COBIOT-854; NO. OF PAGES 8 Please cite this article in press as: Lovley DR, Nevin KP. A shift in the current: New applications and concepts for microbe-electrode electron exchange, Curr Opin Biotechnol (2011), doi:10.1016/ j.copbio.2011.01.009 Available online at www.sciencedirect.com A shift in the current: New applications and concepts for microbe-electrode electron exchange Derek R Lovley and Kelly P Nevin Perceived applications of microbe-electrode interactions are shifting from production of electric power to other technologies, some of which even consume current. Electrodes can serve as stable, long-term electron acceptors for contaminant-degrading microbes to promote rapid degradation of organic pollutants in anaerobic subsurface environments. Solar and other forms of renewable electrical energy can be used to provide electrons extracted from water to microorganisms on electrodes at suitably low potentials for a number of groundwater bioremediation applications as well as for the production of fuels and other organic compounds from carbon dioxide. The understanding of how microorganisms exchange electrons with electrodes has improved substantially and is expected to be helpful in optimizing practical applications of microbe-electrode interactions, as well as yielding insights into related natural environmental phenomena. Address Department of Microbiology, University of Massachusetts, Amherst, MA 01003, United States Corresponding author: Lovley, Derek R ([email protected]) Current Opinion in Biotechnology 2011, 22:1–8 This review comes from a themed issue on Environmental biotechnology Edited by Lindsay Eltis and Ariel Kushmaro 0958-1669/$ – see front matter # 2011 Elsevier Ltd. All rights reserved. DOI 10.1016/j.copbio.2011.01.009 Introduction Electrodes can supply electrons to support the respiration of some microorganisms [1,2 ] or can accept electrons, serving as an electron acceptor to support anaerobic oxidation of organic compounds or inorganic electron donors such as hydrogen and elemental sulfur [3,4 ]. Electron flow between microorganisms and electrodes in both directions is of significance, not only because these are interesting forms of microbial respiration, which may provide insights into how microorganisms may function in natural environments, but also because the ability of microorganisms to consume or produce electrical current has potential practical applications in the environmental and bioenergy fields. Although there has been intense focus on producing elec- trical power with microbial fuel cells over the last decade, some of the early optimism for power production has waned and there is now a major shift in focus to other applications. After hundreds of studies, it is apparent that just about any form of organic matter that microbes can degrade can be converted to current [4 ] and powering electronic equipment with electricity harvested from the complex organic matter in aquatic sediments with benthic microbial fuel cells continues to be a promising application [5–8]. However, after some of the rather obvious design flaws in early microbial fuel cells were rectified, there has been little increase in the power output of microbial fuel cells in recent years [9]. Furthermore, effectively scaling microbial fuel cells to sizes that can handle large volumes of organic waste may be problematic [10 ]. Economic assess- ments indicate that even if the current density and scaling issues can be resolved, current harvesting will probably need to be supplemented with some value-added reaction for the treatment of wastewaters with microbial fuel cell technology to be competitive with other, more mature technologies [11 ,12]. One strategy may be to add elec- trical energy to the wastewater treatment system to over- come electrochemical limitations and focus on product formation [13]. In addition to the well-known possibility of producing hydrogen at the cathode [14 ], it has been suggested that is also feasible to generate peroxide [15] or caustic [16] through abiotic processes at the cathode. Water desalination may also be feasible with energy derived from wastewater in a novel microbial fuel cell design [17]. However, until solutions for increasing power output and scaling are conceived, wastewater-related processes may be one of the less attractive applications of microbe- electrode interactions. Therefore, this review focuses on other technologies in which microbe-electrode interactions might be employed. Many of the most promising applications for microbe- electrode interactions are based on directly supplying electrons to microorganisms at a cathode to permit them to catalyze useful processes. It is possible to indirectly transfer electrons from electrodes to microorganisms via the production of hydrogen gas or the reduction of electron shuttle molecules, but as previously reviewed [1,18], these indirect approaches have serious limitations in practical application and will not be discussed in detail here. A major conceptual shift in such studies is to move away from linking cathode processes to the oxidation of organic matter in wastewater at the anode as the source of www.sciencedirect.com Current Opinion in Biotechnology 2011, 22:1–8

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

COBIOT-854; NO. OF PAGES 8
Available online at www.sciencedirect.com
A shift in the current: New applications and concepts formicrobe-electrode electron exchangeDerek R Lovley and Kelly P Nevin
Perceived applications of microbe-electrode interactions are
shifting from production of electric power to other
technologies, some of which even consume current.
Electrodes can serve as stable, long-term electron acceptors
for contaminant-degrading microbes to promote rapid
degradation of organic pollutants in anaerobic subsurface
environments. Solar and other forms of renewable electrical
energy can be used to provide electrons extracted from water
to microorganisms on electrodes at suitably low potentials for
a number of groundwater bioremediation applications as well
as for the production of fuels and other organic compounds
from carbon dioxide. The understanding of how
microorganisms exchange electrons with electrodes has
improved substantially and is expected to be helpful in
optimizing practical applications of microbe-electrode
interactions, as well as yielding insights into related natural
environmental phenomena.
Address
Department of Microbiology, University of Massachusetts, Amherst, MA
01003, United States
Corresponding author: Lovley, Derek R ([email protected])
Current Opinion in Biotechnology 2011, 22:1–8
This review comes from a themed issue on
Environmental biotechnology
Edited by Lindsay Eltis and Ariel Kushmaro
0958-1669/$ – see front matter
# 2011 Elsevier Ltd. All rights reserved.
DOI 10.1016/j.copbio.2011.01.009
IntroductionElectrodes can supply electrons to support the respiration
of some microorganisms [1,2��] or can accept electrons,
serving as an electron acceptor to support anaerobic
oxidation of organic compounds or inorganic electron
donors such as hydrogen and elemental sulfur [3,4��].Electron flow between microorganisms and electrodes in
both directions is of significance, not only because these
are interesting forms of microbial respiration, which may
provide insights into how microorganisms may function in
natural environments, but also because the ability of
microorganisms to consume or produce electrical current
has potential practical applications in the environmental
and bioenergy fields.
Please cite this article in press as: Lovley DR, Nevin KP. A shift in the current: New applications an
j.copbio.2011.01.009
www.sciencedirect.com
Although there has been intense focus on producing elec-
trical power with microbial fuel cells over the last decade,
some of the early optimism for power production has
waned and there is now a major shift in focus to other
applications. After hundreds of studies, it is apparent that
just about any form of organic matter that microbes can
degrade can be converted to current [4��] and powering
electronic equipment with electricity harvested from the
complex organic matter in aquatic sediments with benthic
microbial fuel cells continues to be a promising application
[5–8]. However, after some of the rather obvious design
flaws in early microbial fuel cells were rectified, there has
been little increase in the power output of microbial fuel
cells in recent years [9]. Furthermore, effectively scaling
microbial fuel cells to sizes that can handle large volumes of
organic waste may be problematic [10��]. Economic assess-
ments indicate that even if the current density and scaling
issues can be resolved, current harvesting will probably
need to be supplemented with some value-added reaction
for the treatment of wastewaters with microbial fuel cell
technology to be competitive with other, more mature
technologies [11��,12]. One strategy may be to add elec-
trical energy to the wastewater treatment system to over-
come electrochemical limitations and focus on product
formation [13]. In addition to the well-known possibility
of producing hydrogen at the cathode [14��], it has been
suggested that is also feasible to generate peroxide [15] or
caustic [16] through abiotic processes at the cathode. Water
desalination may also be feasible with energy derived from
wastewater in a novel microbial fuel cell design [17].
However, until solutions for increasing power output
and scaling are conceived, wastewater-related processes
may be one of the less attractive applications of microbe-
electrode interactions. Therefore, this review focuses on
other technologies in which microbe-electrode interactions
might be employed.
Many of the most promising applications for microbe-
electrode interactions are based on directly supplying
electrons to microorganisms at a cathode to permit them
to catalyze useful processes. It is possible to indirectly
transfer electrons from electrodes to microorganisms via
the production of hydrogen gas or the reduction of electron
shuttle molecules, but as previously reviewed [1,18], these
indirect approaches have serious limitations in practical
application and will not be discussed in detail here.
A major conceptual shift in such studies is to move away
from linking cathode processes to the oxidation of organic
matter in wastewater at the anode as the source of
d concepts for microbe-electrode electron exchange, Curr Opin Biotechnol (2011), doi:10.1016/
Current Opinion in Biotechnology 2011, 22:1–8

2 Environmental biotechnology
COBIOT-854; NO. OF PAGES 8
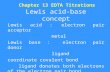
Figure 1
Sediment-Water Interface
OrganicContaminants
Adsorption andConcentration ofContaminants onAnode
Cells Co-Localized withContaminants andElectron Acceptor
OrganicContaminants
CO2
O2 2H2O
Current Opinion in Biotechnology
Strategy for stimulating the anaerobic oxidation of organic contaminants
in sediments.
electrons. Although such anodes can supply electrons at a
low potential and thus lower the energetic costs of sup-
plying electrons to the cathode [14��], wastewater anodes
tie processes to the wastewater treatment concept. In
addition to practical concerns about scaling, wastewater
treatment plants will often not be located near sites where
they are required for applications such as groundwater
bioremediation. Water is abundant and ubiquitous, mak-
ing it an ideal electron donor for many perceived cathode
applications [1].
There have also been recent conceptual shifts in models
for microbe-electrode interactions that may impact on
strategies for the optimization of proposed microbe-elec-
trode technologies. The purpose of this review is to
update progress since a similar recent review [19], with
a focus on new applications and microbial physiology.
Non-microbiological aspects, such as reactor design and
materials, or descriptive studies documenting the com-
position of microbial communities colonizing electrodes
are not reviewed here.
Beyond power production: novel applicationsof microbe-electrode exchangeMicrobe-electrode systems designed for wastewater
treatment have to compete with mature, proven technol-
ogies, and require large capital investments. However,
there are other applications for microbial fuel cell tech-
nology where the competition may not be as intense,
especially when generation of electric power is not the
goal.
One area ripe for application is bioremediation of aquatic
sediments and groundwater. For example, inexpensive
but durable graphite electrodes deployed in sediments
not only serve as a low-maintenance, long-lived, desir-
able electron acceptor for anaerobic respiration, but also
can adsorb contaminants from the surrounding sediment,
co-localizing the contaminants, contaminant-degrading
microorganisms, and an electron acceptor in the same
location (Figure 1). This possibility of accelerating
anaerobic oxidation of organic contaminants in this man-
ner was first proposed back in 2002 (as reviewed in [20])
and has now been demonstrated for aromatic hydrocar-
bons [20], 1,2-dichloroethane [21], pyridine [22], phenol
[23], and possibly alkanes [24]. It seems likely that any
organic contaminants that microbes have been shown to
anaerobically oxidize with other electron acceptors can
be oxidized with electron transfer to an electrode.
Although it has been suggested that salt bridges would
need to be introduced into the subsurface to promote
proton flux between the anode and cathode for subsur-
face bioremediation with this technology [25], this is
unlikely [26�].
Electrodes deployed in anoxic subsurface environments
might also make it feasible to monitor microbial activity
Please cite this article in press as: Lovley DR, Nevin KP. A shift in the current: New applications an
j.copbio.2011.01.009
Current Opinion in Biotechnology 2011, 22:1–8
in groundwater in real time [26�]. As has previously been
observed in many other environments, Geobacter species
readily colonized graphite electrodes deployed in the
subsurface of a uranium-contaminated aquifer, producing
current levels that corresponded with the availability of
acetate added to the groundwater to promote U(VI)
reduction. This approach could be useful for monitoring
microbial activity in a diversity of environments, in-
cluding other planets [27].
Electron transfer from electrodes to cells may also have
bioremediation applications. Providing electrons to
microorganisms with electrodes shows substantial
promise as a strategy for bioremediation of groundwater
contaminated with metals or chlorinated solvents
(Figure 2). Geobacter-mediated cathode-driven bioreme-
diation of uranium-contaminated groundwater via reduc-
tive precipitation may be more effective, simpler, and less
expensive than the more familiar approach of enhancing
the activity of metal-reducing microorganisms with the
addition of organic compounds [1]. Other metal contami-
nants, such as Cr(VI), might be bioremediated in a similar
manner [28,29]. Promoting reductive dechlorination with
electrodes as the electron source [30–34] also has signifi-
cant potential advantages, but it seems important that this
be achieved with direct electron transfer to the dechlor-
inating microorganisms, rather than promoting electron
transfer with toxic electron shuttles or generating hydro-
gen at the cathode as has been done in some studies.
Other contaminants such as perchlorate [35] or nitrate
[36] may also be removed via reduction at the cathode.
Although the treatment of these contaminants was dis-
cussed in terms of wastewater treatment, these current-
d concepts for microbe-electrode electron exchange, Curr Opin Biotechnol (2011), doi:10.1016/
www.sciencedirect.com

A shift in the current: New applications and concepts for microbe-electrode electron exchange Lovley and Nevin 3
COBIOT-854; NO. OF PAGES 8
Figure 2
PowerSupply
PCE
PCE PCE
DCE
DCE DCEDCE
DCE
DCE
PCE
DCE
GW flow
Cathode
Anode
O2 CO2
H2O
e-
Current Opinion in Biotechnology
ContaminantSource Zone
Strategy for sequentially stimulating reductive dechlorination and aerobic degradation of partially dechlorinated products in the subsurface with solar
power.
driven microbial reduction may also be a viable ground-
water bioremediation strategy. Electrodes offer the
possibility of supplying electrons for bioremediation in
very specific locations and effectively co-localizing the
electron donor and the appropriate organisms, offering
the possibility of pre-colonizing the electrodes with the
desired organisms. The possibility of using solar technol-
ogy to sustainably generate the electricity necessary to
supply the electrons for such groundwater bioremediation
efforts is particularly attractive [32].
Increasing reliance on solar energy as a renewable source
of electricity is the major impetus for another cathode-
driven application, termed microbial electrosynthesis
(Box 1). Acetogenic microorganisms, which naturally
reduce carbon dioxide with hydrogen as the electron
donor to produce acetate as an endproduct, can substitute
an electrode as the electron donor for carbon dioxide
reduction, producing primarily acetate and small amounts
of other organic acids and alcohols (Nevin KP, Hensley
SA, Franks AE, Summers ZM, Ou J, Woodard TL,
Snoeyenbo-West OL, Lovley DR: Electrosynthesis of
organic compounds from carbon dioxide catalyzed by a
diversity of acetogenic microorganisms, unpublished
Please cite this article in press as: Lovley DR, Nevin KP. A shift in the current: New applications an
j.copbio.2011.01.009
www.sciencedirect.com
data) [37]. Recovery of electrons consumed in products
was high (ca. 85%) and ca. 70% of the electrical energy
expended was recovered in products. The cathode bio-
films of acetogens maintained sustained activity and
viability on the electrodes for months, without forming
thick biofilms, suggesting that the microbes gain small
amounts of energy from the reduction of carbon dioxide.
Acetyl-CoA, the central intermediate in acetate pro-
duction in acetogens, is the building block for microbial
synthesis of a wide diversity of desirable organic products
[38] and it has been demonstrated that it should be
possible with genetic engineering to divert carbon and
electron flow in acetogenic microbes toward the pro-
duction of butanol [39��], a fuel that can be transported
through existing pipelines and burned in automobile
engines without modification. Thus, microbial electro-
synthesis offers the possibility of converting renewable
but intermittent forms of energy, such as solar and wind,
into liquid transportation fuels or desirable chemicals
much more efficiently and with less potential environ-
mental degradation than biomass-based strategies (Box
1). It would be conceivable to use microbial electrosynth-
esis as a carbon sequestration process, producing organic
compounds that are resistant to degradation for long-term
d concepts for microbe-electrode electron exchange, Curr Opin Biotechnol (2011), doi:10.1016/
Current Opinion in Biotechnology 2011, 22:1–8

4 Environmental biotechnology
COBIOT-854; NO. OF PAGES 8
Box 1 Microbial electrosynthesis: artificial photosynthesis for
direct production of fuels
Electrosynthesis of organic compounds via abiotic or enzymatic
catalysis of carbon dioxide reduction at electrode surfaces has been
evaluated as a strategy for converting electricity into useful organic
products for some time [77,78]. Microbial catalysts might be better.
They are inexpensive to grow and, if the microbes catalyzing the
reactions gain enough energy for cell maintenance, are self-
sustaining and long-lived.
Microbial electrosynthesis can be powered with any source of
electricity, but when solar power is utilized the overall reaction
(CO2 + H2O! organic compounds + O2) powered by light is the
same as photosynthesis. However, microbial electrosynthesis is
ca. 100-fold more efficient than plants in converting solar energy
into organic compounds and can produce desired products
directly whereas producing fuels from biomass requires additional
energy inputs and only a fraction of the energy in the biomass is
recovered as fuel. Microbial electrosynthesis also does not require
arable land; the large quantities of water required for growing
biomass and then processing biomass to fuel; and can avoid the
environmental degradation associated with large-scale biomass
production. Thus, microbial electrosynthesis could transform the
bioenergy field because it offers the possibility of converting
renewable, but intermittent, sources of electricity into fuels or other
desirable organic compounds that are energy dense and can
readily be stored, distributed, and utilized within the existing
infrastructure [1,37].
carbon removal. However, the study of microbial electro-
synthesis is in its infancy and significant engineering of
the microbes and the reactors are required for practical
application.
It was suggested that some methanogens could also
directly accept electrons from electrodes, catalyzing the
reduction of carbon dioxide to methane [40], but sub-
sequent studies have questioned whether hydrogen pro-
duced at the cathode was the actual electron donor [1,41]
because hydrogen is typically produced at the low poten-
tials that were required for active methanogenesis. Micro-
organisms accepting electrons from cathodes may
catalyze proton reduction to hydrogen gas with hydro-
genases [14��,42,43]. Pure cultures that are capable of
reducing protons with electrons derived from electrodes
have not yet been described, but such organisms could
eliminate the need for expensive platinum catalysts for
hydrogen production.
Microorganisms can promote electron transfer to oxygen
on cathodes [1,2��]. This may not be linked to aerobic
respiration [44,45], but rather the result of interaction of
oxygen with a diversity of reduced cell components.
There is often an enrichment of specific microorganisms
on the cathodes of microbial fuel cells deployed in open
environments, and thus it is not clear whether preemptive
colonization of cathodes with microorganisms highly
effective in catalyzing oxygen reduction would improve
the long-term kinetics of oxygen reduction in microbial
fuel cells for most practical applications.
Please cite this article in press as: Lovley DR, Nevin KP. A shift in the current: New applications an
j.copbio.2011.01.009
Current Opinion in Biotechnology 2011, 22:1–8
Mechanisms for microbe-electrode electronexchangeOptimization of any of the applications discussed above, as
well as conceptualization of novel applications, will prob-
ably profit from an understanding of how microorganisms
exchange electrons with the electrode surface. The dis-
cussion here will be limited to mechanistic studies with
defined pure cultures for which the results are more readily
interpreted. Pure culture studies have primarily focused on
Geobacter species, chosen because they are often the pre-
dominant organisms that naturally colonize high current
density anodes in a diversity of environments, or Shewanellaspecies, which are rarely found as anode inhabitants, but
many consider easier to cultivate in the laboratory because
they can also use oxygen as an electron acceptor.
Geobacter sulfurreducens has been intensively studied
because it is often cited as the species most closely related
to those Geobacter species that predominate on anodes and
because it produces the highest current density of any
known pure culture [46,47]. Electrochemical studies [48–50] have confirmed multiple lines of earlier research [51]
that soluble electron shuttles are not important for elec-
tron transfer from G. sulfurreducens to anodes. Despite this,
cells at the outer edge of the relatively thick (>50 mm)
anode biofilms of G. sulfurreducens appear to be metabo-
lically active [52] as are cells near the anode surface, even
though they generate relatively low pH [53]. Microarray
analysis of gene expression and gene deletion studies
suggest that the electrically conductive pili and the c-type
cytochrome OmcZ are essential for optimal current pro-
duction [54]. Adaptive evolution studies in which there
was strong selective pressure for enhanced current pro-
duction yielded a hyperpiliated strain [46]. Characteriz-
ation of OmcZ demonstrated that it contained eight
hemes with potentials ranging from �420 to �60 mV
[55]. Localization of OmcZ with gold-labeled antibodies
and transmission electron microscopy revealed that
OmcZ was not closely associated with cells and accumu-
lated near the anode surface [56]. This is consistent with
the prediction of increased c-type cytochromes at the
biofilm-electrode interface [57,58]. These findings,
coupled with electrochemical analyses [49], suggest that
OmcZ functions as an ‘electrochemical gate’ to promote
electron transfer onto the electrode [56]. This model
(Figure 3a) of long-range electron conduction via pili
and then electron transfer onto the electrode via OmcZ,
is similar to the model for Fe(III) oxide reduction
(Figure 3b), in which electrons are conducted away from
the cell via pili and then electrical contact between pili
and Fe(III) oxide is facilitated by the c-type cytochrome
OmcS, which is localized along the pili [59�]. In both
instances, long-range extracellular transfer is along pili
and c-type cytochromes are required to promote the final
transfer to the insoluble electron acceptor. The pathway
for electron transfer from the inner membrane to the
outer cell surface during electron transfer to electrodes is
d concepts for microbe-electrode electron exchange, Curr Opin Biotechnol (2011), doi:10.1016/
www.sciencedirect.com

A shift in the current: New applications and concepts for microbe-electrode electron exchange Lovley and Nevin 5
COBIOT-854; NO. OF PAGES 8
Figure 3
(a) (b)
(c)
Pilin-MediatedConductionThroughBiofilm
OmcZ-MediatedElectron Transfer fromBiofilm to Anode
Electron Transfer Along Pili with OmcS-Mediated Electron Transfer to Fe(III) Oxide
OxidizedFlavin
ReducedFlavin
ElectrodeorFe(III) Oxide
Current Opinion in Biotechnology
Models for the predominant mechanisms for electron transfer to electrodes (a) and Fe(III) oxides (b) by Geobacter sulfurreducens and electron transfer
to electrodes and Fe(III) oxides by Shewanella oneidensis (c).
considered to involve a series of periplasmic and outer-
membrane c-type cytochromes. The abundant c-type
cytochromes also appear to act as capacitors to store
electrons when electron transfer is impeded [58,60�], as
previously proposed [61]. Other redox-active proteins
such as multi-copper proteins may also be important in
the electron transfer [62]. Electrochemical analysis
suggests that the final electron transfer from Geobacterbiofilms to the anode is fast enough to permit cells in the
biofilm to respire as fast as they can with soluble Fe(III) as
the electron acceptor [60�], suggesting that engineering
Geobacter to respire faster [63], or to pack more cells into
the biofilm, could result in higher current densities.
The current model for extracellular electron transfer in
Shewanella oneidensis is significantly different (Figure 3c).
Electron transfer to electrodes does not require direct
contact [64,65] as had previously been demonstrated for
Fe(III) oxides, and although S. oneidensis also has con-
ductive pili [66], they are not required for electron trans-
fer to Fe(III) or electrodes [67��]. The primary outer-
surface c-type cytochromes required for reduction of
Fe(III) and electrodes appear to be associated with the
cell surface, not pili [68] and although they may be able to
poorly transfer electrons to insoluble electron acceptors,
the primary role of the cytochromes appears to be the
Please cite this article in press as: Lovley DR, Nevin KP. A shift in the current: New applications an
j.copbio.2011.01.009
www.sciencedirect.com
reduction of flavins which are released from the cell and
serve as an electron shuttle for Fe(III) and electrode
reduction [69,70��,71,72��].
Electron transfer from electrodes to microbes may not be
a simple reversal of electron transfer from cells to elec-
trodes. Current-consuming biofilms of G. sulfurreducenshad much lower expression of genes for pili and OmcZ,
which are essential for optimal current production, than
current-producing biofilms and deletion of the genes for
pili or OmcZ production had no impact on the capacity for
current uptake [73]. One of the most highly up-regulated
genes in current-consuming biofilms was a gene for a
putative, mono-heme c-type cytochrome predicted to be
located in the periplasm [73]. Deleting the gene for this
cytochrome had no impact on the capacity for current
production, but completely inhibited the ability of cells to
accept electrons from electrodes [73].
ConclusionsAs the understanding of microbe-electrode electron
exchange improves, it is becoming apparent that producing
electric current may not be the most important, short-term,
practical application of this phenomenon. In fact, some of
the most promising microbe-electrode technologies may
require aninput ofelectricalpower. Additionalapplications,
d concepts for microbe-electrode electron exchange, Curr Opin Biotechnol (2011), doi:10.1016/
Current Opinion in Biotechnology 2011, 22:1–8

6 Environmental biotechnology
COBIOT-854; NO. OF PAGES 8
not only in environmental biotechnology and bioenergy,
but also in other fields, are likely to continue to emerge.
Furthermore, understanding how microorganisms electro-
nically interact with electrodes may provide important
insights into how microorganisms may electronically inter-
act with conductive materials [74�] or other cells [75] in
natural environments, which should be helpful in under-
standing interesting phenomena, such as apparent rapid
electron transfer through marine sediments [76�]. Thus, for
both natural science and practical applications, a basic un-
derstanding of how cells exchange electrons with materials
outside the cell is essential.
References and recommended readingPapers of particular interest, published within the period of review,have been highlighted as:
� of special interest
�� of outstanding interest
1. Lovley DR: Powering microbes with electricity: direct electrontransfer from electrodes to microbes. Environ Microbiol Rep2010 doi: 10.1111/j.1758-2229.2010.00211.x.
2.��
Rosenbaum M, Aulenta F, Villano M, Angenent LT: Cathodes aselectron donors for microbial metabolism: which extracellularelectron transfer mechanisms are involved? BioresourceTechnol 2010 doi: 10.1016/j.biortech.2010.07.008.
Thought-provoking speculation on the potential mechanisms for electrontransfer from cathodes to microbes.
3. Lovley DR, Nevin KP: Electricity production with electricigens.In Bioenergy. Edited by Wall JD, Harwood CS, Demain AL. ASMPress; 2008:295-306.
4.��
Pant D, Van Bogaert G, Diels L, Vanbroekhoven K: A review ofsubstrates use in microbial fuel cells (MFCs) for sustainableenergy production. Bioresource Technol 2010, 101:1533-1543.
Excellent summary of the range of electron donors that can be oxidized inmicrobial fuel cells.
5. Tender LM, Gray SM, Groveman E, Lowy DA, Kauffman P,Melhado J, Tyce RC, Flynn D, Petrecca R, Dobarro J: The firstdemonstration of a microbial fuel cell as a viable powersupply: powering a meterological buoy. J Power Sources 2008,179:571-575.
6. Nielsen ME, Reimers CE, White HK, Sharma S, Girguis PR:Sustainable energy from deep ocean cold seeps. EnergyEnviron Sci 2008, 1:584-593.
7. Donovan C, Dewan A, Heo D, Beyenal H: Batteryless wirelesssensor powered by a sediment microbial fuel cell. Environ SciTechnol 2008, 42:8591-8596.
8. Dewan A, Donovan C, Heo D, Beyenal H: Evaluating theperformance of microbial fuel cells powering electronicdevices. J Power Sources 2010, 195:90-96.
9. Logan BE: Exoelectrogenic bacteria that power microbial fuelcells. Nat Rev Microbiol 2009, 7:375-381.
10.��
Dewan A, Beyenal H, Lewandowski Z: Scaling up microbial fuelcells. Environ Sci Technol 2008, 42:7643-7648.
Demonstration that microbial fuel cells do not scale in a linear manner.
11.��
Foley JM, Rozendal RA, Hertle CK, Lant PA, Rabaey K: Life cycleassessment of a high-rate anaerobic treatment, microbial fuelcells and microbial electrolysis cells. Environ Sci Technol 2010,44:3629-3637.
Insights into the economic realities of wastewater treatment with micro-bial fuel cell technology.
12. Cusick RD, Kiely PD, Logan BE: A monetary comparison ofenergy recovered from microbial fuel cells and microbialelectrolysis cells fed winery or domestic wastewaters. Int JHydrogen Energy 2010, 35:8855-8861.
Please cite this article in press as: Lovley DR, Nevin KP. A shift in the current: New applications an
j.copbio.2011.01.009
Current Opinion in Biotechnology 2011, 22:1–8
13. Rabaey K, Rozendal RA: Microbial electrosynthesis-revisitingthe electrical route for microbial production. Nat Rev Microbiol2010, 8:706-716.
14.��
Geelhoed JS, Hamelers HVM, Stams AJM: Electricity-mediatedbiological hydrogen production. Curr Opin Microbiol 2010,13:307-315.
Excellent review of this novel bioenergy strategy.
15. Rozendal RA, Leone E, Keller J, Rabaey K: Efficient hydrogenperoxide generation from organic matter in a bioelectricalsystem. Electrochem Commun 2009, 11:1752-1755.
16. Rabaey K, Butzer S, Brown S, Keller J, Rozendal RA: High currentgeneration coupled to caustic production using a lamellarbioelectrochemical system. Environ Sci Technol 2010,44:4315-4321.
17. Mehanna M, Saito T, Yan J, Hickner M, Cao X, Huang X, Logan BE:Using microbial desalination cells to reduce water salinityprior to reverse osmosis. Energy Environ Sci 2010, 3:1114-1120.
18. Thrash JC, Coates JD: Review: direct and indirect electricalstimulation of microbial metabolism. Environ Sci Technol 2008,42:3921-3931.
19. Lovley DR: The microbe electric: conversion of organic matterto electricity. Curr Opin Biotechnol 2008, 19:564-571.
20. Zhang T, Gannon SM, Nevin KP, Franks AE, Lovley DR:Stimulating the anaerobic degradation of aromatichydrocarbons in contaminated sediments by providing anelectrode as the electron acceptor. Environ Microbiol 2010,12:1011-1020.
21. Pham H, Boon N, Marzorati M, Verstraete W: Enhanced removalof 1,2-dichloroethane by anodophilic microbial consortia.Water Res 2009, 43:2936-2946.
22. Zhang C, Li M, Liu G, Luo H, Zhange R: Pyridine degradation inthe microbial fuel cells. J Hazard Mater 2009, 172:465-471.
23. Luo H, Liu G, Zhang R, Jin S: Phenol degradation in microbialfuel cells. Chem Eng J 2009, 147:259-264.
24. Morris JM, Jin S, Pruden A: Microbial fuel cell in enhancinganaerobic biodegradation of diesel. Chem Eng J 2009,146:161-167.
25. Morris JM, Jin S: Feasibility of using microbial fuel celltechnology for bioremediation of hydrocarbons ingroundwater. J Environ Sci Health Part A 2008,43:18-23.
26.�
Williams KN, Nevin KP, Franks AE, Englert A, Long PE, Lovley DR:Electrode-based approach for monitoring in situ microbialactivity during subsurface bioremediation. Environ Sci Technol2010, 44:47-54.
Demonstration that current can be produced with long-range separationof anode and cathode in the subsurface and introduction of a newconcept for estimating rates of microbial metabolism in the subsurface.
27. Miller LG, Oremland RS: Electricity generation by anaerobicbacteria and anoxic sediments from hypersaline soda lakes.Extremophiles 2008, 12:837-848.
28. Tandukar M, Huber SJ, Onodera T, Pavlostathis SG: Biologicalchromium(VI) reduction in the cathode of a microbial fuel cell.Environ Sci Technol 2009, 43:8159-8165.
29. Huang L, Chen J, Quan X, Yang F: Enhancement of hexavalentchromium reduction and electricity production from abiocathode microbial fuel cell. Bioprocess Biosyst Eng 2010,33:937-945.
30. Aulenta F, Canosa A, De Roma L, Reale P, Panero S, Rossetti S,Majone M: Influence of mediator immobilization on theelectrochemically assisted microbial dechlorination oftrichlroethene (TCE) and cis-diechloroethene (cis-DCE). JChem Technol Biotechnol 2009, 84:864-870.
31. Aulenta F, Canosa A, Reale P, Rossetti S, Panero S, Majone M:Microbial reductive dechlorination of trichloroethene toethene with electrodes serving as electron donors without theexternal addition of redox mediators. Biotechnol Bioeng 2009,101:85-91.
d concepts for microbe-electrode electron exchange, Curr Opin Biotechnol (2011), doi:10.1016/
www.sciencedirect.com

A shift in the current: New applications and concepts for microbe-electrode electron exchange Lovley and Nevin 7
COBIOT-854; NO. OF PAGES 8
32. Strycharz SM, Woodward TL, Johnson JP, Nevin KP, Sanford RA,Loeffler FE, Lovley DR: Graphite electrode as a sole electrondonor for reductive dechlorination of tetrachlorethene byGeobacter lovleyi. Appl Environ Microbiol 2008, 74:5943-5947.
33. Strycharz SM, Gannon SM, Boles AR, Nevin KP, Franks AE,Lovley DR: Anaeromyxobacter dehalogens interacts with apoised graphite electrode for reductive dechlorination of 2-chlorophenol. Environ Microbiol Rep 2010:289-294.
34. Aulenta F, Reale P, Canosa A, Rossetti S, Panero S, Majone M:Characterization of an electro-active biocathode capable ofdechlorinating trichloroethene to ethene. Biosens Bioelectron2010, 25:1796-1802.
35. Butler C, Clauwaert P, Green SJ, Verstraete W, Nerenberg R:Bioelectrochemical perchlorate reduction in a microbial fuelcell. Environ Sci Technol 2010, 44:4685-4691.
36. Wrighton KC, Virdis B, Clauwaert P, Read ST, Daly RA, Boon N,Piceno Y, Andersen GL, Coates JD, Rabaey K: Bacterialcommunity structure corresponds to performance duringcathodic nitrate reduction. ISME J 2010, 4:1443-1455.
37. Nevin KP, Woodard TL, Franks AE, Summers ZM, Lovley DR:Microbial electrosynthesis: feeding microbes electricityto convert carbon dioxide and water to multicarbonextracellular organic compounds. mBio 2010,1: doi: 10.1128/mBio.00103-00110.
38. Conner MR, Liao JC: Microbial production of advancedtransportation fuels in non-natural hosts. Curr Opin Biotechnol2009, 20:307-315.
39.��
Kopke M, Held C, Hujer S, Liesegang H, Wiezer A, Wolherr A,Ehrenreich A, Liebl W, Gottschalk G, Durre P: Clostridiumljungdahlii represents a microbial production platform basedon syngas. Proc Natl Acad Sci USA 2010, 107:13087-13092.
Demonstrates the promise of whole-genome sequencing and geneticmanipulation for novel bioenergy strategies with acetogenic bacteria.
40. Cheng S, Xing D, Call DF, Logan BE: Direct biological conversionof electrical current into methane by electromethanogenesis.Environ Sci Technol 2009, 43:3953-3958.
41. Villano M, Aulenta F, Ciucci C, Ferri T, Giuliano A, Majone M:Bioelectrochemical reduction of CO2 to CH4 via direct andindirect extracellular electron transfer by a hydrogenophilicmethanogenicculture.BioresourceTechnol2010,101:3085-3090.
42. Rozendal RA, Jeremiasse AW, Hamelers HVM, Buisman CJN:Hydrogen production with a microbial biocathode. Environ SciTechnol 2008, 42:629-634.
43. Jeremiasse AW, Hamelers HVM, Buisman CJN: Microbialelectrolysis cell with a microbial biocathode.Bioelectrochemistry 2010, 78:39-43.
44. Parot S, Nercessian O, Delia M-L, Achouak W, Bergel A:Electrochemical checking of aerobic isolates fromelectrochemically active biofilms formed in compost. J ApplMicrobiol 2009, 106:1350-1390.
45. Freguia S, Tsujimura S, Kano K: Electron transfer pathwaysin microbial oxygen biocathodes. Electrochim Acta 2010,55:813-818.
46. Yi H, Nevin KP, Kim B-C, Franks AE, Klimes A, Tender LM,Lovley DR: Selection of a variant of Geobacter sulfurreducenswith enhanced capacity for current production in microbialfuel cells. Biosens Bioelectron 2009, 24:3498-3503.
47. Nevin KP, Richter H, Covalla SF, Johnson JP, Woodard TL, Jia H,Zhang M, Lovley DR: Power output and columbic efficienciesfrom biofilms of Geobacter sulfurreducens comparable tomixed community microbial fuel cells. Environ Microbiol 2008,10:2505-2514.
48. Marsili E, Baron DB, Shikhare I, Coursolle D, Gralnick JA, Bond DR:Shewanella secretes flavins that mediate extracellularelectron transfer. PNAS 2008, 105:3968-3973.
49. Richter H, Nevin KP, Jia H, Lowy DA, Lovley DR, Tender LM:Cyclic voltammetry of biofilms of wild type and mutantGeobacter sulfurreducens on fuel cell anodes indicatespossible roles of OmcB, OmcZ, type IV pili, and protons
Please cite this article in press as: Lovley DR, Nevin KP. A shift in the current: New applications an
j.copbio.2011.01.009
www.sciencedirect.com
in extracellular electron transfer. Energy Environ Sci 2009,2:506-516.
50. Marsili E, Rollefson JB, Baron DB, Hozalski RM, Bond DR:Microbial biofilm voltammetry: direct electrochemicalcharacterization of catalytic electrode-attached biofilms. ApplEnviron Microbiol 2008, 74:7329-7337.
51. Lovley DR: Extracellular electron transfer: wires, capacitors,iron lungs, and more. Geobiology 2008, 6:225-231.
52. Franks AE, Nevin KP, Glaven RH, Lovley DR: Microtomingcoupled with microarray analysis to evaluate potentialdifferences in the metabolic status of Geobactersulfurreducens at different depths in anode biofilms. ISME J2010, 4:509-519.
53. Franks AE, Nevin KP, Jia H, Izallalen M, Woodard TL, Lovley DR:Novel strategy for three-dimensional real-time imaging ofmicrobial fuel cell communities: monitoring the inihibtoryeffects of proton accumulation within the anode biofilm.Energy Environ Sci 2009, 2:113-119.
54. Nevin KP, Kim B-C, Glaven RH, Johnson JP, Woodard TL,Methe BA, DiDonato RJ Jr, Covalla SF, Franks AE, Liu A et al.:Anode biofilm transcriptomics reveals outer surfacecomponents essential for high currency power production inGeobacter sulfurreducens fuel cells. PLoS ONE 2009, 4:e5628.
55. Inoue K, Qian X, Morgado L, Kim B-C, Mester T, Izallalen M,Salgueiro CA, Lovley DR: Purification and characterization ofOmcZ an outer-surface, octaheme, c-type cytochromeessential for optimal current production by Geobactersulfurreducens. Appl Environ Microbiol 2010, 76:3999-4007.
56. Inoue K, Leang C, Franks AE, Woodard TL, Nevin KP, Lovley DR:Specific localization of the c-type cytochrome OmcZ at theanode surface in current-producing biofilms of Geobactersulfurreducens. Environ Microbiol Rep 2010. 10.1111/j.1758-2229.2010.00210.x.
57. Busalmen JP, Esteve-Nunez A, Berna A, Feliu JM: C-typecytochromes wire electricity-producing bacteria toelectrodes. Angnew Chem Int Ed 2008, 47:4874-4877.
58. Busalmen JP, Esteve-Nunez A, Berna A, Feliu JM: ATR-SEIRAscharacterization of surface redox processes in G.sulfurreducens. Bioelectrochemistry 2010, 78:25-29.
59.�
Leang C, Qian X, Mester T, Lovley DR: Alignment of the c-typecytochrome OmcS along pili of Geobacter sulfurreducens.Appl Environ Microbiol 2010, 76:4080-4084.
New model for coupling electron flow through pili and cytochromes forextracellular electron transfer.
60.�
Marsili E, Sun J, Bond DR: Voltammetry and growth physiologyof Geobacter sulfurreducens biofilms as a function ofgrowth stage and imposed potential. Electroanalysis 2010,22:865-874.
In depth analysis of electron transfer through Geobacter sulfurrreducensanode biofilms identifying the key aspects of this process.
61. Esteve-Nunez A, Sosnik J, Visconti P, Lovley DR:Fluorescent properties of c-type cytochromes revealtheir potential role as an extracytoplasmic electronsink in Geobacter sulfurreducens. Environ Microbiol 2008,10:497-505.
62. Holmes DE, Mester T, O’Neil RA, Larrahondo MJ, Adams LA,Glaven R, Sharma ML, Ward JA, Nevin KP, Lovley DR: Genes fortwo multicopper proteins required for Fe(III) oxide reduction inGeobacter sulfurreducens have different expression patternsboth in the subsurface and on energy-harvesting electrodes.Microbiology 2008, 145:1422-1435.
63. Izallalen M, Mahadevan R, Burgard A, Postier B, DiDonato R,Sun J, Schilling CH, Lovley DR: Geobacter sulfurreducens strainengineered for increased rates of respiration. Metabolic Eng2008, 10:267-275.
64. Lanthier M, Gregory KB, Lovley DR: Electron transfer toelectrodes with high planktonic biomass in Shewanellaoneidensis fuel cells. FEMS Microbiol Lett 2008, 278:29-35.
65. Jiang X, Hu J, Fitzgerald LA, Biffinger JC, Xie P, Ringeisen BR,Lieber CM: Probing electron transfer mechanisms in
d concepts for microbe-electrode electron exchange, Curr Opin Biotechnol (2011), doi:10.1016/
Current Opinion in Biotechnology 2011, 22:1–8

8 Environmental biotechnology
COBIOT-854; NO. OF PAGES 8
Shewanella oneidensis MR-1 using a nanoelectrode platformand single-cell imaging. Proc Natl Acad Sci USA 2010 http://www.pnas.org/cgi/doi/10.1073/pnas.1011699107.
66. El-Naggar MY, Gorby YA, Xia W, Nealson KH: The moleculardensity states in bacterial nanowires. Biophys J 2008,95:L10-L12.
67.��
Bouhenni RA, Vora GJ, Biffinger JC, Shirodkar S, Brockman K,Ray R, Wu P, Johnson BJ, Biddle EM, Marshall MJ et al.:The role of Shewanella oneidensis MR-1 outer surfacestructures in extracellular electron transfer. Electroanalysis2010, 22:856-864.
Important study using genetic approach to rigorously test several modelsfor extracellular electron transfer.
68. Shi L, Richardson DJ, Wang Z, Kerisit SN, Rosso KM, Zachara JM,Fredrickson JK: The roles of outer membrane cytochromes ofShewanella and Geobacter in extracellular electron transfer.Environ Microbiol Rep 2009, 1:220-227.
69. Peng L, You S-J, Wang J-Y: Electrode potential regulatescytochrome accumulation on Shewanella oneidensis cellsurface and the consequence to bioelectrocatalytic currentgeneration. Biosens Bioelectron 2010, 25:2530-2533.
70.��
Ross DE, Brantley SL, Tien M: Kinectic characterization ofOmcA and MtrC, terminal reductases involved in respiratoryelectron transfer for dissimilatory iron reduction inShewanella oneidensis MR-1. Appl Environ Microbiol 2009,75:5218-5226.
Demonstrates likely in vivo role of outer surface cytochromes in aquantitative manner.
71. Coursolle D, Baron DB, Bond DR, Gralnick JA: The Mtr respiratorypathway is essential for reducing flavins and electrodes inShewanella oneidensis. J Bacteriol 2010, 192:467-474.
72.��
Baron DB, LaBelle E, Coursolle D, Gralnick JA, Bond DR:Electrochemical measurements of electron transfer
Please cite this article in press as: Lovley DR, Nevin KP. A shift in the current: New applications an
j.copbio.2011.01.009
Current Opinion in Biotechnology 2011, 22:1–8
kinetics by Shewanella oneidensis MR-1. J Biol Chem 2009,284:28865-28873.
Just one of a series of studies by the Minnesota team that has eloquentlydemonstrated the power of combining electrochemical and more tradi-tional physiological studies to elucidate extracellular electron transfermechanisms.
73. Strycharz SM, Glaven RH, Coppi MV, Gannon SM, Perpetua LA,Liu A, Nevin KP, Lovley DR: Gene expression and deletionanalysis of mechanisms for electron transfer fromelectrodes to Geobacter sulfurreducens. Bioelectrochemistry2010 doi: 10.1016/j.bioelechem.2010.07.005.
74.�
Kato S, Nakamura R, Kai F, Wantanabe K, Hashimoto K:Respiratory interactions of soil bacteria with(semi)conductive iron-oxide minerals. Environ Microbiol 2010doi: 10.1111/j.1462-2920.2010.02284.x.
Novel concept for electron transfer in sedimentary environments.
75. Summers ZM, Fogarty H, Leang C, Franks AE, Malvankar NS,Lovley DR: Direct exchange of electrons within aggregates ofan evolved syntrophic co-culture of anaerobic bacteria.Science 2010, 330:1413-1415.
76.�
Nielsen LP, Risgaard-Petersen N, Fossing H, Christensen PB,Sayama M: Electric currents couple spatially separatedbiogeochemical processes in marine sediment. Nature 2010,463:1071-1074.
Surprising finding of apparent rapid long-range electron conductionthrough marine sediments.
77. Cole EB, Bocarsly AB: Photochemical, electrochemical, andphotochemical reduction of carbon dioxide. In Carbon Dioxideas Chemical Feedstock. Edited by Aresta M.. Wiley-VCH VerlagGmbH & Co; 2010:291-316.
78. Centi G, Perathoner S: Opportunities and prospects in thechemical recycling of carbon dioxide to fuels. Catal Today2009, 148:191-205.
d concepts for microbe-electrode electron exchange, Curr Opin Biotechnol (2011), doi:10.1016/
www.sciencedirect.com
Related Documents