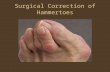
Smart Toe ® II Book your case today. Over 100,000 already sold. Stryker’s premier hammertoe treatment, Smart Toe II, pioneered the use of a one-piece, dynamic compression implant for hammertoe correction. Now with over 100,000 implants sold, Smart Toe II continues to provide demonstrated clinical outcomes. 2,3,4 PIP DIP

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

Smart Toe® II
Book your case today.Over 100,000 already sold.
Stryker’s premier hammertoe treatment, Smart Toe II, pioneered the use of a one-piece, dynamic compression implant for hammertoe correction. Now with over 100,000 implants sold, Smart Toe II continues to provide demonstrated clinical outcomes. 2,3,4
PIP
DIP

100,000+ implants sold globally
2x rate of successful fusion when compared to Kirschner wires1
Successful clinical fusion rates of over 90%2
Less than 1% reported rates of implant failure3
Clinically reported high patient post-operative satisfaction rates4
Joint line compression
Rotational stability
No risk of pin-tract infection
No violation of the distal interphalangeal joint
The Smart Toe II implant is stored below 32˚fahrenheit for two hours prior to surgery. Upon insertion, as the implant returns to normal temperature, the head and feet expand, providing compression across the joint line.
Smart Toe IIShape memory PIP & DIP implantsProven. Flexible. Supported.2,3,4
Flexible patient sizing options
Dynamic compression
Smart Toe II vs. Kirschner Wire1
A surgeon must always rely on his or her own professional clinical judgment when deciding whether to use a particular product when treating a particular patient. Stryker does not dispense medical advice and recommends that surgeons be trained in the use of any particular product before using it in surgery. The information presented is intended to demonstrate the breadth of Stryker product offerings. A surgeon must always refer to the package insert, product label and/or instructions for use before using any Stryker product. Products may not be available in all markets because product availability is subject to the regulatory and/or medical practices in individual markets. Please contact your Stryker representative if you have questions about the availability of Stryker products in your area. Stryker Corporation or its divisions or other corporate affiliated entities own, use or have applied for the following trademarks or service marks: Howmedica, Osteonics, Smart Toe, Stryker. All other trademarks are trademarks of their respective owners or holders.
ST2-SS-1_Rev-1, 03-2016 • Copyright © 2016 Stryker
1. Angirasa AK, Barrett MJ, Silvester D. Smarttoe implant compared with Kirschner wire fixation for hammer digit corrective surgery: a review of 28 patients. J Foot Ankle Surg. 2012;51(6):711-713.
2. Sandhu, J.S.; Hofbauer, M.H.; DeCarbo, W.T. Digital Arthrodesis with a One-Piece Memory Nitinol Intramedullary Fixation Device: A Retrospective Review. Foot and Ankle Specialist, October 2013, 6(5):364-366
3. According to a Medical Device reporting analysis performed in the Super and Trackwise systems for US reported events on August 3rd, 2015, the reported rate of implant failure for Smart Toe implant breakage between July 1, 2011 and August 3rd, 2015 was less than 1% of total Smart Toe implants sold.
4. Catena F, Doty JF, Jastifer J, Coughlin MJ, Stevens F. Prospective study of hammertoe correction with an intramedullary implant. Foot & Ankle International 2014, Vol. 35(4):319-325.
Visit footankle.stryker.com or email us at [email protected].
Manufactured by:Stryker GmbHBohnackerweg 1CH-2545 SelzachSwitzerland
www.stryker.com
Pre-operative Post-operative
Neutral 0˚
Angled 0˚
PIP range 15 16 19 20 21 22 DIP range
Arthodesis line
Neutral 0˚ ST0-15P ST0-16P ST0-19P ST0-20P ST0-21P ST0-22P xs xs
Angled 0˚ ST0A-15P ST0A-16P ST0A-19P ST0A-20P ST0A-21P ST0A-22P ST0XS-11 ST0XS-13
Related Documents