Allele Biotech-Introducing Cost Effectiveness to Research G reen uorescent proteins (GFP) and variants thereof are widely used to study protein localization and dynamics. However, among the most commonly used tags for immuno- precipitation (a brief review in Box 1), the use of GFP is limited due to the previously available anti-GFP antibod- ies, either polyclonal or monoclonal, not being comparable to those against other tags. GFP-multiTrap® is a high qual- ity GFP-bingding protein based on a single domain antibody immobilized in wells. It is characterized by a small barrel shaped structure (13 KDa, 2.5 nm X 4.5 nm) and a very high stability (stable up to 70°C, functional within 2 M NaCl or 0.5% SDS). From detailed in vitro binding analysis, we deter- mined that one molecule GFP-Trap® binds one molecule GFP in a stable stoichiometric complex. The dissocia- tion constant (Kd) lies with 0.59 nm within the picomolar range compara- ble to conventional antibodies. GFP-multiTrap® is available in black 96-well plate format with clear bottom for colorimetric, chemiluminescence and uorescence detection methods. With much greater stability, specicity, and afnity, GFP-Trap®, the recent addition to antibodies for immunopre- cipitation should make GFP in line to become the most suitable tags for im- munoprecipitation assays. Datas from direct comparison of the GFP-Trap® with conventional antibodies will be shown in Box 2. Besides wtGFP, GFP-multiTrap® can also bind to eGFP and GFPS65T as well as to YFP and eYFP. It recog- nizes and binds a three dimensional epitope at the beta barrel structure. Interestingly it does not bind to CFP, which is due to the fact, that CFP ex- hibit an amino acid exchange within the recognized epitope. In addition we could not detect any binding to red uorescent proteins derived from DsRed (RFP-Trap® is available as another product line). Meanwhile, as GFP-Trap® recognizes the beta barrel structuure of GFP, it does not recognise unfolded or denatured GFP (e.g. on immunoblots). Chromotek-GFP-multiTrap ® , GFP-Trap® immobilized in wells to test your GFP fusion proteins for pep- tide, protein, DNA or RNA binding. Box 1 | Tags for Immunoprecipitation T o achieve effective immunoprecipitation, a researcher must rst overcome the difculty of nding usable antibodies against a target of interest. Using tags that are fused to the C- or N-terminus of the targe t protein is common practice. In general, while keeping mindful of the unique nuances with each biological system, choosing tags that have been tested in many situations and been proven to be non-interfering is ideal. The most commonly used tags are: FLAG, Myc, HA, V5, T7, and His, which are quite small in size and in theory less likely to interfere. GST and GFP are well documented to form self-contained and stable structures independent of their fusion partners and proven to not interfere in many cases despite their larger size (in be- tween 20-30kD). A top choice for pulldown experiments, GST c an bind to glutathione beads directly. GFP and variants are excellent tags having both the advantages of being a visualization module to follow the protein both inside cells and during pull- down. However its use is limited due to the previously available anti-GFP antibodies, either polyclonal or monoclonal, not being comparable to those against other tags. With much greater stability, specicity, and afnity, GFP-Trap®, the recent addition to antibodies for immunoprecipitation should make GFP in line to become the most suitable tags for immunoprecipitation assays. Box 2 | Comparative Immunoprecipitation Assay [A] GFP-multiTrap® Immunoprecipitations (IP) of GFP and GFP-fusion proteins (CBX1) from ex- tracts of GFP-producing human cells. Input (I), non-bound (FT), and bound (B) fractions were sepa- rated by SDS-PAGE. [B] Fraction input percentages of GFP and GFP-CBX1 and their GFP brightness displaying the efciency of pulldown using the GFP-multiTrap®. A B

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

8/4/2019 ChromoTek GFP-multiTrap
http://slidepdf.com/reader/full/chromotek-gfp-multitrap 1/3
Allele Biotech-Introducing Cost Effectiveness to Research
Green uorescent proteins (GFP)and variants thereof are widely
used to study protein localization and
dynamics. However, among the most
commonly used tags for immuno-
precipitation (a brief review in Box
1), the use of GFP is limited due to the
previously available anti-GFP antibod-
ies, either polyclonal or monoclonal,
not being comparable to those against
other tags.
GFP-multiTrap® is a high qual-
ity GFP-bingding protein based on a
single domain antibody immobilizedin wells. It is characterized by a small
barrel shaped structure (13 KDa, 2.5
nm X 4.5 nm) and a very high stability
(stable up to 70°C, functional within 2
M NaCl or 0.5% SDS). From detailed
in vitro binding analysis, we deter-
mined that one molecule GFP-Trap®
binds one molecule GFP in a stable
stoichiometric complex. The dissocia-
tion constant (Kd) lies with 0.59 nm
within the picomolar range compara-
ble to conventional antibodies.
GFP-multiTrap® is available in black
96-well plate format with clear bottom
for colorimetric, chemiluminescence
and uorescence detection methods.
With much greater stability, specicity,
and afnity, GFP-Trap®, the recent
addition to antibodies for immunopre-
cipitation should make GFP in line to
become the most suitable tags for im-munoprecipitation assays. Datas from
direct comparison of the GFP-Trap®
with conventional antibodies will be
shown in Box 2.
Besides wtGFP, GFP-multiTrap® can
also bind to eGFP and GFPS65T as
well as to YFP and eYFP. It recog-
nizes and binds a three dimensionalepitope at the beta barrel structure.
Interestingly it does not bind to CFP,
which is due to the fact, that CFP ex-
hibit an amino acid exchange within
the recognized epitope. In addition
we could not detect any binding to
red uorescent proteins derived from
DsRed (RFP-Trap® is available as
another product line). Meanwhile,
as GFP-Trap® recognizes the beta
barrel structuure of GFP, it does not
recognise unfolded or denatured GFP
(e.g. on immunoblots).
Chromotek-GFP-multiTrap®, GFP-Trap®
immobilized in wells to test your GFP fusion proteins for pep-tide, protein, DNA or RNA binding.
Box 1 | Tags for Immunoprecipitation
To achieve effective immunoprecipitation, a researcher must rst overcome the
difculty of nding usable antibodies against a target of interest. Using tags that are
fused to the C- or N-terminus of the target protein is common practice. In general,
while keeping mindful of the unique nuances with each biological system, choosing
tags that have been tested in many situations and been proven to be non-interfering
is ideal. The most commonly used tags are: FLAG, Myc, HA, V5, T7, and His, which
are quite small in size and in theory less likely to interfere. GST and GFP are well
documented to form self-contained and stable structures independent of their fusion
partners and proven to not interfere in many cases despite their larger size (in be-
tween 20-30kD). A top choice for pulldown experiments, GST can bind to glutathione
beads directly. GFP and variants are excellent tags having both the advantages of
being a visualization module to follow the protein both inside cells and during pull-down. However its use is limited due to the previously available anti-GFP antibodies,
either polyclonal or monoclonal, not being comparable to those against other tags.
With much greater stability, specicity, and afnity, GFP-Trap®, the recent addition
to antibodies for immunoprecipitation should make GFP in line to become the most
suitable tags for immunoprecipitation assays.
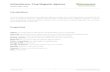
Box 2 | Comparative Immunoprecipitation Assay
[A] GFP-multiTrap® Immunoprecipitations (IP) of GFP and GFP-fusion proteins (CBX1) from ex-tracts of GFP-producing human cells. Input (I), non-bound (FT), and bound (B) fractions were sepa-rated by SDS-PAGE.
[B] Fraction input percentages of GFP and GFP-CBX1 and their GFP brightness displaying theefciency of pulldown using the GFP-multiTrap®.
A
B

8/4/2019 ChromoTek GFP-multiTrap
http://slidepdf.com/reader/full/chromotek-gfp-multitrap 2/3
Allele Biotech-Introducing Cost Effectiveness to Research
Protocols
1. Immunoprecipitation
•For one immunoprecipitation reaction resuspend cell pel-
let (~107 cells) in 200 μL lysis buffer by pipetting (or using
a syringe)
•Place the tube on ice for 30 min with extensively pipetting
every 10 min•Spin cell lysate at 20.000x g for 5-10 min at 4°C
•Transfer supernatant to a precooled tube. Adjust volume
with dilution buffer to 500 μL-1000 μL. Discard pellet.
The cell lysate can be frozen at this point for long-term stor-
age at minus 80°C.
For immunoblot analysis dilute 50 μL cell lysate with 50 μL
4x SDS-sample buffer (→refer as input)
•Add 50 μL (half area GBP-Plate) or 100 μL (full area GBP
plate) cell lysate per well
•Incubate for 2 hours at 4°C under shaking (800rpm)
For western blot analysis dilute 50 μL cell lysate with 50 μL4x SDS-sample buffer (→refer as ow-through)
•Discard remaining supernatant
•Wash wells two times with 100 μL- 200 μL wash buffer
•Add 100 μL dilution buffer for Fluorescence intensity mea-
surements
Add 10 μL of elution buffer to wells and transfer it to a tube.
Buffer with 1M Tris pH 7.5 to an end concentration of 100
mM Tris and dilute with 10 μL 4x SDS-sample buffer (→refer
as bound)
2. In vitro binding assays after IP
2.1. In vitro histone-tail peptide binding assay
•After one-step purication of GFP fusion proteins with
the 96-well GBP plate in half-area plates (1), the wells are
blocked with 100 μL blocking buffer for 30 minutes at 4°C
under shaking (800rpm)
•Discard blocking solution
•Equilibrated wells with 50 μL dilution buffer supplemented
with 0.05% Tween
•Add peptides to a nal concentration of 0.15 μM
•Incubate at room temperature for 20 min under shaking
(800rpm)
•Discard supernatant
•Wash wells two times with 100 μL wash buffer
•Add 100 μL dilution buffer for Fluorescence intensity mea-
surements
2.2. In vitro DNA binding assay
•After one-step purication of GFP fusion proteins with the
96-well GBP plate in full-area plates(1), the wells are equilibrated with 100 μL dilution buffer sup-
plemented with 2mM DTT and
100ng/μL BSA
•Add uorescent-labeled DNA probe to a nal concentration
of 0.15 μM
•Incubate at room temperature for 30 min under shaking
(800rpm)
•Discard supernatant
•Wash wells two times with 100 μL dilution buffer
•Add 100 μL dilution buffer for Fluorescence intensity mea-
surements
2.3. In Vitro Protein-Protein binding assay
•After one-step purication of GFP fusion proteins with the
96-well GBP plate in full-area plates
(1), the wells are equilibrated with 100 μL dilution buffer sup-
plemented with 2mM DTT and
100ng/μL BSA
•Prepare cell lysate of RFP-fusion protein as described for
GFP-fusion proteins
•Add 50 μL (half area GBP-Plate) or 100 μL (full area GBP
plate) cell lysate per well•Incubate for 30 minutes at 4°C under shaking (800rpm) For
western blot analysis dilute 50 μL cell lysate with 50 μL 4x
SDS-sample buffer (→refer as ow-through)
•Discard remainning supernatant
•Wash wells two times with 100 μL- 200 μL wash buffer
•Add 100 μL dilution buffer for Fluorescence intensity mea-
surements. Add 10 μL of elution buffer to wells and transfer it
to a tube. Buffer with 1M Tris pH 7.5 to an end concentration
of 100 mM Tris and dilute with 10 μL 4x SDS-sample buffer
(→refer as bound)
Box 1 | Product List
GFP-multiTrap® Plates
Chromotek-GFP-multiTrap® ABP-CM-GMULT1 1 Plate
ABP-CM-GMULT5 5 Plates

8/4/2019 ChromoTek GFP-multiTrap
http://slidepdf.com/reader/full/chromotek-gfp-multitrap 3/3
Allele Biotech-Introducing Cost Effectiveness to Research
ProtocolsContinued
3. Fluorescence Measurements
3.1. Quantication with Tecan Innite M1000 plate reader
•GFP: : 490±10 nm and 511±10 nm
•RFP: 586±5 nm and 608±10 nm
•TAMRA: 560±5 nm and 586±5 nm
•DNA: according to label
For quantication, uorescence intensity measurements are
adjusted using standard curves from labelled probes with
known concentrations.
Suggested Buffers
Lysis buffer
20 mM Tris/HCl pH7.5
150 mM NaCl
0.5 mM EDTA
0.5% NP-40
Optional:
1 mM PMSF has to be freshly added
1x Protease Inhibitor Cocktail has to be freshly added
For nuclear/chromatin proteins:
DNase nal conc. 1 μg/μl
2.5 mM MgCl2
Dilution buffer
20 mM Tris/HCl pH7.5
150 mM NaCl
0.5 mM EDTA
Wash buffer
20 mM Tris/HCl pH7.5
50-500 mM NaCl
0.5 mM EDTA
Blocking Buffer
3% milk solved in TBS-T (0.075% Tween)
Elution buffer (freshly prepared)
300 mM Glycin pH2.5
Box 3 | GFP-multiTrap Overview
Welcome to Join in the Discussion at Allele’s Network:
Allele News: news.allelebiotech.com
Allele Blog: blogs.allelebiotech.com
Allele Facebook: http://www.facebook.com/…
Allele Twitter: http://www.twitter.com/allele_biotech
Allele Myspace: http://www.myspace.com/allelebiotech
Related Documents