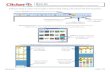
5/21/2018 10-22-13TranslationClickerSupplement-slidepdf.com http://slidepdf.com/reader/full/10-22-13-translation-clicker-supplement 1/18 Clicker Question If a codon consists of 3 nucleic acids, how many possible different codons exist? A. 3 B. 16 C. 64 (4x4x4) D. 128 E. 8675309

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript
Found in translation: from mRNA to protein
Clicker QuestionIf a codon consists of 3 nucleic acids, how many possible different codons exist?316 64 (4x4x4)1288675309
Codons: What a bunch of degenerates!comprised of 3 consecutive nucleotides
written 5 to 3
4 nucleotides (U, C, A, G) so there are 43 (or 64) codons
3 codons signal for stop (stopping translation)
So there are 64 - 3 = 61 different codons for amino acids
Remember, there are 20 amino acids so some amino acids must be encoded by more than one codon = the code is degenerateUniversal decoder chart
See Figure 11.40 on page 463 of Karp.Clicker QuestionIf a codon for serine is 5-AGC-3, what is the anticodon on the tRNA for this triplet? 5-AGC-3 5-UCG-3 5-GCU-3 5-GCT-3 5-SOS-3
In Class ActivityWrite a possible DNA sequence that would produce a protein with the following amino acid sequence: N-M-Y-P-R-D-E-C
mRNA: 5- AUG CCA UAC AGA GAC GAA UAA -3DNA: 3- TAC GGT ATG TCT CTG CTT ATT -5
OrDNA: 5- TTA TTC GTC TCT GTA TGG CAT -3In Class ActivityWrite a possible DNA sequence that would produce a protein with the following amino acid sequence: N-M-Y-P-R-D-E-CREMOVE6Synthesis and Processing of Messenger RNAsSplit Genes: An Unexpected FindingThe difference between heterogeneous nuclear RNA (hnRNA) and mRNA provided early clues about RNA processing.Eukaryotic genes contain intervening sequences which are missing from mature mRNAs.The presence of genes with intervening sequences are called split genes.The difference in size betweenhnRNAs and mRNAs
The discovery of introns in a eukaryotic gene
Figure 11.24cDNA is a DNA made in vitro by reverse transcriptase using the mRNA as a template.Figure 11.24 The discovery of introns in a eukaryotic gene. As discussed in Chapter 18, bacteria contain restriction enzymes that recognize and cleave DNA molecules at the site of certain nucleotide sequences. The drawing shows a map of restriction enzyme cleavage sites in the region of the rabbit - globin gene (upper) and the corresponding map of a cDNA prepared from the - globin mRNA (lower). A cDNA is a DNA made in vitro by reverse transcriptase using the mRNA as a template. Thus, the cDNA has the complementary sequence to the mRNA. cDNAs had to be used for this experiment be-cause restriction enzymes dont cleave RNAs.) The letters indicate the sites at which various restriction enzymes cleave the two DNAs. The upper map shows that the globin gene contains a restriction site for the enzyme BamH1 (B) located approximately 700 base pairs from a restriction site for the enzyme EcoR1 (E). When the globin cDNA was treated with these same enzymes (lower map), the corresponding B and E sites were located only 67 nucleotides apart. It is evident that the DNA prepared from the genome has a sizeable region that is absent from the corresponding cDNA (and thus absent from the mRNA from which the cDNA was produced.9Pre-mRNA transcripts areprocessed cotranscriptionally
Figure 11.27Pre- mRNA transcripts are processed as they are synthesized Figure 11.27 Pre- mRNA transcripts are processed as they are synthesized ( i. e., cotran-scriptionally). (a) Electron micrograph of a nonribosomal transcription unit showing the presence of ribonucleoprotein particles attached to the nascent RNA transcripts. (b) Interpretive tracing of the micrograph shown in part a. The dotted line represents the chromatin (DNA) strand, the solid lines represent ribonucleopro-tein (RNP) fibrils, and solid circles represent RNP particles associated with the fibrils. Individual transcripts are numbered, beginning with 1, which is closest to the point of initiation. The RNP particles are not distributed randomly along the nascent transcript, but rather are bound at specific sites where RNA processing is taking place.10Synthesis and Processing of Messenger RNAsThe pre-mRNA is typically not capable of self-splicingrequires small nuclear RNAs (snRNAs).As each hnRNA is transcribed, it becomes associated with a heterogeneous ribonucleoprotein particle (hnRNP).Processing occurs as each intron becomes associated with a complex called spliceosome.The spliceosome consists of small nuclear ribonucleoproteins (snRNPs).Heterogeneous nuclear ribonucleoproteins (hnRNPs) are complexes of RNA and protein present in the cell nucleus during gene transcription and subsequent post-transcriptional modification of the newly synthesized RNA (pre-mRNA). The presence of the proteins bound to a pre-mRNA molecule serves as a signal that the pre-mRNA is not yet fully processed and ready for export to the cytoplasm. Since most mature RNA is exported from the nucleus relatively quickly, most RNA-binding protein in the nucleus exist as heterogeneous ribonucleoprotein particles. After splicing has occurred, the proteins remain bound to spliced introns and target them for degradation.The proteins involved in the hnRNP complexes are collectively known as heterogeneous ribonucleoproteins. They include protein K and polypyrimidine tract-binding protein (PTB), which is regulated by phosphorylation catalyzed by protein kinase A and is responsible for suppressing RNA splicing at a particular exon by blocking access of the spliceosome to the polypyrimidine tract.[1]:32611Model of the assembly of the splicing machinery
Figure 11.32Figure 11.32 Schematic model of the assembly of the splicing machinery and some of the steps that occur during pre- mRNA splicing. Step 1 shows the portion of the pre-mRNA to be spliced. In step 2, the first of the splicing components, U1 snRNP, has be-come attached at the 5 splice site of the intron. The nucleotide se-quence of U1 snRNA is comple-mentary to the 5 splice site of the pre- mRNA, and evidence in-dicates that U1 snRNP initially binds to the 5 side of the intron by the formation of specific base pairs between the splice site and U1 snRNA ( see inset A). The U2 snRNP is next to enter the splic-ing complex, binding to the pre-mRNA ( as shown in inset A) in a way that causes a specific adeno-sine residue ( dot) to bulge out of the surrounding helix ( step 3). This is the site that later becomes the branch point of the lariat. U2 is thought to be recruited by the protein U2AF, which binds to the polypyrimidine tract near the 3 splice site. U2AF also interacts with SR proteins that bind to the ex-onic splicing enhancers ( ESEs).12A mechanism for the coordination of transcription, capping, polyadenylation, and splicing.
Figure 11.34Figure 11.34 Schematic representation of a mechanism for the coordination of transcription, capping, polyadenylation, and splicing. In this simplified model, the C- terminal domain (CTD) of the large subunit of the RNA polymerase (page 443) serves as a flexible scaffold for the organization of factors involved in processing pre- mRNAs, including those for capping, polyadenylation, and intron removal. In addition to the proteins depicted here, the polymerase is probably associated with a host of transcription factors, as well as enzymes that modify the chromatin template. The proteins bound to the polymerase at any particular time may depend on which of the serine residues of the CTD are phosphorylated. The pattern of phosphorylated serine residues changes as the polymerase proceeds from the beginning to the end of the gene being transcribed (compare to Figure 11.20). The phosphate groups linked to the # 5 residues are largely lost by the time the polymerase has transcribed the 3 end of the RNA. 13Maturation of the mRNA - reviewModifications on the 5 and 3 ends occur in nearly all eukaryotic mRNAs5 ends usually have a short methylguanosine cap and often a noncoding region3 ends often have a noncoding region and a poly-adenosine (polyA) tail
Mutations & their effects on protein synthesisBase-pair substitutions, insertions, and deletionsframe-shift many amino acids affectednon frame-shift only one amino acid affectedmissense add start or stop
Frameshift with major effect
Clicker QuestionWhich mutation would have an effect on the corresponding amino acid sequence: 5 - CGAUGAAGGCAUAAGC - 3
5 - CCAUGAAGGCAUAAGC - 35 - CGAUGAAGGUCAUAAGC - 35 - CGAUGAAGGCAUAAUC - 35 - CGAUGAAGGCCUAAGC - 3
Clicker QuestionWhich mutation would have an effect on the corresponding amino acid sequence: 5 - CGAUGAAGGCAUAAGC - 3
5 - CCAUGAAGGCAUAAGC - 35 - CGAUGAAGGUCAUAAGC - 35 - CGAUGAAGGCAUAAUC - 35 - CGAUGAAGGCCUAAGC - 3
Related Documents