children", section on 'Preparation'.) Anesthesiologists have used bedside evaluation tools to identify patients for whom airway management may be difficult [1]. None of these indicators has been tested in emergency
departments or in children. Nevertheless, a reasonable approach can be developed using evidence from the operating room and clinical experience in the emergency department to identify children who may have difficult airways [2]. Bag mask ventilation Bag mask ventilation may be difficult in children with the following features:
A misshapen head (as the result of trauma or a congenital anomaly) or limited neck mobility (such as a patient whose cervical spine is immobilized) can interfere with proper positioning (picture 1). Facial burns or any disruption of lower facial continuity (as can occur with facial trauma or a congenital anomaly with facial asymmetry) can make it difficult to achieve an adequate seal between the face and the mask. Patients who are obese or who have significant lung disease (such as severe asthma) may be difficult to ventilate with a bag and mask [7].
Laryngoscopy or intubation A combination of several clinical features appears to be a sensitive predictor of difficult laryngoscopy or intubation for adults [8-10]. These features include:
Interincisor gap is the distance between the upper and lower incisors with the mouth open as wide as possible. For adult patients, the width of three of the patient's fingers is considered an adequate distance for laryngoscopy [2]. Mallampati score assesses the view of the posterior pharynx with the mouth wide open (figure 1). Intubation may be difficult for patients with a poor view (Class III or IV). However, when used alone, the score has limited accuracy for predicting a difficult airway [11]. Thyromental distance is the distance between the tip of the chin and the thyroid notch. Typically, the width of three of the patient's fingers is considered normal for adults [2]. Difficulty visualizing the larynx may occur when the distance is longer or shorter.
Cricothyroidotomy Needle cricothyroidotomy, which permits percutaneous transtracheal ventilation, should always be considered a difficult technique in children because normal landmarks are difficult to identify and the caliber of the airway is small. In addition, few if any practitioners are able to gain proficiency with these techniques because clinical scenarios that require them occur rarely. (See 'Surgical airway' below.) The LEMON approach to difficult airway assessment The mnemonic LEMON has been developed by researchers in emergency airway management as a tool for rapidly identifying adult patients who may have a difficult airway (table 3) [2,12]. The tool has not been tested in children. Components of the mnemonic include the following:
L: Look externally for indicators of a difficult airway (such as a misshapen head, facial abnormalities, or neck masses). E: Evaluate mouth opening, thyromental distance, and the distance between the mandible and the thyroid cartilage (this correlates with the distance between the
base of the tongue and the larynx) (figure 2). Adequate mouth opening and thyromental distance should be the width of three of the patient's fingers. The distance between the mandible and thyroid cartilage should be the width of two fingers. M: Mallampati score: Assigning a Mallampati score may be difficult in young children. For the obtunded, supine patient, a crude assessment can be made using a tongue blade (figure 1) [2]. O: Obstruction: Signs of airway obstruction (such as stridor, a muffled voice, or difficulty handling secretions) always indicate that airway management may be difficult. Upper airway obstruction can interfere with bag mask ventilation, as well as with laryngoscopy and intubation. N: Neck mobility: Conditions that limit neck mobility (such as congenital anomalies or cervical spine immobilization) can usually be identified by observation.
ALTERNATIVE AIRWAY TECHNIQUES Alternative strategies for providing oxygenation and ventilation must be considered for the child who may be difficult to intubate with direct laryngoscopy. These techniques may be temporizing (such as laryngeal mask airway, combitube, or a percutaneous needle cricothyrotomy) or provide alternative approaches to tracheal intubation (as with fiberoptic intubation or a lighted stylet). Several factors impact the choice of device including the clinical situation, type of airway difficulty, and experience of the operator. (See "Devices for difficult endotracheal intubation in children", section on 'Choice of device' and "Emergency rescue devices for difficult pediatric airway management", section on 'Choice of device'.) Laryngeal mask airway The laryngeal mask airway (LMA) consists of a cuffed mask, designed to fit over the larynx, which is attached to a tube similar to an endotracheal (ET) tube. The LMA is inserted into the mouth and blindly passed along the palate into the posterior pharynx until resistance is met. The cuff is then inflated and the mask forms a partial seal around the larynx. Positive pressure ventilation can then be delivered through the tube. The procedure for placing the LMA in children is described in detail separately. (See "Emergency rescue devices for difficult pediatric airway management", section on 'Laryngeal mask airway (LMA)'.) The device is available in multiple sizes suitable for infants, children, and adults. The appropriate size is based on the patient's weight (table 4). The LMA has been used extensively by anesthesiologists in the operating room for children with normal and difficult airways [13-16]. It is relatively easy to insert, although complications with its use have been reported in infants and small children [17]. It has been used as a primary airway and as an adjunct for fiberoptic tracheal intubation [18,19]. (See 'Flexible fiberoptic intubation' below.) The LMA may provide effective airway management for adults during resuscitation [20]. In addition, successful oxygenation and ventilation with an LMA have been described in adults with upper airway obstruction from supraglottic edema [21]. However, experience with the device as a rescue airway in children is limited [22]. Case series and reports suggest that an adequate airway can be achieved with an LMA in neonates when bag-mask ventilation and tracheal intubation have failed [23].
An LMA with a modified cuff (Proseal LMA) appears to provide a better seal over the larynx for children, allowing for more effective delivery of positive pressure ventilation [24,25]. The device also has an esophageal drainage tube through which a gastric tube can be placed to empty the stomach. It is available in the full range of pediatric sizes. Intubating introducers (gum elastic bougie) Intubating introducers are helpful when the epiglottis is visible but the vocal cords cannot be seen. These devices are semi-rigid solid or hollow rods with the distal tip bent at a 30 degree angle (figure 3 and figure 4). Pediatric sized introducers allow placement of endotracheal tubes as small as 4.0 mm (internal diameter). Lighted stylet The pediatric lighted stylet is a rescue device that does not require direct visualization of the vocal cords [26]. A stylet with a fiberoptic light source is inserted into an endotracheal tube that is then blindly placed into the posterior pharynx and advanced. The tube is usually located in the trachea when a cherry-red glow is noted at the suprasternal notch. The stylet can then be carefully removed, endotracheal tube placement confirmed, and the tube secured. This technique has been used successfully for infants and children but has a relatively low success rate (75-83%) when the technique is performed by inexperienced users [27,28]. It can often be performed with minimal movement of the patient's head and neck, making it particularly useful for trauma patients whose cervical spines are immobilized [29]. (See "Devices for difficult endotracheal intubation in children", section on 'Lighted stylet'.) Fiberoptic stylets These devices combine features of the lighted stylet with features of a flexible fiberoptic bronchoscope to create a device that can be used for blind intubation similar to a lighted stylet or visually guided intubation. With this technique an endotracheal tube is loaded onto a stylet with a fiberoptic lens that allows the clinician to visualize the glottis. The endotracheal tube is then threaded through the glottis and secured. (See "Devices for difficult endotracheal intubation in children", section on 'Fiberoptic stylets'.) Flexible fiberoptic intubation Flexible fiberoptic techniques have been used extensively by anesthesiologists for difficult intubations [1,19]. Experience with this approach in the emergency department is almost exclusively in adult patients [30]. Typically, an endotracheal tube is threaded onto the end of a flexible fiberoptic bronchoscope. The scope is then introduced into the nose or mouth. The trachea is visualized and intubated with the scope and endotracheal tube. The scope is then withdrawn, placement of the endotracheal tube in the trachea is confirmed, and the tube is secured. (See "Devices for difficult endotracheal intubation in children", section on 'Flexible fiberoptic bronchoscope'.) Fiberoptic intubation should be considered in cases where the pre-intubation assessment suggests that orotracheal intubation via RSI (preferred in most emergency department intubation scenarios) is unlikely to be successful. Examples include congenital airway anomalies such as micrognathia or conditions where difficulty aligning the oral, pharyngeal, and laryngeal axes is predicted (such as when neck mobility is limited). The small size of the nasal passages in very young children may preclude the nasal route for intubation.
In the hands of an experienced clinician, flexible fiberoptic laryngoscopy is an excellent method for endotracheal intubation for a patient with a difficult airway who is breathing spontaneously. Availability of equipment and experienced personnel, as well as time considerations, are usually the limiting factors for using this technique for emergency airway management. Video laryngoscopy Video laryngoscopes provide indirect laryngoscopy and display the glottic view on a video monitor during endotracheal intubation. Several devices are available in sizes appropriate for infants and children. (See "Devices for difficult endotracheal intubation in children", section on 'Video laryngoscope'.) Combitube The Combitube is a dual-cuff, dual-lumen tube that is placed blindly in the esophagus. The distal balloon is designed to occlude the esophagus, while the proximal balloon will occlude the hypopharynx. Positive pressure ventilation can be delivered through side ports when the tube is in the esophagus, or through the tip when it is placed in the trachea, which rarely occurs. Minimal training is required to use the device effectively, and it can be placed quickly, with minimal movement of the cervical spine [31]. The Combitube does not provide a definitive airway. In addition, it is only available in sizes appropriate for patients taller than 48 inches (1.2 m) [32]. (See "Emergency rescue devices for difficult pediatric airway management", section on 'Combitube'.) Complications may occur more commonly with the Combitube than with an LMA [33]. Reported complications with the Combitube include esophageal rupture, pyriform sinus perforation, and tongue engorgement [34-36]. Mucosal ischemia may occur as the result of pressure from the balloons [37]. Surgical airway Rarely, noninvasive rescue devices fail to provide an airway. As a result, emergency healthcare providers should be familiar with surgical airway techniques, such as needle or surgical cricothyroidotomy, although in reality few if any practitioners have enough opportunity with these approaches to gain proficiency. Surgical cricothyroidotomy should be avoided in infants and young children. Equipment required for needle and surgical cricothyroidotomy, particularly a setup for delivering transtracheal ventilation, should be organized in advance and readily available in locations where emergency airway procedures are performed. Step by step instructions on how to perform needle or surgical cricothyroidotomy and on how to perform percutaneous transtracheal ventilation are discussed separately. (See "Needle cricothyroidotomy with percutaneous transtracheal ventilation" and "Emergent surgical cricothyrotomy (cricothyroidotomy)".) MANAGEMENT Anticipating and preparing for advanced airway management, including intubation, for a critically ill or injured child who may have a difficult airway should begin before the patient arrives in the emergency department. Emergency departments should have equipment and supplies available in a readily identifiable location, such as a "difficult airway box". (See 'Alternative airway techniques' above.)
Once the child with a difficult airway is identified, a specific plan for management must be developed that includes mobilizing appropriate personnel and assembling specialized equipment (algorithm 1 and algorithm 2 and algorithm 3). The child may improve with supportive care and aggressive treatment of the underlying condition. For children who require intubation, airway management must include a rescue plan and preparation for a failed airway (algorithm 4). Supportive care Care for all patients with respiratory difficulties who may have a difficult airway should include the following:
Provide supportive care and careful monitoring. Children who are developing respiratory compromise must be rapidly identified. (See "Initial assessment and stabilization of children with respiratory or circulatory compromise".) Aggressively treat the underlying condition. As an example, a patient with a congenitally abnormal airway who develops croup should quickly receive nebulized epinephrine and corticosteroids. In most cases, the child's condition will improve and advanced airway management will not be necessary. (See "Approach to the management of croup".) Avoid situations that could worsen airway compromise. As an example, a child with a retropharyngeal abscess who requires sedation for imaging studies should receive reversible agents, whenever possible. (See "Procedural sedation and analgesia in children".) Anticipate the need for advanced airway management. Children with conditions that rapidly and predictably progress to involve edema and distortion of normal airway anatomy despite aggressive medical management (such as thermal or chemical airway burns) should be intubated early, in as controlled a setting as possible.
Airway management In the case of a predicted difficult airway, the first intervention should be to call for help if such help is available. The most expert physician available may be from anesthesia or otorhinolaryngology rather than emergency medicine or pediatrics, and they may provide valuable assistance in the rare case of a difficult pediatric airway. An approach to management decisions should consider the urgency of establishing an airway ("crash airway") and the likelihood that rapid sequence intubation (RSI) will be successful. In comparison to adults, fewer interventions are available for children who have a failed airway. Crash airway Children who are in extremis are considered a "crash airway" and should receive bag mask ventilation, followed by orotracheal intubation (algorithm 3). Numerous studies have demonstrated that effective BMV, especially in the prehospital arena, is an effective means of supporting respirations [38,39]. BMV may provide oxygenation and ventilation as personnel and equipment are being mobilized for endotracheal intubation, even in situations with significant soft tissue obstruction such as epiglottitis [40]. Alternative airway techniques (such as laryngeal mask airway or needle cricothyrotomy) should be employed when attempts to intubate the trachea are unsuccessful. (See 'Alternative airway techniques' above and 'Approach to the failed airway' below.)
Rapid sequence intubation Rapid sequence intubation should be considered for children who are not in extremis when the clinician is confident that the child can be adequately ventilated with a bag and mask and that oral tracheal intubation will be successful (algorithm 1). Preparations should always be made for alternative airway management (such as a laryngeal mask airway). (See "Rapid sequence intubation in children" and 'Alternative airway techniques' above.) Awake intubation Awake intubation, using sedation and local anesthesia, is an approach that is frequently used for adults [41]. With this technique, the patient is sedated but not paralyzed and continues to breath spontaneously. There are no reports describing experience with this technique for children in the emergency department. It is likely that the degree of sedation required to perform awake intubation in a frightened young child would depress airway protective reflexes and spontaneous respiration, placing the patient at risk for aspiration and hypoxia. Alternative airway techniques Alternatives for airway management when RSI or awake intubation are not feasible include a laryngeal mask airway or fiberoptic intubation. (See 'Alternative airway techniques' above and 'Approach to the failed airway' below.) Approach to the failed airway A child with respiratory failure for whom bag mask ventilation is not effective and the trachea cannot be intubated has a failed airway. This situation is often referred to as a "can't ventilate, can't intubate" scenario [2]. Prompt intervention to improve oxygenation and ventilation is essential (algorithm 4). The most expert clinician available should be managing the airway. There is no evidence to guide recommendations for management of these rare, but lifethreatening, situations. Therefore, any intervention that could be possibly helpful and is unlikely to worsen the patient's condition, should be considered. An LMA should be used initially for most children with failed airways who do not have complete airway obstruction. Positive pressure ventilation through a device that is sealed around the larynx may be effective for those with partial airway obstruction, even if the obstruction is subglottic (such as with croup or a subglottic foreign body). Temporary improvement in oxygenation may be life-saving, while other interventions are implemented. A surgical airway should be performed for a child with a complete upper airway obstruction. Needle cricothyroidotomy is recommended for children 10 years of age. A surgical airway should also be considered for children with airway conditions that could be worsened by injury from attempts to place an LMA (such as expanding hematomas, significant midface trauma, or large abscesses). (See "Needle cricothyroidotomy with percutaneous transtracheal ventilation".) SUMMARY AND RECOMMENDATIONS Effective airway management includes anticipating and planning for difficulties. A reasonable, systematic approach for children can be developed from experience with adult patients.
Problems with airway management can occur with positioning, positive pressure ventilation, laryngoscopy, visualizing and/or intubating the trachea, or identifying landmarks for performing a surgical airway. (See 'Definition' above.) Conditions in children that may make airway management difficult include characteristics of the normal airway and congenital or acquired conditions. (See 'Causes of the difficult pediatric airway' above.) Airway characteristics that may identify a difficult airway can be rapidly assessed using the mnemonic LEMON (table 3). (See 'The LEMON approach to difficult airway assessment' above.) L: Look externally for indicators of a difficult airway E: Evaluate mouth opening, thyromental distance, and the distance between the mandible and the thyroid cartilage (figure 2) M: Mallampati score (figure 1) O: Obstruction: Signs of airway obstruction N: Neck mobility Alternative airway techniques that may be useful for managing a difficult airway include a laryngeal mask airway (LMA), fiberoptic laryngoscopy, video laryngoscopy, a lighted stylet, a combitube, or performing a surgical airway. (See 'Alternative airway techniques' above.) General management issues for all patients include providing supportive care, monitoring, treating the underlying condition, avoiding situations that could worsen airway compromise, and anticipating the need for advanced airway management. (See 'Management' above.) Once the child with a difficult airway is identified, a specific plan for management must be developed that includes mobilizing appropriate personnel and assembling specialized equipment (algorithm 1 and algorithm 2 and algorithm 3). The child may improve with supportive care and aggressive treatment of the underlying condition. For children who require intubation, airway management must include a rescue plan and preparation for a failed airway (algorithm 4). We suggest that an LMA be used as the initial rescue device for a child with a failed airway who does not have complete upper airway obstruction or an airway condition that could be worsened by injury from attempts to place an LMA (Grade 2B). (See 'Approach to the failed airway' above.) A surgical airway is the only option for a child who has a failed airway with complete upper airway obstruction or an airway condition that could be worsened by injury from attempts to place an LMA. Children 10 years of age should receive a needle cricothyrotomy. For children who are older than 10 years, the surgical approach may be dictated by experience of the clinician. (See 'Approach to the failed airway' above and "Needle cricothyroidotomy with percutaneous transtracheal ventilation".) We suggest that oxygen be delivered from a low pressure source when a needle cricothyrotomy is performed (Grade 2C). Jet ventilation is unlikely to improve ventilation and the risk of injury may be significant.
Use of UpToDate is subject to the Subscription and License Agreement.
Emergency airway management in the adult with direct airway trauma Authors Trevor J Mills, MD, MPH Peter DeBlieux, MD Section Editor Ron M Walls, MD, FRCPC, FAAEM Deputy Editor Jonathan Grayzel, MD, FAAEM Disclosures All topics are updated as new evidence becomes available and our peer review process is complete. Literature review current through: feb 2012. | This topic last updated: feb 9, 2012. INTRODUCTION Airway management in patients who have sustained direct trauma to the airway is among the most challenging problems for emergency clinicians. Blunt or penetrating injuries to the head, oropharynx, neck, or upper chest can result in immediate or delayed airway obstruction. Immediate, definitive airway management is needed when the patient cannot protect their airway or is unable to adequately oxygenate or ventilate. Emergent or urgent airway management is indicated when a patient develops respiratory distress or when symptoms are progressing rapidly. In addition, airway management often is indicated when the patient appears clinically stable, but the clinician anticipates clinical decline (eg, smoke inhalation, edema, subcutaneous air, hematoma) or feels that an unprotected airway presents a risk to the patient who requires transport to another facility or to radiology for extensive diagnostic studies. The higher rate of complicated airways in this population mandates that the clinician be prepared to use advanced airway techniques, including a surgical airway. Airway assessment and management in adults with direct airway trauma will be reviewed here. Other aspects of airway management, including a general approach to the difficult airway, the decision to intubate, and advanced tools for airway management, are discussed separately. (See "The difficult airway in adults" and "Rapid sequence intubation in adults" and "Devices for difficult emergency airway management in adults" and "The decision to intubate" and "Basic airway management in adults".) The general management of trauma to the head and neck is also discussed separately. (See "Facial trauma in adults" and "Penetrating neck injuries" and "Skull fractures in adults".) CAUSES OF AIRWAY TRAUMA Common causes of direct airway trauma include the following:
Blunt or penetrating maxillofacial injury Blunt or penetrating neck injury Smoke inhalation or facial burns Caustic ingestion
Face and neck injuries from blunt or penetrating trauma can cause severe bleeding into the oropharynx, expanding hematomas within soft tissue, and disruption of bone and soft tissue. Smoke inhalation, burns of the face and oropharynx, and caustic ingestions are all capable of causing mucosal injury and severe swelling and edema within the oropharynx, larynx, and tracheobronchial tree. It is crucial that emergency airway managers recognize that such injuries are dynamic and that conditions can deteriorate quickly. Hematomas and soft tissue swelling can expand rapidly, converting a partially obstructed airway into a completely obstructed airway. The general management of the injuries listed here is discussed separately. (See "Facial trauma in adults" and "Penetrating neck injuries" and "Smoke inhalation" and "Emergency care of moderate and severe thermal burns in adults" and "Caustic esophageal injury in adults".) AIRWAY ASSESSMENT Determining the need for immediate intervention The first step in managing patients with direct airway trauma is to rapidly assess the patient and their airway to determine whether a definitive airway is needed emergently. Unresponsive patients and those with inadequate respiratory function are intubated during or immediately following evaluation. Patients in obvious respiratory distress also require prompt intubation. This includes patients struggling to breathe because of their injuries and those who have sustained severe burns of the face or who demonstrate blistering or edema of the oropharynx. Patients incapable of protecting their airway, as demonstrated, for example, by inability to clear debris from the oropharynx (eg, teeth, bone fragments, foreign bodies, emesis), also require prompt intubation. A simple assessment consisting of four basic questions often distinguishes patients requiring intubation from those who may be observed. An affirmative answer to any of the following questions identifies the need for intubation in nearly all scenarios involving direct airway trauma:
Is there failure of airway maintenance or protection? Is there failure of ventilation? Is there failure of oxygenation? Is deterioration, particularly of the airway, anticipated? (ie, What is the expected clinical course?)
This approach to intubation and management of the failed airway is discussed in detail separately. (See "The decision to intubate" and "The failed airway in adults".) Direct trauma to the airway can cause conditions that deteriorate precipitously leading to complete airway obstruction. Examples include expanding hematomas following blunt or penetrating trauma and soft tissue swelling following smoke inhalation or caustic ingestion. Of note, the progression of an airway injury, such as a soft tissue hematoma, may involve the deep tissue planes of the neck and therefore not become clinically apparent until airway
obstruction is nearly complete and the chance for successful intervention is slim. Therefore, the risk of rapid airway compromise is a common and important reason for early intubation in patients with direct airway trauma. Signs of airway compromise In patients with direct trauma to the face, neck, or upper chest who do not have a crash airway, the clinician performs a careful examination looking for signs of airway compromise. These signs may include any of the following:
Direct signs of airway compromise: Dyspnea Stridor Indirect signs of airway compromise: Drooling Trismus Painful swallowing (odynophagia) Tracheal deviation or other anatomical abnormality involving the larynx or trachea Signs of developing airway compromise: Nonsuperficial burns of the face or neck Severe bleeding in the oropharynx or nasopharynx Subcutaneous air (crepitus) in the neck or upper chest Hematoma in the neck or lower face Hoarseness or other alterations in voice Subjective sense of shortness of breath despite adequate oxygen saturation
If any such signs are identified, it is generally prudent to secure the patient's airway early, before significant further deterioration occurs. Of note, the signs listed above may not be present during the clinician's initial examination. Frequent reexamination is needed in patients who have sustained significant direct trauma to the airway but whose airway is not secured early. (See 'Patients appropriate for observation' below.) A subset of initially stable patients is at higher risk of progressing to an unstable and potentially difficult airway. Patients with any of the following signs or conditions often require early intubation to prevent subsequent airway compromise or collapse:
Unstable mandible or midface injuries Steady bleeding into the oropharynx or nasopharynx Worsening or fluctuating level of consciousness
Determining difficulty with management Emergency clinicians should assess the patient's airway for potential difficulty with bag-mask ventilation, endotracheal intubation, rescue device placement, and cricothyrotomy. Mnemonics that can be used for these
assessments are provided here and discussed in detail separately (table 1 and table 2 and table 3 and table 4). (See "The difficult airway in adults".) [1]. Difficulty with bag-mask ventilation should be anticipated in patients who have sustained severe maxillofacial injuries that disrupt bones and create instability or disfigurement in the middle or lower face. Such injuries make it difficult to maintain a proper mask seal. Obstruction from heavy bleeding, soft tissue swelling, or hematoma can interfere with effective bag-mask ventilation. Posterior displacement of severe fractures of the maxilla or mandible can also obstruct the patient's airway [2]. Subcutaneous air in the neck or communicating injury that establishes an external opening from any part of the airway (mouth, oropharynx, larynx, trachea) makes bag-mask ventilation impossible and also increases the likelihood that bag-mask ventilation will further distort anatomy, making subsequent airway rescue maneuvers more difficult. Bleeding and disruption of normal anatomy can make laryngoscopy and intubation extremely difficult. Mouth opening may be limited and should be carefully assessed with the anterior portion of the cervical collar removed. Bleeding, soft tissue swelling, and debris can obscure the view of the glottis during laryngoscopy, making effective suction essential. In-line stabilization of the neck is recommended in all blunt trauma patients during intubation, although it increases the difficulty of the procedure. In penetrating trauma, the role of in-line stabilization is controversial. Victims of isolated penetrating trauma who are neurologically intact rarely have unstable bony spinal columns and so are at low risk of sustaining a spinal cord injury during intubation [3]. However, a patient with penetrating injury may have concomitant blunt injury. Examples include a patient who is shot then falls down a flight of stairs or a patient who sustains a stab wound to the neck while also being beaten about the head and neck with blunt objects. Although in-line stabilization should be maintained if the clinician harbors any doubt about spinal column injury, the risk of unstable bony injury is so low in isolated penetrating injury with intact neurological examination that, if the operator is unable to see the glottis satisfactorily to intubate, it may be preferable to relax spinal immobilization somewhat to achieve a gentle intubation rather than allow hypoxemia to occur. Judgment is required to determine which of the two threats (hypoxemia caused by a failed airway versus spinal cord injury caused by spinal column movement) represents a more realistic or serious risk to the patient. Placement of a rescue device (eg, laryngeal mask airway) can be difficult if mouth opening is limited, if the airway is disrupted or distorted (eg, by swelling), or if debris such as teeth or bone fragments are present. Cricothyrotomy can be difficult if normal anatomic relationships are disrupted or a hematoma is present at the anterior neck. MANAGEMENT
Guiding principle: Secure the airway early Injuries sustained from direct trauma to the airway are often dynamic and conditions can deteriorate quickly [4]. As examples, hematomas and soft tissue swelling can expand rapidly, converting a partially obstructed airway into a completely obstructed airway. Signs suggestive of imminent obstruction are described above. (See 'Signs of airway compromise' above.) It is best to secure the airway early whenever signs of active or impending obstruction are identified or there is doubt about the extent of the injuries or their likely course. Doing so enables clinicians to secure the airway under relatively controlled circumstances before complete obstruction occurs and a crisis ensues. The basic and advanced techniques used to manage the airway are discussed separately. (See "Rapid sequence intubation in adults" and "The difficult airway in adults" and "Basic airway management in adults" and "Devices for difficult emergency airway management in adults".) Crash airway: No time available Patients with direct trauma to their airway may present in extremis, unresponsive to the examiner and without effective ventilation or circulation (ie, crash airway). The basic approach to the crash airway remains unchanged in such patients. Algorithms outlining the basic approach to the traumatized airway and the crash airway are provided (algorithm 1 and algorithm 2). The airway management algorithms are discussed in detail separately. (See "Advanced emergency airway management in adults" and "The failed airway in adults".) Management of the patient with an exposed trachea, most likely from a stab wound to the neck or a "clothesline" type injury, differs from the standard management of a crash airway. In such a circumstance, the airway manager prevents the inferior portion of the trachea from retracting into the chest by grasping it with a towel clip or clamp and the exposed trachea is then intubated directly. The general management of penetrating neck wounds is discussed separately. (See "Penetrating neck injuries".) Time available and difficult airway anticipated The difficult airway algorithm provides the fundamental approach to the patient with direct airway trauma who requires intubation and whose airway is anticipated to be difficult (algorithm 3 and algorithm 1). The specific approach selected is determined by the patient's injuries, patient attributes that suggest difficult airway management, the skills of the airway manager, and the resources available. The traumatized airway can be difficult to manage and it is important to obtain whatever help is available. The general approach to the difficult airway is discussed separately; aspects of management related to the traumatized airway are discussed below. (See "The difficult airway in adults".) The most important questions to ask when faced with a traumatized airway (or any difficult airway) are:
Is there time? In other words, can the patient's oxygen saturation (SpO2) be maintained above 90 percent? Is difficulty with bag-mask ventilation (BMV) anticipated?
If the SpO2 can be maintained above 90 percent, there is some time to consider different approaches and to make preparations. If adequate oxygenation cannot be maintained, a failed airway is present, and a definitive airway must be established promptly (algorithm 4). (See "The failed airway in adults".) If the SpO2 remains above 90 percent and no risk factors for difficult BMV are identified, the clinician may elect to use standard rapid sequence intubation (RSI) to secure the airway. If difficult BMV is anticipated, RSI may pose risks, and an awake approach to intubation may be best. Alternatively, RSI may be undertaken using a "double set-up" in which the patient undergoes RSI with one or two brief attempts at laryngoscopy, proceeding directly to a cricothyrotomy if intubation is not possible. Both the intubation and the cricothyrotomy are prepared for in advance. (See "Rapid sequence intubation in adults".) Of note, subcutaneous emphysema usually represents a contraindication to BMV because gases forced into the airway during BMV can expand the neck's soft tissues, compromising subsequent efforts to ventilate or to intubate. In patients with very minimal detectable subcutaneous air, gentle, controlled bag-mask ventilation might be attempted, but its effectiveness is not assured and it should be abandoned if the subcutaneous air increases in volume. The awake approach to securing the airway involves sedation to the level used for common emergency department procedures (eg, using propofol or ketamine) in conjunction with topical airway anesthesia (eg, using atomized or nebulized lidocaine, or lidocaine paste or jelly). This approach allows the patient to continue to breathe spontaneously while sedation and topical anesthesia enable the clinician to overcome the patient's protective airway reflexes. Excessive blood or secretions in the airway limit the effectiveness of topical anesthetics and may preclude use of the awake approach if adequately deep sedation without topical anesthesia cannot be achieved. (See "The difficult airway in adults", section on 'Awake look'.) The awake but sedated patient can undergo standard direct laryngoscopy, video laryngoscopy, or fiberoptic laryngoscopy. The presence of a large amount of upper airway blood will likely make flexible fiberoptic laryngoscopy difficult or impossible. If the vocal cords are visualized, the clinician can opt to intubate during the awake look without additional medications or to withdraw the laryngoscope and perform standard RSI. We believe in general it is best not to remove the laryngoscope and perform RSI in a patient with direct airway trauma due to the risk of the glottic view deteriorating during the interim. Direct, fiberoptic, and video laryngoscopy are the primary awake intubation techniques used in the setting of the traumatized airway. Flexible fiberoptics require patient stability, time, and operator expertise. Rigid fiberoptic devices (eg, optical stylet) may enable clinicians to obtain a more rapid view of the glottis. Copious blood or secretions in the airway can make fiberoptic laryngoscopy difficult or impossible. Devices used for difficult
airway management are discussed separately. (See "Devices for difficult emergency airway management in adults".) In some instances the clinician will judge RSI to be the best approach despite the presence of a potentially difficult airway, particularly if performed early before significant deterioration occurs (ie, when the anatomy is still close to normal). Several observational studies suggest that RSI is effective in patients with traumatized airways [5,6]. An approach incorporating a double set-up is often prudent when
Related Documents