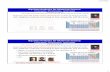
The Periodic Table

The Periodic Table. Dmitri Mendeleev (1834-1907) determined the properties of every known element at the time Atomic Mass Density Colour Melting Point.
Jan 11, 2016
Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

The Periodic Table

The Periodic Table Dmitri Mendeleev (1834-1907)
determined the properties of every known element at the time Atomic Mass Density Colour Melting Point Boiling Point

The Periodic Table: Periods
Mendeleev arranged the known elements in order of increasing atomic mass
He found that the properties of the elements repeated at definite, or PERIODIC, intervals Na has similar properties to Li and K

The Modern Periodic Table:Atomic Number In 1915, the Periodic Table was
reorganized based on the element’s atomic structure
Each element has an ATOMIC NUMBER, which is unique to each element
The atomic number begins with H (1) in the upper-left hand corner, and increases as you move from left to right

The Modern Periodic Table: Groups of Elements
3 4 5 6 7 8 9 10 11 12
13 14 15 16 17
1812

The Modern Periodic Table:Groups
Vertical columns in the periodic table Chemical families Elements in a group share very
similar properties Numbered from 1-18 Elements in the same GROUP
have the same number of VALENCE ELECTRONS (electrons in the valence or outermost shell)
1 2 13 14 15 16 17 18

Group 1: Alkali Metals
Very reactive metals that do not occur freely in nature (Cs, Fr the most reactive)
Valence Electrons: One; ready to lose that one electron to bond with other elements.
Properties: Malleable, ductile, good conductors of heat and electricity, softer than most metals
Can explode if exposed to water.
alkali metals in water

Group 2: Alkaline Earth Metals Very reactive Valence Electrons: 2; can
bond easily with other elements by losing these electrons
Not found free in nature

Groups 3-12: Transition Metals Properties: Ductile, malleable, conduct
electricity and heat Valence Electrons: transfer electrons to
nonmetals to form compounds; the number of electrons transferred can vary
Iron, cobalt, and nickel are the only elements known to produce a magnetic field.

Group 17: Halogens
“Salt-former” because they are very reactive non-metals; form salts when they react with metals
Valence Electrons: 7; will bond easily with Alkali Metals.
Exist as solids (I, At), liquid (Br), and gas (F, Cl)
reactivity of halogens

Group 18: Noble Gases Valence Electrons: full
valence shell; prevents gases from forming compounds
Very stable because they have the maximum electrons in their outer shell

Rare Earth Elements Lanthanide and Actinide series (Group 3
and Period 6-7) One element of the lanthanide series and
most of the elements in the actinide series are trans-uranium (synthetic or man-made)

Mendeleev’s Periodic Law
If the elements are arranged
according to their atomic mass, a
pattern can be seen in which
similar properties occur regularly
Related Documents