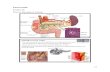
27.05.2015 1 endocrine diseases PITUITARY The normal pituitary. (A) The gland is bean shaped and covered by a fibrous capsule. It hangs from the hypothalamus by the pituitary stalk or infundibulum. (B) On horizontal cross-section the gland is readily seen to have two distinct components, the anterior lobe representing the larger part (top) and the posterior lobe, or neurohypophysis, representing the smaller component (bottom). Between them is the intermediate lobe, which consists only of vestigial structures that are recognized grossly as tiny cysts with gelatinous contents

Pathology Lecture 4
Sep 16, 2015
Summary of Pathology - compromised - Endocrinology
Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript
-
27.05.2015
1
endocrine diseases
PITUITARY
The normal pituitary. (A) The gland is bean shaped and covered by a fibrous capsule. It hangs from the hypothalamus by the pituitary stalk or infundibulum. (B) On horizontal cross-section the gland is readily seen to have two distinct components, the anterior lobe representing the larger part (top) and the posterior lobe, or neurohypophysis, representing the smaller component (bottom). Between them is the intermediate lobe, which consists only of vestigial structures that are recognized grossly as tiny cysts with gelatinous contents
-
27.05.2015
2
-
27.05.2015
3
-
27.05.2015
4
ADRENAL GLAND
-
27.05.2015
5
The cortex, of course, has three "zones": (1) ZONA GLOMERULOSA: mineralocorticoid
production. Thin and patchy, small cells.
(2) ZONA FASCICULATA: glucocorticoid production (now seems settled), resting cells (the reserve cells are at the ZG-ZR interface). Yellow.
(3) ZONA RETICULARIS: glucocorticoid production, androgen and estrogen production, grossly darker than outer layers. Brown.
(Salt, sugar, and sex: the deeper you go, the sweeter it gets.)
CUSHINGS SYNDROME (too much glucocorticoids)
Symptoms and signs: truncal obesity with "buffalo hump" ("upper trunk fat") and "moon
face" increased appetite insomnia and mental changes (Cushing's psychosis) vascular and connective tissue changes: thinning of the dermis,
bruisability, striae ("purple stripes"), very poor healing, purpura high blood pressure glucose intolerance (diabetes, with complications) osteoporosis loss of normal circadian rhythm of cortisol secretion other: acne, cellulitis, edema, gastric ulcers, hirsutism,
oligomenorrhea, hypokalemia, muscle wasting,
CUSHINGS SYNDROME
Etiology (both Cushing's disease and glucocorticoid-secreting
adenomas are most common in women ages 15 to 45, but can be diagnosed in anybody and anytime)
Adrenals are: atrophic if glucocorticoids were administered
hyperplastic if ACTH was administered
CUSHINGS SYNDROME. etiology 1. IATROGENIC (the most common).
2. ACTH-PRODUCING PITUITARY LESION, usually a
basophilic microadenoma ("Cushing's disease", "pituitary Cushingism")
the adrenals usually are diffusely enlarged (but may be nodular),
"Nelson's syndrome" - rapid enlargement of the pituitary adenoma leading to hyperpigmentation, blindness and death - followed adrenalectomy in many of these patients
CUSHINGS SYNDROME. etiology
3. ADRENAL CORTICAL ADENOMA OR CARCINOMA ("adrenal Cushingism") the tumor may be primary, or an autonomous adrenal tumor may develop after years of "pituitary Cushingism")
4. ACTH- (OR CRH-) PRODUCING CANCERS OF OTHER ORGANS: small-cell carcinoma, bronchial and thymic carcinoids, medullary thyroid carcinoma, islet cell cancer; other APUDomas.
Full-blown Cushingism is rare in oat cell patients, only because they don't live long enough
CUSHINGS SYNDROME. etiology
5. Really "primary" adrenal hyperplasia (not due to excess ACTH): "primary pigmented nodular adrenocortical
hyperplasia" (part of the autosomal dominant Carney complex)
circulating antibodies against the ACTH receptor (analogous to Graves disease) can occasionally stimulate the gland into hyperactivity
6. Cushingism with a burst of cortisol after eating: inappropriate expression of GIP receptors on the adrenal cortex/adrenal adenoma
7. Recurrent cushingism of pregnancy
-
27.05.2015
6
PRIMARY HYPERALDOSTERONISM (Conn's syndrome)
"low-renin hyperaldosteronism": too much mineralocorticoid
Etiology: "idiopathic" adrenal hyperplasia, or an adrenal adenoma. the most familiar cause is an "autonomous" adrenal
cortical adenoma (CONN'S SYNDROME), often very small
a cause of surgically-correctable high blood pressure (0.5% of hypertensives have primary hyperaldosteronism)
PRIMARY HYPERALDOSTERONISM
clinic: classically, patients exhibit:
hypokalemia (muscle weakness, and even paralysis),
alkalosis, and low rennin, surprisingly, these patients do not have
edema
SECONDARY HYPERALDOSTERONISM
is much more common then primary part of the picture in:
chronic heart failure, cirrhosis, nephrotic syndrome, and other problems.
ADRENAL CORTICAL ADENOMAS
round, yellow (like the adrenal cortex) nodules ("nodular hyperplasia")
adrenal cortical adenomas are found in 2% of autopsies and abdominal CT scans ("incidentalomas", as far as there is no evidence of steroid or catecholamine over-production)
may cause: Cushing's syndrome, Conn's syndrome, or virilization, the vast majority are clinically silent
ADRENAL CORTICAL CARCINOMA
rare tumor, but often lethal many are hormonally active (Cushing's, Conn's,
and/or virilization), mixed endocrine syndromes usually mean cancer (Adrenal tumors that feminize, or that produce androgens without glucocorticoids, are most often malignant)
histology: usually obviously malignant, grossly and microscopically, with ten or more mitotic figures per high power field
HYPOADRENOCORTICISM ("Addisonism")
Insufficient glucocorticoid (and usually insufficient mineralocorticoid) production.
chronic hypoadrenocorticism (ADDISON'S DISEASE, now regardless of etiology)
troubles start when 80% of the gland tissue is gone, There are several etiologies of chronic
hypoadrenocorticism:
-
27.05.2015
7
HYPOADRENOCORTICISM INFECTIONS most of Dr. Addison's patients had bovine TB of the
adrenals, worldwide:
fungal infections (histoplasmosis, coccidioidomycosis and South American blastomycosis)
other important causes: leprosy, now AIDS is too.
HYPOADRENOCORTICISM AUTOIMMUNE the most prevalent non-iatrogenic cause of Addison's
disease in the US the adrenals are typically loaded with lymphocytes. most of patients have autoantibodies against 21-
hydroxylase ("adrenal cortex antibodies) autoimmune adrenalitis often occurs jointly with Hashimoto's
thyroiditis, type I diabetes mellitus, vitiligo, gluten enteropathy, and/or pernicious anemia ("autoimmune polyendocrine deficiency syndrome II", "Schmidt's syndrome", etc.)
HYPOADRENOCORTICISM IATROGENIC results from too-rapid withdrawal of glucocorticoid
medication, post-adrenalectomy for breast cancer or Cushingism,
etc., ketoconazole or fluconazole (antifungal drug therapy), removal of a "non-functioning adenoma" (rare). CORTICOSTEROID INSUFFICIENCY OF CRITICAL
ILLNESS a newly-characterized entity, seen especially in severe
systemic infections, in which the body does not put out enough glucorticoid to handle the extra stress
HYPOADRENOCORTICISM OTHERS: amyloidosis congenital hypoplasia hemochromatosis (a common disease still under-
diagnosed in the U.S.) sarcoidosis DAX1 mutations metastatic cancer to adrenal glands anticoagulant therapy CMV infection (in AIDS, it's almost the rule) adrenal leukodystrophy (one male in 17,000)
SECONDARY HYPOADRENOCORTICISM (ACTH deficiency)
patients almost always with lost of adenohypophysis and "panhypopituitarism".
less often, selective, presumably autoimmune, loss of the ACTH-producing cells.
SECONDARY HYPOADRENOCORTICISM (ACTH deficiency)
Clinical picture ("Addisonian" patients): weakness, nausea, and weight loss, and are usually
hypotensive and other complaints (like in most endocrine patients, the problems are likely to appear "emotional")
In primary hypoadrenocorticism, the skin and buccal mucosa will usually be hyperpigmented, due to increased ACTH (MSH?)
Lab studies typically show hyponatremia, hyperkalemia, metabolic acidosis, hypoglycemia, low serum cortisol, low urinary 17-OH-steroids, and (most important) failure to respond to various "stimulation tests" by increasing cortisol output.
commonly patients die suddenly and unexpectedly before anyone thinks of adrenocortical insufficiency
-
27.05.2015
8
ACUTE HYPOADRENOCORTICISM
"adrenal apoplexy", "Addisonian crisis
Sudden collapse, often fatal (opening of the peripheral vasculature and shock with high
cardiac output)
It may result from undiagnosed adrenal insufficiency iatrogenic, infection, surgery, or treatment
WATERHOUSE-FRIDERICHSEN SYNDROME
"adrenal apoplexy" features hemorrhage, fibrin thrombi, and sometimes
necrosis in the adrenals in a setting of sepsis is not rare (often overlooked (patients in shock
WITHOUT elevated serum cortisol) occurs when there is overwhelming sepsis with
hemorrhage into, and destruction of, the adrenals, patients develop: purpura, shock, and die in a few hours. etiology:
classically the meningococcus, staphylococci pneumococci, and H. influenza
ADRENAL MEDULLA
around 10% of the normal adrenal by weight
produce "adrenalin" (epinephrine, also norepinephrine)
"Adrenal medullary hyperplasia" is a marker for MEN II and a few rarities
any nodule bigger than 1 cm is a pheochromocytoma. - pheochromocytoma (well-differentiated, adults) - neuroblastoma (poorly-differentiated, children)
PHEOCHROMOCYTOMA
"paraganglioma", formerly "10% tumor":
in young patients (10-20 yrs) cases involve both adrenals, are familial, and 10% metastasize
- is named for its colorful reaction in fixatives containing chromic acid salts
- secrete norepinephrine (most common) and/or epinephrine (usually less, and often others: dopamine, serotonin, ACTH, somatostatin, neuropeptide Y, and/or VIP)
- IHC: chromogranin+ and/or synaptophysin+
PHEOCHROMOCYTOMA
Clinic: The infamous paroxysms of extreme hypertension,
accompanied by sweating, headache, and other autonomic disturbances, probably result from physical compression and/or ischemia
Even a tiny (1 gm) benign pheochromocytoma can make a person very sick and will eventually cause death
Regardless of location and appearance, the patients will report anxiety, headache, palpitations, "panic attacks", sweating, dizziness, etc. (the basic problem is emotional. "Pheo is a great imitator.")
PHEOCHROMOCYTOMA
"pheochromocytoma" is defined to arise in the adrenal medulla, however similar tumors (less common) arise elsewhere are called "extra-adrenal paragangliomas."
Macro: - are very bloody (because they are very vascular), and
often show fibrosis, calcification, cystic change, or even fatty change
Microscopically: resemble adrenal medulla, there are no histologic criteria
for malignancy, not even vascular invasion. 5-year survival rate with malignant pheo is around 50%
Related Documents