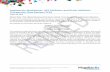
TNFα Inhibitors Therapeutic Drug Monitoring Compiled by Prof E Vardas NEWSLETTER 3 Quarter 2017 rd Introduction Tumour necrosis factor alpha (TNFα) is a pro-inflammatory cytokine involved in the pathogenesis of several chronic inflammatory conditions. The introduction of monoclonal antibody derived biologic drugs targeting TNFα has redefined the treatment of multiple chronic inflammatory diseases (see Figure 1) and has led to considerable improvement in clinical outcomes for patients with these conditions. TNFα inhibitors (TNFi) These biologic drugs are monoclonal antibodies of various designs (see Figure 2). Infliximab (Revellex ) was the first TNFα inhibitor to be approved for the treatment of inflammatory bowel disorders. Infliximab is a chimeric monoclonal IgG1 antibody composed of a murine variable region and a constant human region. Alternative tradenames for many of these monoclonal antibody drugs exist because biosimilars produced by other manufacturers, after the patent of the original drug has expired, have entered the market. Biosimilars contain a similar version of the active substance of an original biological medicinal product and are very close to the reference product in terms of quality characteristics, biological activity, safety, and efficacy based on comprehensive comparability studies done before registration of the product as well as phase 4 effectiveness studies. Rheumatoid arthitis Juvenile idiopathic arthritis Ankylosing Spondylitis Beҫhets Disease uveitis Vasculitic disease Inammatory Bowel Diseases - Crohns disease - Ulcerative colitis Psoriatic arthritis Psoriasis TNFα Inhibitors Figure 1. Spectrum of chronic inammatory diseases in which TNFα inhibitors are used Figure 2. Schematic representation of the structure of a few common TNFα inhibitors ®

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

TNFα Inhibitors Therapeutic Drug MonitoringCompiled by Prof E Vardas
NEWSLETTER
3 Quarter 2017rd
IntroductionTumour necrosis factor alpha (TNFα) is a pro-inflammatory cytokine involved in the pathogenesis of several chronic
inflammatory conditions. The introduction of monoclonal antibody derived �biologic� drugs targeting TNFα has
redefined the treatment of multiple chronic inflammatory diseases (see Figure 1) and has led to considerable
improvement in clinical outcomes for patients with these conditions.
TNFα inhibitors (TNFi)These biologic drugs are monoclonal antibodies of various designs (see Figure 2). Infliximab (Revellex ) was the first
TNFα inhibitor to be approved for the treatment of inflammatory bowel disorders. Infliximab is a chimeric monoclonal
IgG1 antibody composed of a murine variable region and a constant human region. Alternative tradenames for many of
these monoclonal antibody drugs exist because �biosimilars� produced by other manufacturers, after the patent of the
original drug has expired, have entered the market. Biosimilars contain a similar version of the active substance of an
original biological medicinal product and are very close to the reference product in terms of quality characteristics,
biological activity, safety, and efficacy based on comprehensive comparability studies done before registration of the
product as well as phase 4 effectiveness studies.
Rheumatoidarthitis
Juvenileidiopathicarthritis
Ankylosing Spondylitis
BeҫhetsDiseaseuveitis
Vasculiticdisease
In�ammatory BowelDiseases
- Crohn�s disease- Ulcerative colitis
PsoriaticarthritisPsoriasis
TNFα Inhibitors
Figure 1. Spectrum of chronic in�ammatory diseases in which TNFα inhibitors are used
Figure 2. Schematic representation of the structure of a few common TNFα inhibitors
®

TNFi therapeutic drug monitoringThe management of patients receiving TNFα inhibitors has until recently been empirical, with dose adaptations based
on various complex clinical measures and derived scales that have been developed to quantitate patient symptoms,
and which may include both objective and subjective measures of improvement. Recent technological advances have
made therapeutic drug monitoring (TDM) of TNFα inhibitors possible. Consequently treatment with these very costly
biologicals can now be more personalised for the individual patient, and ultimately more cost-effective.
Multiple clinical studies have demonstrated a link between trough concentrations of in�iximab (IFX), the concentration
of the drug just before administration of the next dose, and clinical response in patients with in�ammatory bowel
disease (IBD). Low or undetectable concentrations correlated with poor response and adequate trough levels are
associated with a sustained response to IFX. In rheumatoid arthritis (RA), the use of a biologic disease-modifying anti-
rheumatic drug (DMARD) or the targeted kinase inhibitor tofacitinib is recommended for patients with moderate or high
disease activity despite monotherapy with a conventional (non-biologic) DMARD. The most commonly used biologics in
these patients are the TNFα inhibitors etanercept, adalimumab, and in�iximab. Newer TNFi (e.g. certolizumab pegol and
golimumab) are also used, but less frequently.
For any patient on TNFi biological therapy, treatment failure may occur and it can be either primary or secondary.
Primary failure occurs in approximately 30% of patients who are refractory to, and do not improve clinically while on
induction therapy with TNFα inhibitors. Secondary treatment failure, de�ned as decreased or lack of response in a
previously responsive patient, may also occur. Table 1 outlines the most common reasons for secondary treatment
failure on TNFi, which may occur in up to 50% of IBD/RA patients that responded initially.
The production of anti-drug antibodies (ADA), which neutralise and accelerate the clearance of the particular TNFα
inhibitor, results in reduced drug availability. ADA may occur transiently in some patients, which are of no clinical
consequence. High levels of ADA that in�uence treatment outcomes may also occur. The point prevalence of ADA is
approximately 60% in episodic treatment with TNFi and 6 � 25% in scheduled treatment. Most patients that develop
ADA do so within the �rst 12 months of treatment.
Table 1 also outlines the major adverse events that can occur in patients on biologicals. Especially important are
allergic reactions to the drug, increased risk for the development of active tuberculosis, major bacterial and viral
infections, as well as various malignancies.
Table 1. Common causes of treatment failure and complications of TNFα inhibitors
Causes of treatment failure
Complications
Severity of disease
Duration of disease
Smoking
High BMI
Pharmacokinetics � drug elimination
Drug binding
Anti-drug antibodies
Alternative non-TNFα mediated disease pathways
Concomitant treatment with immunosuppressants
Prior treatment failure with another TNFα inhibitor
Allergic reactions to drugs, e.g. urticaria, dif�culty swallowing or breathing, joint pain, fever or chills, swelling of the face or hands
Serious viral or bacterial infections, incl. tuberculosis, especially in people aged > 65 years
Skin reactions, incl. psoriasis (red scaly patches), rashes, skin lesions, ulcers and hives, swollen face and lips
Worsening of pre-existing cardiac condition
Increased cancer risk, e.g. lymphoma
Liver in�ammation

Multiple studies and cost effectiveness analyses provide guidance on the use of TNFi in patients with IBD. Locally
guidance is provided by the South African Gastroenterology Society (SAGES), which mostly uses the approach of the
TAXIT study in their guidelines. Similarly EULAR/SARAA recommendations exist for the use of TNFα inhibitors in
rheumatological conditions. Figure 3 outlines a symptomatic approach to IFX TDM for in�ammatory bowel diseases.
Lancet Laboratories is using an ELISA based system that detects levels of in�iximab and its biosimilars with a
reportable range of 0.5 � 48 �g/mL, and anti-drug antibodies (ADA) with a reportable range of 2.5 � 1 000 ng/mL. These
particular assays were chosen because of their superior performance compared to other TNFα inhibitor immunoassays
available in the market, in particular their ability to detect both in�iximab and its biosimilars, as well as increased
sensitivity to detect anti-drug antibodies.
Soon to be introduced is an adalimumab (ADM) assay with anti-ADM antibodies as well as more rapid TDM systems for
both in�iximab and adalimumab (Humira ) and its biosimilars.
References1. Aaltonen KJ, et al. Systematic review and meta-analysis of the ef�cacy and safety of existing TNF blocking
agents in the treatment of rheumatoid arthritis. PlosOne 2012; 7(1): e30275.
2. Bendtzen K. Anti-TNFα Biotherapies: perspectives for evidence-based personalized medicine. Immunotherapy
2012; 4(11): 1167 � 1179.
3. Freeman K, et al. Clinical effectiveness and cost-effectiveness of use of therapeutic monitoring of tumour
necrosis factor alpha (TNF-α) inhibitors [LISA-TRACKER® enzyme-linked immunosorbent assay (ELISA) kits,
TNF-α-Blocker ELISA kits and Promonitor® ELISA kits] versus standard care in patients with Crohn's disease:
systematic reviews and economic modelling. Health Technol Assess 2016; 20(83): 1 � 288.
4. Jørgensen KK, et al. Switching from originator in�iximab to biosimilar CT-P13 compared with maintained
treatment with originator in�iximab (NOR-SWITCH): a 52 week, randomised, non-inferiority trial. Lancet 2017;
389(10086): 2304 � 2316.
5. Khanna R, et al. Review article: A clinician's guide for therapeutic drug monitoring of in�iximab in in�ammatory
bowel disease. Aliment Pharmacol Ther 2013; 38(5): 447 � 459.
6. Schmitz EM, et al. Therapeutic drug monitoring of in�iximab: performance evaluation of three commercial
ELISA kits. Clin Chem Lab Med 2016; 54(7): 1211 � 1219.
7. Silva LC, et al. Anti-TNF-α agents in the treatment of immune-mediated in�ammatory diseases: mechanisms of
action and pitfalls. Immunotherapy 2010; 2(6): 817 � 833.
8. Ungar B, et al. The temporal evolution of antidrug antibodies in patients with in�ammatory bowel disease
treated with in�iximab. Gut 2014; 63(8): 1258 � 1264.
9. Van Stappen T, et al. An optimised anti-in�iximab bridging enzyme-linked immunosorbent assay for
harmonization of anti-in�iximab antibody titers in patients with in�ammatory bowel diseases. In�amm Bowel
Dis 2015; 21(9): 2172 � 2177.
10. Vande Casteele, N et al. Trough concentrations of in�iximab guide dosing for patients with in�ammatory bowel
disease. Gastroenterol 2015; 148(7): 1320 � 1329.
11. Watermeyer G, et al. Recommendations on the use of anti-TNFs in adults with in�ammatory bowel disease:
SAGES guidelines. South African Gastroeneterology Review, 2015; 13(3): 21 � 24.
®

Johannesburg
Pretoria
Durban
| 0861 LANCET
| (012) 483 0100
| (031) 308 6500
Polokwane
Rustenburg
Nelspruit
| (015) 294 0400
| (014) 597 8500
| (013) 745 9000
Cape Town
Kimberly
Bloemfontein
| (021) 673 1700
| (053) 836 4460
| (051) 410 7100
(051) 410 4000
Google playANDROID APP ON
www.lancet.co.za@LancetLab App StoreAvailable on the
LancetLaboratories
design done and printed by: ELECTRONIC LABORATORY SERVICES (PTY) LTD 0027 (0) 11 358-0798/99corporate branding/newsletters/south africa/2017/N00054 TNFα Inhibitors Therapeutic Drug Monitoring A3 Duplex170gsm leo Jul2017.cdr | rev000
ITEM CODE: N00054
Figure 3. Symptom-guided approach to TNFα inhibitor and drug antibody monitoring
(Ad
apte
d fr
om
Kh
ann
a R
, et
al. A
limen
t P
har
mar
colT
her
2013
; 38:
44
7 –
459)
Incr
ease
TN
Fid
ose
an
dre
‐eva
luat
e d
rug
leve
ls
TN
Fαin
hib
ito
r (T
NFi
) in
clu
des
infl
ixim
ab p
lus
bio
sim
ilars
TD
M: t
her
apeu
�c
dru
g m
on
ito
rin
g; A
DA
: an
�‐d
rug
an�
bo
die
s;
AZ
A: a
zath
iop
rin
e, M
TX: m
eth
otr
exat
e.
*T
NFi
leve
ls: l
ow
< 3
.0 μ
g/m
L, a
deq
uat
e 3.
1‐7
.0 μ
g/m
L**
AD
A d
etec
�o
n li
mit
s 2.
5 –
1 0
00 n
g/m
L
TD
M n
ot
req
uir
ed
YES
NO
Stab
le s
ymp
tom
s th
rou
gho
ut
infu
sio
n in
terv
al
Res
po
nse
to
infu
sio
ns?
Ini�
al r
esp
on
se b
ut
sym
pto
ms
wo
rsen
ing
pri
or
to n
ext
do
se
Excl
ud
e al
tern
ate
dis
ease
pro
cess
.
Do
TN
Fian
d A
DA
leve
ls
Loo
k fo
r o
ther
re
aso
ns
for
dis
ease
ac
�vi
ty
AD
A le
vels
N
EG
AD
Ale
vels
P
OS*
*
Swit
ch w
ith
in c
lass
an
d e
nsu
re
con
com
itan
t im
mu
no
sup
ress
ion
Ad
equ
ate
TN
File
vels
*Lo
w T
NFi
leve
ls*
Sco
pe
or
imag
ing
dep
end
ing
on
acc
ess
Do
AD
Ale
vels
Co
nsi
der
sw
itch
ing
to o
ut
of
clas
s ag
ent,
ad
din
g A
ZA o
r M
TX
, ch
angi
ng
con
com
itan
t im
mu
no
sup
pre
ssiv
e st
ero
ids,
ad
din
g st
ero
ids
or
surg
ery
No
res
po
nse
to
TN
Fid
rug
infu
sio
ns
End
osc
op
y an
d p
oss
ibly
imag
ing
Ac�
ve d
isea
seIn
ac�
ve d
isea
se
Loo
k fo
r o
ther
re
aso
ns
for
sym
pto
ms
Do
TN
Fitr
ou
gh
leve
ls,
CR
P, a
nd
fa
ecal
cal
pro
tec�
n
Co
nsi
der
sw
itch
ing
to o
ut
of
clas
s ag
ent,
ad
din
g A
ZA o
r M
TX, c
han
gin
g co
nco
mit
ant
imm
un
osu
pp
ress
ive
ster
oid
s,
add
ing
ster
oid
s o
r su
rger
y
Ad
equ
ate
TN
File
vels
* Lo
w T
NFi
leve
ls*
Do
AD
Ale
vels
AD
A le
vels
N
EG
AD
Ale
vels
P
OS*
*
Incr
ease
TN
Fid
ose
an
dre
‐eva
luat
e d
rug
leve
ls
Swit
ch w
ith
in c
lass
an
d e
nsu
re
con
com
itan
t im
mu
no
sup
ress
ion
Related Documents