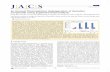
135 Chapter 5 Synthesis of highly functionalized benzo[h]quinoline and tetracyclic dilactam fluorophores 5.1 INTODUCTION In this chapter, we report the synthesis of tricyclic quinoline (tetrahydrobenzo [h] quinoline) (16) and tetracyclic quinoline (3-(epiminomethano) benzo[h]quinoline-2, 12(3H)-dione) (17) fluorophores in an efficient and simple method starting from simple precursor α-tetralone, aryl aldehyde and cyanoacetamide. When the equivalence of the addition of cyanoacetamide to the reaction changed, the product formation also differed (i.e.) when one equivalence of cyanoacetamide was used in this reaction, we got tricyclic monoamide fluorophore (16) but when the reaction was carried out with two equivalence of cyanoacetamide, we got tetracyclic diamide fluorophore (17). This domino reaction is more interesting and fascinating and provides a quick access to the synthesis of highly functionalized tetracyclic quinoline derivatives which give access to tetracyclic dilactams possessing two quaternary amino functionality among four stereogenic centers. Such observation is truly interesting and very rare in organic chemistry. We are the first to report such skeletal arrangement of dilactam in fused tetracyclic ring system using four components via a domino reaction. Initially, we optimized the reaction condition and found the optimal condition for this reaction. Then the reaction was carried out with diverse range of aryl aldehyde, and their reaction mechanism was studied. Several reactions were carried out to investigate the exact mechanism involved in this reaction and the most feasible mechanism for the formation of tetracyclic dilactam was proposed. Figure 5.1 Tricyclo monolactam and multifunctionalized tetracyclic dilactam – A new class of quinoline fluorophore

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript
-
135
Chapter 5
Synthesis of highly functionalized benzo[h]quinoline and
tetracyclic dilactam fluorophores
5.1 INTODUCTION
In this chapter, we report the synthesis of tricyclic quinoline (tetrahydrobenzo [h]
quinoline) (16) and tetracyclic quinoline (3-(epiminomethano) benzo[h]quinoline-2,
12(3H)-dione) (17) fluorophores in an efficient and simple method starting from
simple precursor α-tetralone, aryl aldehyde and cyanoacetamide. When the
equivalence of the addition of cyanoacetamide to the reaction changed, the product
formation also differed (i.e.) when one equivalence of cyanoacetamide was used in
this reaction, we got tricyclic monoamide fluorophore (16) but when the reaction was
carried out with two equivalence of cyanoacetamide, we got tetracyclic diamide
fluorophore (17). This domino reaction is more interesting and fascinating and
provides a quick access to the synthesis of highly functionalized tetracyclic quinoline
derivatives which give access to tetracyclic dilactams possessing two quaternary
amino functionality among four stereogenic centers. Such observation is truly
interesting and very rare in organic chemistry. We are the first to report such skeletal
arrangement of dilactam in fused tetracyclic ring system using four components via a
domino reaction. Initially, we optimized the reaction condition and found the optimal
condition for this reaction. Then the reaction was carried out with diverse range of
aryl aldehyde, and their reaction mechanism was studied. Several reactions were
carried out to investigate the exact mechanism involved in this reaction and the most
feasible mechanism for the formation of tetracyclic dilactam was proposed.
Figure 5.1 Tricyclo monolactam and multifunctionalized tetracyclic dilactam – A
new class of quinoline fluorophore
-
136
5.2 LITERATURE DISCUSSION
Numerous synthetic methods have been reported for the synthesis of benzo [h]
quinoline derivatives and several quinoline derivatives were reported to show
fluorescence properties. Several research groups have established the unique
behaviour of fluorescence property of numerous quinoline derivatives. Following are
the literature found for the synthesis of various fused cyclic lactam derivatives
(quinoline).
5.2.1 SYNTHESIS OF TRICYCLO MONOLACTAM
In 1975, Zahran et. al., reported the synthesis of few benzoquinolines and
benzacridines from 2-Arylidene-3,4-dihydro-l(2H)-naphthalenones and ethyl
cyanoacetate in presence of ammonium acetate (Zahran et al., 1975). Initially they
synthesised 2-benzylidene-3,4-dihydronaphthalen-1(2H)-one starting from α-tetralone
and benzaldehyde, with the obtained aldol product they treated this product with
various reagents like cyanoacetamide, cyanoacetamide with cyclohexanone/ α-
tetralone, ammonium acetate. From these various reactions, they obtained several
benzoquinoline and benzacridine derivatives. 2-oxo-4-phenyl-1, 2, 5, 6-
tetrahydrobenzo [h] quinoline-3-carbonitrile was obtained from the reaction between
2 -benzylidene-3, 4-dihydronaphthalen-1(2H)-one and cyanoacetamide as shown in
Scheme 5.1.
Scheme 5.1 Synthesis of tetrahydrobenzo [h] quinolines
Otto et. al., in 1979, described the synthesis of tetrahydrobenzo [h] quinoline
derivatives starting from 2-arylidene-tetralone and cyanoacetamide under mild
condition (Otto et al., 1979). They synthesised the same derivatives in other way by
reacting 2-arylidene-tetralone with cyanoacetates under the same conditions. The
benzo [h] quinoline-2-ones were formed by cyclisation of 2-arylidene-tetralone with
the carboxamide group of cyanoacetamide; and they did not isolate the intermediate.
-
137
Scheme 5.2 Synthesis of benzo [h] quinolines in different ways.
Rong et. al., in 2009 reported an efficient method for the synthesis of 4-aryl-3-cyano-
1, 2, 5, 6- tetrahydrobenzo[h]quinolin-2-one derivatives under solvent free conditions
(Rong et al., 2009). This group carried out the reaction in one-pot in the presence of
sodium hydroxide as catalyst. Initially they optimized this reaction with various
catalysts and also with various polar solvents. They found an appropriate method for
these reactions to synthesise 4-aryl-3-cyano-1, 2, 5, 6- tetrahydrobenzo[h]quinolin-2-
one. The same reaction conditions were further extended to synthesise 4-aryl-3-
cyano-2,5-dihydro-1H-indeno[ 1,2-b]pyridin-2-one starting from indan-1-one, aryl
aldehyde and cyanoacetamide.
Scheme 5.3 The reaction of α-tetralone, aryl aldehydes, and 2-cyanoacetamide.
5.2.3 SYNTHESIS OF FUSED CYCLIC DILACTAM
In 1964, Paquette et. al., described the use of 1,3-Dihydro-2H-azepin-2-one on
various Diels-Alder Studies and synthesised fused cyclic lactams (Paquette et al.,
1964). 1, 3, 5, 7-tetramethyl and l-ethyl-3, 5, 7-trimethyl-1,3-dihydro-2H-azepin-2-
ones reacted rapidly with tetracyanoethylene thereby the solution immediately turned
into violet-brown colour. This got restored to colourless solution when the reaction
mixture was kept at room temperature for about 30 min. The mixture was allowed to
stand overnight to give rise to dimethyl 2-ethyl-1,4,4,6-tetramethyl-3-oxo-2-
-
138
azabicyclo [3.2.2] non-6-ene-8,9-dicarboxylate and dimethyl 1,2,4,4,6-pentamethyl-3-
oxo-2-azabicyclo[3.2.2]non-6-ene-8,9-dicarboxylate.
Scheme 5.4 Synthesis of fused cyclic lactams via Diels-Alder reaction.
Sato et. al., in 1989, presented the photoaddition of conjugated dienes to 2-Pyridones
and 2-Quinolones (Sato et al., 1989). Irradiation of 2-Pyridones with cyclopentadiene
(10 equivalence) through a Pyrex filter with a 500-W high-pressure mercury lamp
gave intermolecular product 7-azatricyclo [4.2.2.12, 5
] undeca-3, 9-dien-8-one (dimer)
as shown in Scheme 5.5. Similarly they carried out [4+4] cycloaddition on 2-
Pyridones with 1, 3-butadiene followed by Diels-Alder reaction between the adduct
thus formed with 1, 3-butadiene to give 1, 4, 4a, 5, 6, 9, 10, 10a-octahydro-6, 9-
(epiminomethano) benzo [8] annulen-11-one as shown in Scheme 5.6.
ν
Scheme 5.5 Photoaddition of 2-Pyridones with cyclopentadiene.
ν ν
Scheme 5.6 Photoaddition of 2-Pyridones with 1, 3-butadiene.
-
139
Kalme et. al., in 1989 accounted the synthesis of 2, 7-diazabicyclo [2.2.2] octane-3, 8-
diones from 3, 4-dihydropyridin-2(IH)-ones under alkaline medium (Kalme et al.,
1989). They found that the synthesis of diazabicyclo [2.2.2] octane-3, 8-diones was
obtained by intramolecular cyclisation of 6-hydroxy-2-oxo-4, 6-diphenylpiperidine-3-
carboxamide and 3-carbamoyl-4, 6-diphenyl -3,4-dihydropyridin-2(IH)-one in an
alkaline medium as shown in Scheme 5.7. Similarly, they tried to synthesise
diazabicyclo [2.2.2] octane derivatives possessing thiones by replacing carbonyl
group with thio carbonyl group and successfully synthesised diazabicyclo [2.2.2]
octane-3, 8-dithiones.
Scheme 5.7 Synthesis of bicyclic dilactams from tetrahydropyridine.
Guigen Li et. al., in 2009 accounted the synthesis of multifunctionalized quinazoline
derivatives starting from cyclic ketone, cyanoacetamide and aryl aldehydes (Guigen et
al., 2009) as shown in Scheme 5.8. Initially they optimized the reaction condition
with various solvents and bases, and found an optimal condition. They proposed the
most feasible mechanism for this reaction which underwent the tandem formations of
two different Knoevenagel intermediates by [4+2] cycloaddition. This was followed
by intramolecular Michael addition and nucleophilic reaction. They tried the same
reaction with aliphatic aldehyde to synthesise the corresponding quinazoline
derivatives, but this reaction proceeded in another way to form tricyclo [6.2.2.01, 6
]
dodecane. The same group, in 2010, reported the synthesis of highly functionalized
tricyclo [6.2.2.01, 6
] dodecane derivatives via four components domino reaction
providing an access tricyclic monolactam (Guigen et al., 2010) as shown in Scheme
-
140
5.9. They were the first to report the skeleton possessing one quaternary carbon-amino
attachment among four stereogenic centers.
Scheme 5.8 Synthesis of multifunctionalized Quinazoline derivatives.
Scheme 5.9 Synthesis of highly functionalized tricyclo [6.2.2.01, 6
] dodecanes.
5.2.3 FLUORESCENCE STUDIES
Tang et. al., in 2001 reported the aggregation induced emission of 1-methyl-1, 2, 3, 4,
5-pentaphenylsilole which greatly increased the efficiency of silole emission (Tang et
al., 2001). Initially they diluted the 1-methyl-1, 2, 3, 4, 5-pentaphenylsilole with
ethanol solution; excited the sample at 381 nm and found no photoluminescence
signals. At the same concentration they prepared the sample with large amount of
water; excited the sample at 381 nm and found that intense photoluminescence signals
were observed. However, water is non-solvent for pentaphenylsilole. Use of water
system aggregated in the solvent mixture and it was found that the solution was
macroscopically homogenous with no precipitate. This suggests that the silole
aggregates are of nanodimension.
Figure 5.2 Molecular structure and conformational rotamers of silole.
In 2012, Jianbing et. al., reported the aggregation-induced emission enhancement to
dual-channel fluorescence response for conjugated copolymers consisting of
tetraphenylethylene (Jianbing et al., 2012). This group designed a series of new
conjugated polymers which consisted of tetraphenylethylene and fluorene units and
-
141
were synthesized by Suzuki cross coupling polymerization as shown in Scheme 5.10.
When they tried to aggregate tetraphenylethylene in solution, the polymers exhibited
aggregation-induced emission enhancement and dual-channel fluorescence response
as shown in Fig 5.3.
Scheme 5.10 Synthetic routes of tetraphenylethylene and fluorene.
Figure 5.3 Photographs of the polymer fluorescence under UV illumination at
365 nm [RU] = 10 μM in 99:1 water/THF mixture.
In 2013, Ryousuke et. al., described the synthesis of a series of boron ketoiminate
derivatives from 1,3-enaminoketone derivatives and boron trifluoride-diethyl etherate
(Ryousuke et al., 1989). From the result obtained, they suggested that the AIE effect
was derived from rotational or vibrational molecular motions of boron-chelating rings
with a boron-nitrogen bond. This group was the first to state that boron complexes
possessing four-coordination would inherently provide strong effect on the generation
of AIE properties.
Figure 5.4 Photograph of boron complex of 3-amino-1,3-bis(4-methoxyphenyl)
prop-2-en-1-one with solvent compositions of the THF/H2O mixture.
-
142
5.3 RESULTS AND DISCUSSION
For the first time, we report the tetracyclic quinoline (3-(epiminomethano)
benzo[h]quinoline-2, 12(3H)-dione (17) fluorophores skeletal motif which was
synthesised from an efficient method using NaOH as catalyst from simple starting
materials α-tetralone, aryl aldehyde and cyanoacetamide. We tried to synthesize
quinolines starting from α-tetralone, aryl aldehyde and cyanoacetamide using the
same condition which is used in chapter 4. Interestingly, the obtained 2-oxo-4-
phenyl-1, 2, 5, 6-tetrahydrobenzo[h]quinoline-3-carbonitrile possesses fluorescence
properties. To clarify further, we recorded single crystal XRD to evaluate the
interaction of the molecule in crystal lattice, and it clearly showed that the synthesized
molecule exhibited aggregation induced emission which provoked the fluorescence
emission (i.e.) quinoline dimers linked through dimeric N–H•••O hydrogen bonds as
shown in Spectra 5.5. From the obtained quinoline fluorophore derivatives, we
extended the same reaction condition by adding one more equivalence of
cyanoacetamide to synthesize quinazolines as Guigen Li et. al. reported (Guigen et al.,
2009). Guigen Li et. al. reported the synthesis of multifunctionalized quinazolines
starting from cycloketones, aryl aldehyde and cyanoacetamide (Guigen et al., 2010).
But, unexpectedly, we obtained tetracyclic dilactam of highly functionalized
benzo[h]quinoline fluorophores. This synthesized tetracyclic dilactam also showed
fluorescence property due to the formation of aggregation in the crystal packing
(Ryousuke et al., 2013). One more interesting phenomenon for the formation of
aggregation in these tetracyclic dilactam was water hydrogen bonding between the
molecules. When the equivalence of cyanoacetamide used in the reaction was reduced
to 1 eq. instead of 2 eq., the reaction proceeded in different way to give 2-oxo-4-
phenyl-1, 2, 5, 6-tetrahydrobenzo[h]quinoline-3-carbonitrile. General representation
for the synthesis of tricyclo monolactam and tetracyclic dilactam is shown in Scheme
5.11.
Scheme 5.11 Synthesis of tricyclo monolactam and tetracyclic dilactam
-
143
5.3.1 OPTIMISATION OF REACTION CONDITION
Initially, we evaluated the efficiency of the catalyst for this reaction by investigating
the optimal condition under various circumstances. We screened several metal
hydroxides and found an optimal condition for this reaction. From Table 1 (entry 1-9),
it is very clear that the reaction did not proceed in the absence of catalyst and that the
reaction proceeded only in the presence of any metal hydroxide to yield highly
functionalized benzo[h]quinoline.
Table 5.1: Screening of catalyst and solvent effect on four component domino
reaction
Entry Catalyst (mol %) Solvent (ml) Yield (%)
a
1 None Methanol -
2 LiOH (1) Methanol 24
3 LiOH (0.5) Methanol 11
4 KOH (1) Methanol 28
5 KOH (0.5) Methanol 16
6 NaOH (2) Methanol 82
7 NaOH (1) Methanol 82
8 NaOH (0.5) Methanol 82
9 NaOH (0.25) Methanol 71
10 NaOH (0.5) None -
11 NaOH (0.5) CH3CN -
12 NaOH (0.5) DCM -
13 NaOH (0.5) Ethanol 73
14 NaOH (0.5) IPA 64
15 NaOH (0.5) Benzene -
16 NaOH (0.5) Hexane -
aIsolated yield
-
144
Table 5.2: Domino reaction for the synthesis of multifunctionalized benzo[h]
quinoline-2,12(3H)-dione
Entry ArCHO Product Yield (%)b
1 2a
16a
81
2 2b
16b
76
3 2t
16c
72
4 2c
16d
78
5 2g
16e
84
6 2u
16f
85
7 2s
16g
82
8 2e
16h
58
-
145
9 2a
17a
58
10
2v
17b
62
11 2m
17c
69
12 2e
17d
45
13 2g
17e
89
14 2u
17f
65
-
146
15 2s
17g
80
16 2b
17h
83
17 2c
17i
80
18 2f
17j
75
19 2r
17k
89
20 2j
17l
95
aReaction conditions: α-tetralone (10 mmol); Benzaldehyde (10 mmol) and
cyanoacetamide (10/20 mmol) at room temperature (30oC).
bIsolated yield
-
147
When sodium hydroxide was used as catalyst in this reaction, it was significant to
note that we got remarkable yield. So, in order to investigate further on the molar
percentage of the catalyst required to give excellent yield, the reaction was carried out
with 0.1 mol%, which was increased up to 1 mol% of catalyst. A high yield was
observed on using 0.5 mol% of NaOH as catalyst, whereas the use of increased
quantities of catalyst did not further improve the yield significantly. We carried out all
the reactions with 0.5 mol%. The solvent used in the reaction was the next factor
considered for obtaining better yield. So, we carried out these experiments in various
solvents like polar protic, aprotic and nonpolar solvents under same condition (Table
1, entry 8, 10–16). We got better yields with polar protic solvents such as ethanol,
methanol and isopropyl alcohol (IPA) but we did not get the desired product in the
absence of solvent and with polar aprotic solvents like acetonitrile, dichloromethane
(DCM) and nonpolar solvents like hexane, benzene. Thus, the optimal condition for
these reaction transformations was 0.5 mol% of NaOH with methanol as solvent
medium. With the obtained optimal conditions, we carried out these four components
domino reaction with a diverse range of aryl aldehydes under the same conditions to
investigate the generality of the reaction in other system. The results obtained are
summarized in Table 5.2. The synthesised multifunctionalized benzo[h]quinolines
possess two quaternary amine functionalities among four stereogenic centers; such
observation is certainly rare, fascinating and quite interesting in organic chemistry.
We also examined the scope and the limitation of these four component domino
reactions. When the reaction was carried out with other cyclic ketone as substrate
instead of α-tetralone, the reaction proceeded in another path. This is being studied by
other researchers in our laboratories. We also tried these reactions with aliphatic
aldehydes instead of aryl aldehydes under the same condition but failed to get the
desired product.
5.3.2 MECHANISTIC INSIGHT
5.3.3.1 TRICYCLIC MONOLACTAM
There are two possible ways for the formation of tricyclic monolactam (16). One
possible way is the formation of aldol product between α-tetralone and benzaldehyde,
which in turn undergoes cyclisation with cyanoacetamide to yield the expected
product as shown in Scheme 5.12. Another possible way is the formation of
-
148
Knoevenagel product (14a) followed by the cyclisation of 14a with α-tetralone to
yield the desired product as shown in Scheme 5.13.
Scheme 5.12 One possible way for the formation of compound 16.
Thus, to investigate the exact mechanism, we carried out two reactions by changing
the sequence of addition of precursor to the reaction under the same conditions. In one
reaction we took α-tetralone and benzaldehyde initially followed by the addition of
cyanoacetamide to this reaction mixture but the reaction did not proceed to yield the
expected product. In another reaction we took cyanoacetamide and benzaldehyde
initially followed by the addition of α-tetralone to this reaction mixture and monitored
the reaction. In this sequential addition, we got the product 16a.
Scheme 5.13 Another possible ways for the formation of compound 16.
Scheme 5.14 A proposed mechanism for the formation of quinolines (16).
Here, the synthesised Knoevenagel product (14a) under this condition showed
fluorescence property. So, to further confirm the reaction pathway, we synthesised
non- fluorescence Knoevenagel product from cyanoacetamide and benzaldehyde
using polar aprotic solvent. With the obtained non-fluorescence Knoevenagel product,
we carried out the same reaction with 15 but we failed to get the desired product. So,
the synthesized tricyclic monolactam got fluorescence property only via
phenylacrylamide fluorophore. Thus, from these two reactions, we found that the
-
149
mechanism for the formation of quinolines proceeded only via phenylacrylamide
fluorophore followed by cyclisation of 15 as shown in Scheme 5.14.
5.3.3.1 TETRACYCLIC DILACTAM
In this reaction also two ways are possible for the formation of tetracyclic dilactam
(16). One possible mechanism is that aldol product would be formed in the first step;
malonamide would be formed by condensation of cyanoacetamide in the second step
followed by the cyclisation of these two intermediates in the third step, which would
give the desired product as shown in Scheme 5.15.
Scheme 5.15 One possible way for the formation of 17a via int5a and int5c.
Another possible mechanism is that two different Knoevenagel condensations with
cyanoacetamide using α-tetralone and benzaldehyde give two intermediates, which in
turn rearranges C-C bond followed by [4+2] cycloaddition and intramolecular
Michael-type addition. Thus, to examine the exact mechanism, we synthesised all the
intermediates formed in these two possible ways under the same conditions. With
these intermediates, we carried out two reactions by mixing the appropriate
intermediate to obtain the desired product as shown in Scheme 5.16.
Scheme 5.16. Another possible way for the formation of 17a via int5d and 14a.
-
150
One reaction was between int5b and int5c under the same condition, but we failed to
get the product in this way as shown in Scheme 5.15. Another reaction was between
int5d and 14a under the same condition and it was found that the reaction proceeded
smoothly in this way as shown in Scheme 5.16. To further look through the
fluorescence behaviour in these derivatives, we tried the same reaction with non-
fluorescence Knoevenagel product under the same condition, but we couldn’t get the
product. Thus, from these reactions it is clear that the fluorescence properties of
phenylacrylamide were carried over through these reactions to the synthesised
tetracyclic dilactam fluorophore. It also clearly shows that the reaction proceeded only
via phenylacrylamide fluorophore followed by [4+2] cycloaddition and intramolecular
Michael-type addition as shown in Scheme 5.17.
Scheme 5.17. A proposed mechanism for the formation of tetracyclic dilactams.
5.3.3 PHOTOPHYSICAL STUDIES
5.3.3.1 TRICYCLIC MONOLACTAM
UV-visible absorption and fluorescence spectral studies for these synthesised
compounds 16a – 16h were carried out to understand the electronic properties to
explore the influence of electron withdrawing or electron donating nature of the
substituent.
-
151
The absorption spectra of 16 measured in acetonitrile solvent showed peaks at 227-
229 nm. It showed slightly bathochromic-shifted absorption spectrum with respect to
parent compound (16a), and it even showed slightly bathochromic-shifted emission
with respect to 16a as shown in Spectra 5.1 and Fig.5.5.
280 290 300 310 320 330
0.0
0.2
0.4
0.6
0.8
1.0
1.2
Flu
ore
sc
en
ce
In
ten
sit
y (
a.u
)
Wavelength (nm)
16a
16b
16c
16d
16e
16f
16g
16h
Figure 5.5: Emission spectra for the synthesised compound 16a-16h
5.3.3.2 TETRACYCLIC DILACTAM
UV-visible absorption and fluorescence spectral studies for these compounds were
carried out to understand the electronic properties, and also to explore the influence of
electron donating or electron withdrawing nature of the substituent on electronic
properties. The absorption spectra of 17a measured in tetrahydrofuran (THF) solvent
showed peaks at 255, 336, 348, 396, and 412 nm, which was a concentration
dependent behaviour. Interestingly, though the chromophoric unit in this case was
restricted to the benzene moiety, 17l significantly showed the red-shifted absorption
spectrum with respect to benzene where the absorption features were generally
observed below 300 nm.
Table 5.3: Photophysical data of synthesized compound 17a to 17l
S.No Code Abs.max (nm) Flu.max (nm)a Stokes shift (cm
-1 )
1. 17a 393 456 3515
-
152
2. 17b 383 446 3688
3. 17c 391 458 3741
4. 17k 394 460 3642
5. 17d 393 456 3515
6. 17e 395 458 3482
7. 17f 395 458 3482
8. 17g 394 458 3547
9. 17h 392 455 3532
10. 17i 393 456 3515
11. 17j 394 458 3547
12. 17l 393 456 3515 aMolecules are excited at respective absorption maximum.
At lower concentration levels i.e. 10-4
M, only monomer absorption was noticed,
when the concentration was increased further, i.e. greater than critical aggregation
concentration (CAC) the benzo[h]quinoline-2, 12(3H)-dione started aggregation and
showed new absorption bands at lower wavelength regions. The intermolecular
hydrogen bonding interaction through cyclic amide bonds induced the aggregation
process. It has been known, particularly, for benzene based systems that the dimer or
higher aggregates absorbed at longer wavelengths than that of monomers.
Nevertheless, trans-annular interactions between the benzene ring in cyclophane
analogous systems led to longer wavelength absorption peak which was observed
below 300 nm. However, 17a shows the longest absorption spectrum close to 400 nm.
To the best of our knowledge, this is the first observation with benzene aggregates
showing lowest energy absorption in the visible region. The fluorescence spectra of
17a in THF solvent showed maximum emission wavelength at 426 and 448 nm with
mirror image relationship with the lowest energy absorption spectrum. The compound
17l, naphtho[h]quinoline-2, 12(3H)-dione also showed concentration dependent
absorption.
-
153
300 400 500 6000.0
0.2
0.4
0.6
0.8
1.0
1.2
Ab
so
rba
nc
e(a
.u)
Wavelength(nm)
17a-1.603X10-3 M
17e-1.6875X10-3 M
17c-1X10-5 M
17k-1.3267X10-3 M
17d-1X10-5 M
17b-1.0388X10-5 M
17f-1.529X10-3 M
17g-1X10-5 M
17h-1.09X10-3 M
17i-1X10-5 M
17j-1.164X10-3 M
17l-1.6313X10-3 M
Figure 5.6: Absorbance spectra for the synthesised compound 17a-17l
450 500 550 600 6500.0
0.2
0.4
0.6
0.8
1.0
Flu
ore
sc
en
ce
In
ten
sit
y(a
.u)
Wavelength(nm)
17a-1.603X10-3 M
17e-1.6875X10-3 M
17c-1X10-5 M
17k-1.3267X10-3 M
17d-1X10-5 M
17b-1.0388X10-5 M
17f-1.529X10-3 M
17g-1X10-5 M
17h-1.09X10-3 M
17i-1X10-5 M
17j-1.164X10-3 M
17l-1.6313X10-3 M
Figure 5.7: Emission spectra for the synthesised compound 17a-17l
At higher concentration the ground state dimer absorption was predominant at 265,
336, 346, 378, 394 and 414 nm and at lower concentrations monomer spectra were
noticed. The observed spectra matched with the reported value for the dimer
absorption where naphthalene moieties were tethered covalently to the dendrimer
moiety. Indeed, intense fluorescences at 438 and 459 nm were observed for
naphthalene dimers in THF solutions and that of monomer was observed at 328, 340
nm along with the shoulder at 354 nm. The red-shifted absorption and fluorescence
spectra at higher concentration was solely due to the self-assembly formation which
was aided by the hydrogen bonding interaction.
-
154
300 400 500 6000.0
0.2
0.4
0.6
0.8
1.0
Ab
so
rba
nc
e(a
.u)
Wavelength(nm)
100% THF
80% THF & 20% WATER
60% THF & 40% WATER
40% THF & 60% WATER
30% THF & 70% WATER
20% THF & 80% WATER
10% THF & 90% WATER
Figure 5.8 Absorbance Spectrum of Compound 17l in various percentages of
water and THF at 1.356 X 10-2
M
300 400 5000.0
0.2
0.4
0.6
0.8
1.0
Flu
ore
sc
en
ce
In
ten
sit
y(a
.u)
Wavelength(nm)
100% THF
80% THF & 20% WATER
60% THF & 40% WATER
40% THF & 60% WATER
30% THF & 70% WATER
20% THF & 80% WATER
10% THF & 90% WATER
Figure 5.9 Fluorescence Spectrum of Compound 17l exi-270nm in various
percentages of water and THF at 1.356 X 10-2
M
’
Figure 5.10 Fluorescent photographs of compound 17l in THF/water mixtures
with different water fractions taken under UV illumination
-
155
In order to enable the self-assembly even at lower concentrations, we used the
reprecipitation method where the concentrated 17l was injected rapidly into the water,
water-THF solvent mixtures in different ratios (V/V). The concentration of 17l was
maintained at 2.712×10-4
M, where only the monomer absorption was observed.
Addition of 17l to the water-THF mixture caused slight changes in the spectral
maxima; however, a significant reduction in absorbance characteristic to the solvent
ratio has been noticed. Thus, we can say that the formed self-assembly is quite stable
as understood from the lack of time-dependent absorption spectral changes. It should
be noted that solvent aided self-assembled structure formed in water-THF solvent
mixture at lower concentrations does not form the naphthalene dimers. Nonetheless,
the fluorescence from the monomeric units becomes intense when the water-THF
ratio is 20-80, 40-60, 60-40%. (SI, Figure,). The dimer also exists in the solid state as
can be understood from the UV-visible diffuse reflectance spectra of the solid
samples.
-
156
5.4 SPECTRAL DISCUSSION
Characterization of compound 16h and 17b was discussed as a representative
compound of this chapter 16a – 16h and 17a – 17l respectively.
Figure 5.11: Structure of 4-(4-methoxyphenyl)-2-oxo-1, 2, 5, 6-tetrahydrobenzo [h]
quinoline-3-carbonitrile (16h) and 4-(2-(trifluoromethyl) phenyl)-4, 4a, 5, 6-tetrahydro-
1H-10b, 3-(epiminomethano) benzo[h]quinoline-2, 12(3H)-dione (17b)
4-(4-methoxyphenyl)-2-oxo-1, 2, 5, 6-tetrahydrobenzo [h] quinoline-3-carbonitrile
(16h): Green crystal was obtained by slow evaporation from ethanol and THF (1:1)
mixture. Yield and melting point are 58 and > 300 ⁰C respectively. FT-IR spectra of
compound 16h, showed the absorption band at 3352 cm-1
representing the secondary
amide N-H stretching. A band at 3035 cm-1
indicates the aromatic C-H stretching. The
absorption band at 2810 cm-1
represents the aliphatic C-H stretching. A band at 2358
cm-1
confirms the presences of CN stretching and a band at 1678 cm-1
confirms the
presence of C=O stretching (Spectra 5.2). 1H and
13C-NMR spectra have been
recorded in 400 MHz Bruker using CDCl3 as solvent. 1H-NMR spectra of compound
16h shows doublet of doublet peak at 1.77-1.85 ppm, corresponding to one aliphatic
CH2 proton and peak at 2.08-2.17 ppm shows doublet of doublet, corresponding to
one aliphatic CH2 proton. Two doublet of doublet peaks showed at 2.82-3.00 ppm
corresponding to two proton of aliphatic CH2. A singlet peak at 3.78 ppm showed the
presence of methoxy (OCH3) proton. The other aromatic CH proton appeared at 6.85-
7.52 ppm and singlet peaks at 7.00 ppm corresponding to one amide NH proton
(Spectra 5.3). 13
C-NMR spectra of compound 16h showed peaks at 23.13, 29.81,
53.04, 56.02, 64.16 ppm corresponding to aliphatic CH2 carbon. The peak at 110.83-
156.37 ppm corresponded to aromatic carbon and peak at 169.91 ppm indicated amide
-
157
carbonyl carbon (Spectra 5.4). Additional information of 16h structure was obtained
from single crystal X-ray diffraction analysis (Spectra 5.5).
4-(2-(trifluoromethyl) phenyl)-4, 4a, 5, 6-tetrahydro-1H-10b, 3-(epiminomethano)
benzo[h]quinoline-2, 12(3H)-dione (17b): Light green crystal was obtained by slow
evaporation from ethanol and THF (1:1) mixture. Yield and melting points were 62
and > 300 ⁰C respectively. FT-IR spectra of compound 17b, showed the absorption
band at 3510 cm-1
representing the secondary amide N-H stretching. A band at 3195
cm-1
indicated the aromatic C-H stretching. The absorption band at 2939 cm-1
represented the aliphatic C-H stretching. A band at 1708 cm-1
confirmed the presence
of C=O stretching (Spectra 5.6). 1H-NMR spectra of compound 17b showed doublet
of doublet peak at 1.46-1.49 ppm, corresponding to one aliphatic CH2 proton and peak
at 1.55-1.58 ppm showed doublet of doublet, corresponding to one aliphatic CH2
proton. Triplet Peak at 2.41-2.43 ppm showed one aliphatic CH proton. Peak at 2.75-
2.78 ppm showed multiplet, corresponding to one aliphatic CH proton (1H) and one
aliphatic CH2 proton (2H). Peak at 3.38-3.41 showed doublet, corresponding to one
aliphatic CH proton (1H). The other aromatic CH proton appeared at 6.60-7. 52 ppm
and singlet peak at 7.63 ppm corresponding to two amide NH proton (Spectra 5.7).
13C-NMR spectra of compound 17b showed peak at 30.42, 46.44, 57.33, 60.21 ppm
corresponding to aliphatic CH2 and CH carbon. Peak at 114.87-138.72 ppm
corresponded to aromatic carbon; peak at 162.22 and 163.16 ppm indicate two amide
carbonyl carbons (Spectra 5.8). Additional information of 17b structure was obtained
from single crystal X-ray diffraction analysis (Spectra 5.9). The structures of all the
synthesised compounds (16a – 16h) were confirmed using FT-IR, 1H,
13C-NMR,
HRMS analysis (Table 5.4) and few of the crystalline product were confirmed using
single crystal XRD analysis.
-
15
8
20
02
50
30
03
50
-0.1
0.0
0.1
0.2
0.3
0.4
0.5
0.6
0.7
0.8
Absorbance (a.u)
Wa
ve
len
gth
(nm
)
16
a
16
b
16
c
16
d
16
e
16
f
16
g
16
h
Sp
ectra 5
.1: A
bso
rban
ce spectra
for th
e syn
thesised
com
pou
nd
16a
-16h
-
15
9
Sp
ectra 5
.2: F
T-IR
of 4
-(4-m
ethoxyp
hen
yl)-2
-oxo-1
, 2, 5
, 6-tetra
hyd
rob
enzo
[h] q
uin
olin
e-3
-carb
on
itrile (16
h)
-
16
0
Sp
ectra 5
.3: 1H
– N
MR
of 4
-(4-m
ethoxyp
hen
yl)-2
-oxo-1
, 2, 5
, 6-tetra
hyd
rob
enzo
[h] q
uin
olin
e-3
-carb
on
itrile (16
h)
-
16
1
Sp
ectra 5
.4: 1
3C –
NM
R o
f 4-(4
-meth
oxyp
hen
yl)-2
-oxo-1
, 2, 5
, 6-tetra
hyd
rob
enzo
[h] q
uin
olin
e-3
-carb
on
itrile (16
h)
-
16
2
Sp
ectra 5
.5: O
RT
EP
dia
gra
m o
f 4-(4
-meth
oxy
ph
enyl)-2
-oxo-1
, 2, 5
, 6-tetra
hyd
rob
enzo
[h] q
uin
olin
e-3
-carb
on
itrile (16h
)
-
16
3
Sp
ectra 5
.6: F
T-IR
of 4
-(2-(triflu
oro
meth
yl) p
hen
yl)-4
, 4a, 5
, 6-tetra
hy
dro
-1H
-10b
, 3-(ep
imin
om
ethan
o) b
enzo
[h]q
uin
olin
e-2
,
12(3
H)-d
ion
e (17b
)
-
16
4
Sp
ectra 5
.7: 1H
– N
MR
of 4
-(2-(triflu
oro
meth
yl) p
hen
yl)-4
, 4a, 5
, 6-tetr
ah
yd
ro-1
H-1
0b
, 3-(ep
imin
om
eth
an
o) b
enzo
[h]q
uin
olin
e-2
,
12(3
H)-d
ion
e (17b
)
-
16
5
Sp
ectra 5
.8: 1
3C –
NM
R o
f 4-(2
-(trifluoro
meth
yl) p
hen
yl)-4
, 4a, 5
, 6-tetra
hyd
ro-1
H-1
0b
, 3-(ep
imin
om
eth
an
o) b
enzo
[h]q
uin
olin
e-
2, 1
2(3
H)-d
ion
e (17b
)
-
16
6
Sp
ectra 5
.9: O
RT
EP
dia
gra
m o
f 4-(2
-(trifluoro
meth
yl) p
hen
yl)-4
, 4a, 5
, 6-tetra
hyd
ro-1
H-1
0b
, 3-(ep
imin
om
ethan
o)
ben
zo[h
]qu
inolin
e-2, 1
2(3
H)-d
ion
e (17b
)
-
16
7
Tab
le 5.4
Sp
ectral D
ata
of co
mp
ou
nd
16a
-16h
an
d 1
7a
-17l
Pro
du
ct
cod
e
FT
-IR
(cm-1)
Rf -
TL
C
(EA
:
hex
ane)
1H-N
MR
(δ) in
pp
m
13C
-NM
R (δ
) in p
pm
HR
MS
MP
(⁰C)
Ca
lcula
ted
Fo
un
d
16
a
33
52
,
30
35
,
28
10
,
23
58
,
16
78
0.8
4
(55
%)
1.9
8-2
.05
(dd
, CH
2 , 2H
), 2.8
2-3
.00
(dd
, CH
2 , 2H
),
7.1
6-7
.61
(ArH
, 9H
), 7.8
3 (s, C
ON
H2 , 2
H)
24
.73,
25.6
4,
54
.83
, 5
5.6
4,
115
.73,
12
0.9
1, 1
26
.84
, 128
.52, 1
29.6
1, 1
30
.85,
13
3.5
1, 1
34
.85
, 14
3.4
1, 1
52.1
4, 1
70
.02
C2
0 H1
4 N2 O
([M]
+)
29
8.1
10
6
29
8.1
10
8
>3
00
16
b
33
73
,
30
93
,
28
73
,
23
64
,
16
92
0.4
5
(55
%)
1.8
3-1
.89
(dd
, CH
2 , 2H
), 2.3
8 (s, C
H3 , 3
H), 2
.95
-
3.0
1 (d
d, C
H2 , 2
H), 6
.97
-7.6
1 (A
rH, 8
H), 7
.86 (s,
CO
NH
2 , 2H
)
19
.41,
25
.82
, 2
8.7
3,
55
.84
, 5
6.7
2,
11
2.7
4, 1
22
.49
, 126
.40, 1
28.8
4, 1
29
.51,
13
1.6
2, 1
33
.81
, 134
.75, 1
41.4
2, 1
45
.14,
16
8.8
4
C2
1 H1
6 N2 O
([M]
+)
31
2.1
26
3
31
2.1
26
3
>3
00
16
c
33
84
,
30
31
,
28
01
,
23
74
,
16
91
0.7
4
(55
%)
1.7
9-1
.85
(dd
, CH
2 , 2H
), 2.2
5 (s, C
H3 , 3
H), 2
.99
-
3.0
6 (d
d, C
H2 , 2
H), 6
.85
-7.5
2 (A
rH, 8
H), 7
.82 (s,
CO
NH
2 , 2H
)
20
.12,
24
.73
, 2
6.9
1,
52
.63
, 5
4.3
4,
11
6.3
4, 1
19
.59
, 122
.54, 1
27.5
4, 1
29
.35,
13
0.6
8, 1
32
.69
, 134
.74, 1
42.7
4, 1
52
.64,
16
7.7
2
C2
1 H1
6 N2 O
([M]
+)
31
2.1
26
3
31
2.1
26
2
>3
00
16
d
34
72
,
31
74
,
28
94
,
23
62
,
16
83
0.7
2
(55
%)
1.9
7-2
.04
(dd
, CH
2 , 2H
), 2.4
7 (s, C
H3 , 3
H), 2
.89
-
2.9
5 (d
d, C
H2 , 2
H), 6
.78
-7.4
9 (A
rH, 8
H), 7
.64 (s,
CO
NH
2 , 2H
)
19
.73,
25
.74
, 2
6.8
2,
51
.43
, 5
2.6
4,
11
8.6
4, 1
22
.86
, 126
.64, 1
28.5
2, 1
29
.18,
13
0.7
6, 1
34
.65
, 136
.15, 1
43.3
8, 1
51
.27,
17
1.5
5
C2
1 H1
6 N2 O
([M]
+)
31
2.1
26
3
31
2.1
26
0
>3
00
-
16
8
16
e
33
17
,
30
73
,
28
94
,
23
28
,
16
46
0.8
4
(55
%)
2.0
3-2
.09
(dd
, CH
2 , 2H
), 2.9
3-3
.05
(dd
, CH
2 , 2H
),
6.9
4-7
.59
(ArH
, 8H
), 7.7
8 (s, C
ON
H2 , 2
H)
27
.65,
28.7
4,
52
.17
, 5
3.0
4,
121
.64,
12
3.6
4, 1
26
.63
, 127
.76, 1
28.3
8, 1
30
.15,
13
1.6
4, 1
32
.41
, 14
0.8
4, 1
47.7
1, 1
68
.83
C2
0 H1
3 FN
2 O
([M] +)
31
6.1
01
2
31
6.1
01
4
>3
00
16
f
32
91
,
30
03
,
28
63
,
23
84
,
16
91
0.6
1
(55
%)
1.8
8-1
.97
(dd
, CH
2 , 2H
), 2.7
6-2
.84
(dd
, CH
2 , 2H
),
6.9
1-7
.50
(ArH
, 8H
), 7.6
3 (s, C
ON
H2 , 2
H)
24
.16,
24.9
3,
55
.74
, 5
3.7
2,
119
.84,
12
0.1
9, 1
25
.39
, 127
.92, 1
29.6
4, 1
30
.83,
13
1.8
5, 1
34
.74
, 14
4.8
1, 1
52.3
7, 1
71
.18
C2
0 H1
3 FN
2 O
([M] +)
31
6.1
01
2
31
6.1
01
1
>3
00
16
g
33
83
,
30
99
,
28
64
,
23
16
,
16
90
0.5
8
(55
%)
1.9
9-2
.07
(dd
, CH
2 , 2H
), 2.9
8-3
.09
(dd
, CH
2 , 2H
),
6.8
1-7
.54
(ArH
, 8H
), 7.8
9 (s, C
ON
H2 , 2
H)
27
.54,
29.3
4,
55
.23
, 5
6.3
4,
112
.53,
11
9.1
2, 1
20
.34
, 124
.59, 1
28.8
4, 1
30
.83,
13
1.9
4, 1
32
.41
, 14
1.8
4, 1
54.8
2, 1
70
.27
C2
0 H1
3 FN
2 O
([M] +)
31
6.1
01
2
31
6.1
01
2
>3
00
16
h
33
95
,
30
71
,
28
75
,
23
42
,
16
12
0.4
0
(55
%)
1.7
7-1
.85
(d, C
H2 , 1
H), 2
.08
-2.1
7 (d
d, C
H2 , 1
H),
2.8
2-3
.00
(d
d,
CH
2 , 2
H),
3.7
8
(s, O
CH
3 , 3
H),
6.8
5-7
.52
(ArH
, 8H
), 7.0
0 (s, C
ON
H2 , 2
H)
23
.13,
29
.81
, 5
3.0
4,
56
.02
, 6
4.1
6,
11
0.8
3, 1
21
.28
, 127
.32, 1
28.2
6, 1
29
.59,
13
0.9
0, 1
33
.90
, 134
.26, 1
45.3
5, 1
56
.37,
16
9.9
1
C2
1 H1
6 N2 O
2
([M] +)
32
8.1
21
2
32
8.1
21
1
>3
00
17
a
35
10
,
31
95
,
29
39
,
17
08
0.8
2
(60
%)
1.7
4-1
.87
(td, C
H2 , 2
H), 2
.44
-2.4
8 (t, C
H, 1
H),
2.9
7-2
.99
(m, C
H, 1
H), 3
.08
-3.1
3 (m
, CH
2 , 2H
),
3.9
7-3
.98
(d, C
H, 1
H), 6
.90
-7. 2
9 (A
rH, 9
H), 8
.72
(s, CO
NH
2 , 2H
)
29
.04,
34
.37
, 4
5.5
2,
50
.12
, 6
0.0
8,
11
8.2
4, 1
28
.35
, 128
.72, 1
29.6
2, 1
30
.89,
13
1.7
6, 1
35
.86
, 16
0.0
5, 1
66.0
6
C2
0 H1
8 N2 O
2
([M] +)
31
8.1
36
8
31
8.1
36
8
>3
00
17
b
35
01
, 0
.79
1
.46
-1.4
9 (d
, CH
2 , 1H
), 1.5
5-1
.58
(dd
, CH
2 , 1H
), 3
0.4
2,
46.4
4,
57
.33
, 6
0.2
1,
114
.87,
C2
1 H1
7 F3 N
2 O2
38
6.1
24
3
>3
00
-
16
9
31
83
,
29
16
,
16
92
(60
%)
2.4
1-2
.43
(t, CH
, 1H
), 2.7
5-2
.78
(m, C
H &
CH
2 ,
3H
), 3.3
8-3
.41
(d, C
H, 1
H), 6
.60
-7. 5
2 (A
rH, 8
H),
7.6
3 (s, C
ON
H2 , 2
H)
11
5.3
7, 1
22
.09
, 124
.79, 1
25.4
2, 1
28
.36,
12
9.1
3, 1
30
.42
, 137
.91
, 13
8.7
2, 1
62
.22,
16
3.1
6
([M] +)
38
6.1
24
2
17
c
35
51
,
31
84
,
29
74
,
17
21
0.6
8
(60
%)
1.7
3-1
.85
(td, C
H2 , 2
H), 2
.58
-2.6
2 (t, C
H, 1
H),
3.0
0-3
.02
(m, C
H2 , 2
H), 3
.36
-3.3
8 (t, C
H, 1
H),
3.9
3-3
.94
(d, C
H, 1
H), 6
.87
-7. 5
6 (A
rH, 8
H), 9
.62
(s, CO
NH
2 , 2H
)
22
.39,
29
.07
, 5
2.3
0,
55
.28
, 6
6.8
3,
11
0.0
9, 1
20
.54
, 126
.58, 1
28.0
4, 1
30
.16,
13
3.5
2, 1
44
.61
, 15
5.6
3, 1
75.9
3
C2
0 H1
7 BrN
2 O2
([M] +)
39
6.0
47
3
39
6.0
47
1
>3
00
17
d
35
84
,
31
41
,
29
82
,
17
12
0.7
1
(60
%)
1.7
1-1
.82
(td, C
H2 , 2
H), 2
.23
-2.2
6 (t, C
H, 1
H),
2.9
8-3
.00
(m, C
H, 1
H), 3
.15
-3.2
0 (t, C
H2 , 2
H),
3.8
1 (s, O
CH
3 , 3H
), 3.8
8-3
.89
(d, C
H, 1
H), 6
.91
-
7. 4
0 (A
rH, 8
H), 8
.53
(s, CO
NH
2 , 2H
)
23
.06,
27
.25
, 5
5.6
4,
56
.33
, 6
9.9
4,
11
1.7
3, 1
16
.15
, 120
.76, 1
25.2
1, 1
27
.08,
12
9.3
4, 1
31
.16
, 13
9.2
9, 1
55.5
4, 1
72
.24
C2
1 H2
0 N2 O
3
([M] +)
34
8.1
47
4
34
8.1
47
3
>3
00
17
e
35
00
,
31
02
,
29
72
,
17
18
0.7
6
(60
%)
1.7
3-1
.89
(td, C
H2 , 2
H), 2
.28
-2.3
0 (t, C
H, 1
H),
2.5
1-2
.54
(m, C
H2 , 2
H), 3
.09
-3.1
1 (t, C
H, 1
H),
3.9
5-3
.96
(d
, C
H,
1H
), 6
.91
-7.
60
(A
rH,
8H
),
10
.25
(s, CO
NH
2 , 2H
)
18
.98,
32.7
4,
58
.63
, 6
0.0
3,
117
.66,
12
5.8
5, 1
27
.10
, 129
.25, 1
30.2
1, 1
34
.24,
14
0.0
0, 1
61
.27
, 16
6.4
8
C2
0 H1
7 FN
2 O2
([M] +)
33
6.1
27
4
33
6.1
27
4
>3
00
17
f
34
72
,
31
36
,
29
84
,
16
92
0.7
8
(60
%)
1.7
2-1
.85
(td, C
H2 , 2
H), 2
.45
-2.4
9 (t, C
H, 1
H),
2.9
7-2
.99
(m, C
H, 1
H), 3
.11
-3.1
5 (t, C
H2 , 2
H),
3.9
8-3
.99
(d, C
H, 1
H), 6
.91
-7. 2
2 (A
rH, 8
H), 9
.23
(s, CO
NH
2 , 2H
)
13
.90,
20
.61
, 3
0.5
0,
57
.72
, 5
9.9
4,
11
4.5
5, 1
16
.03
, 123
.03, 1
29.9
7, 1
30
.33,
13
6.5
7, 1
60
.56
, 16
3.2
2, 1
63.4
8
C2
0 H1
7 FN
2 O2
([M] +)
33
6.1
27
4
33
6.1
27
3
>3
00
17
g
35
18
,
31
81
,
29
25
,
16
71
0.8
1
(60
%)
1.7
4-1
.86
(td, C
H2 , 2
H), 2
.47
-2.5
1 (t, C
H, 1
H),
2.8
8-2
.89
(m, C
H, 1
H), 2
.95
-2.9
7 (t, C
H2 , 2
H),
3.9
8-4
.00
(d, C
H, 1
H), 6
.91
-7. 1
8 (A
rH, 8
H), 8
.83
(s, CO
NH
2 , 2H
)
22
.33,
26
.95
, 4
9.7
1,
54
.72
, 6
2.5
3,
11
3.3
2, 1
22
.21
, 123
.30, 1
25.8
5, 1
28
.98,
13
7.1
7, 1
60
.42
, 16
5.7
0
C2
0 H1
7 FN
2 O2
([M] +)
33
6.1
27
4
33
6.1
27
6
>3
00
17
h
34
86
,
31
72
,
0.7
2
(60
%)
1.6
9-1
.83
(td, C
H2 , 2
H), 2
.29 (s, C
H3 , 3
H), 2
.58
-
2.6
0 (t, C
H, 1
H), 2
.91
-2.9
5 (m
, CH
, 1H
), 3.3
0-
23
.26,
27
.78
, 5
4.6
8,
63
.58
, 6
7.9
3,
11
3.2
3, 1
23
.97
, 126
.64, 1
29.8
3, 1
38
.13,
C2
1 H2
0 N2 O
2
([M] +)
33
2.1
52
4
>3
00
-
17
0
29
79
,
17
09
3.3
2 (m
, CH
2 , 2H
), 3.9
6-3
.97
(d, C
H, 1
H), 6
.88
-7.
30
(ArH
, 8H
), 8.8
2 (s, C
ON
H2 , 2
H)
15
8.5
4, 1
68
.96
3
32
.15
25
17
i
35
51
,
31
62
,
29
84
,
17
08
0.7
5
(60
%)
1.7
1-1
.85
(td, C
H2 , 2
H), 2
.19 (s, C
H3 , 3
H), 2
.34
-
2.3
6 (d
, CH
, 1H
), 2.6
9-2
.71
(t, CH
2 , 2H
), 2.8
9-
2.9
1 (t, C
H, 1
H), 3
.94
-3.9
5 (d
, CH
, 1H
), 6.9
1-7
.
13
(ArH
, 8H
), 8.6
5 (s, C
ON
H2 , 2
H)
20
.96,
35
.52
, 3
8.9
4,
55
.21
, 6
3.2
9,
11
7.8
4, 1
23
.31
, 126
.80, 1
29.3
6, 1
36
.27,
13
7.4
3, 1
63
.57
, 16
6.4
0
C2
1 H2
0 N2 O
2
([M] +)
33
2.1
52
5
33
2.1
52
6
>3
00
17
j
34
77
,
32
01
,
28
91
,
16
82
0.7
6
(60
%)
1.7
6-1
.86
(td, C
H2 , 2
H), 2
.39
-2.4
1 (d
, CH
, 1H
),
2.6
4-2
.68
(m, C
H2 , 2
H), 3
.47-3
.49
(m, C
H, 1
H),
3.9
5-3
.96
(d, C
H, 1
H), 6
.91
-7. 6
6 (A
rH, 8
H), 8
.60
(s, CO
NH
2 , 2H
)
26
.63, 2
9.4
1, 4
1.8
3, 5
0.0
9, 5
8.7
9, 6
9.5
6,
12
6.8
5, 1
28
.09
, 129
.35, 1
30.0
6, 1
33
.76
,
13
7.4
7, 1
72
.12
2
C2
1 H2
0 N2 O
2
([M] +)
33
2.1
52
5
33
2.1
52
5
>3
00
17
k
35
73
,
31
72
,
29
74
,
17
01
0.8
3
(60
%)
1.6
4-1
.78
(td, C
H2 , 2
H), 2
.59
-2.6
1 (d
, CH
, 1H
),
2.8
7-2
.89
(CH
, 1H
), 2.9
9-3
.02
(t, CH
2 , 2H
), 3.8
7-
3.8
8 (d
, CH
, 1H
), 6.9
1-7
. 36
(ArH
, 8H
), 11
.89 (s,
CO
NH
2 , 2H
)
30
.68,
36
.68
, 4
6.2
7,
47
.65
, 5
7.8
9,
11
6.6
1, 1
27
.65
, 129
.12, 1
30.8
9, 1
33
.62,
13
4.4
3, 1
63
.96
, 17
2.0
54
C2
0 H1
7 ClN
2 O2
([M] +)
35
2.0
97
9
35
2.0
97
7
>3
00
17l
35
92
,
31
43
,
29
48
,
17
04
0.8
5
(60
%)
2.0
3-2
.16
(td, C
H2 , 2
H), 2
.93
-2.9
5 (d
, CH
, 1H
),
3.1
3-3
.18
(m, C
H2 , 2
H), 3
.68
-3.7
0 (d
, CH
, 1H
),
4.0
9-4
.10
(d, C
H, 1
H), 6
.91
-8. 0
9 (A
rH, 8
H), 8
.76
(s, CO
NH
2 , 2H
)
22
.19,
32
.13
, 5
0.1
8,
59
.94
, 6
8.6
3,
11
8.3
0, 1
23
.52
, 125
.44, 1
26.3
9, 1
27
.47,
12
8.6
7, 1
31
.15
, 132
.70, 1
33.3
9, 1
59
.28,
16
6.2
9
C2
4 H2
0 N2 O
2
([M] +)
36
8.1
52
5
36
8.1
52
5
>3
00
-
171
5.5 CRYSTALLOGRAPHY DISCUSSION
The diffraction quality crystals for 17b were grown and examined using X-ray
diffraction method. The molecular structure is illustrated in Fig. 5.12. The asymmetric
unit comprised of 2 conformers (A and B) of 17b hydrated with three water
molecules. In the tetracyclic ring, the diazabicyclo-octane-dione (DBOD) and
tetrahydronaphthalene (THP) were fused together. In the reported centrosymmetric
structure, the stereogenic centers of DBOD rings (C10 & C11) in 17b (A) and 17b (B)
assumed R, R configurations. The cyclohexene ring of THP adopts a twisted-chair
configuration and trifluoromethylbenzene substitutent of DBOD ring is in equatorial
position.
Figure 5.12 Molecular structures of 17b, together with the atomic-labeling
scheme. Displacement ellipsoids are drawn at 30% probability levels and
hydrogens are shown as small spheres of arbitrary radii.
In the crystal packing, 17b molecules associate along a-axis to form a channel
occupied by water that form a multiple bridge interactions with one of the amide
groups in DBOD bicyclic ring system. These channels at the unit cell corners (see Fig
5.13 in projected unit cell along a-axis) interacted via closed dimeric N1A-
H1A…O1A interactions between amide groups of DBOD ring and augmented by
Intermolecular C-H…F interactions (Table 5.5).
-
172
Figure 5.13 Crystal packing projected along a-axis displaying nanotube structure
of 17b occupied with water
Table 5.5 Inter and intra-molecular interactions observed in 17b Donor
(D)
H Acceptor
(A)
D-H H…A D…A Angle(D-
H…A)
(Å) (Å) (Å) (°)
N-H…O N1A H1A O1Ai 0.86 2.05 2.886(2) 164
N1B H1B O3Wii 0.86 2.12 2.964(2) 166
N2A H2A O1Biii 0.86 2.03 2.852(2) 161
N2B H2B O2Wiv 0.86 2.05 2.874(3) 159
Ow-H…X O1W H1W1 O2A 0.89(5) 2.10(5) 2.968(3) 167(4)
O1W H1W2 O3Wv 0.94(4) 2.00(4) 2.922(3) 166(3)
O2W H2W1 O1Wvi 0.96(4) 1.91(4) 2.852(3) 167(3)
O2W H2W2 O1Bvii 0.73(6) 2.22(6) 2.927(3) 165(5)
O3W H3W1 O2Aviii 0.78(4) 2.18(4) 2.940(2) 166(3)
O3W H3W2 O2B 0.92(4) 1.89(4) 2.807(2) 175(4)
C-H…O C5A H5A O1Aix 0.93 2.52 3.160(2) 126
C-H….F C4A H4A F1Biii 0.93 2.52 3.354(3) 150
C8A H8A1 F2B 0.97 2.54 3.378(3) 145
C11A H11A F1A 0.98 2.30 3.037(2) 131
C11A H11A F2A 0.98 2.46 3.037(2) 117
C11B H11B F1B 0.98 2.34 2.948(2) 119
C11B H11B F2B 0.98 2.37 3.083(2) 129
C17A H17A F3A 0.93 2.33 2.680(3) 102
C17B H17B F3B 0.93 2.33 2.678(3) 102
Symmetry codes: (i) -x,-y,1-z, (ii) 1-x,2-y,-z, (iii)x,-1+y,z, (iv) x,y,-1+z, (v)1-x,1-y,-z,
(vi) 1-x,1-y,1-z, (vii) 1-x,2-y,1-z, (viii)1+x,1+y,z, (ix) 1+x,y,z.
-
173
5.6 CONCLUSION
We report a synthetic method for the synthesis tetrahydrobenzo [h] quinoline-
3-carbonitrile fluorophore and a novel four-component domino reaction for the
synthesis of highly functionalized benzo[h]quinoline-2, 12(3H)-dione
(tetracyclic dilactams) which gives access to tetracyclic dilactam.
The reaction was easy to perform by simply mixing four common reactants in
the presence of sodium hydroxide as catalyst at room temperature. The
reaction was fast and got completed within 30 minutes. Importantly, it did not
require high energy, and water was the only major by-product, making the
reaction greener and cost effective.
Four stereogenic centers with two quaternary carbon-amino functions were
controlled very well and the stereochemistry was unequivocally determined by
X-ray structural analysis. The resulting product was of significance in organic
and medicinal chemistry research.
At higher concentrations, all the compounds exist as dimers which are aided
by the hydrogen bonding interactions between the amide groups. The dimers
show much red-shifted absorption spectrum than the monomer.
To the best of our knowledge, this report is the first to specify an interesting
finding of tetracyclic dilactam fluorophore which has an inherently strong
effect on the generation of aggregation-induced emission properties.
Indeed, all the compounds show intense blue fluorescence with a maximum at
460 nm. These molecules can be used as blue emitters also the presence of
amide group might help to use these molecules as fluorescent markers for
probing biological functions.
-
174
5.7 EXPERIMENTAL DISCUSSION
Chemicals were purchased from Aldrich and they were used without further
purification. TLC -Thin layer chromatography (Merck, Silica gel 60 F254) was
performed on alumina plates (pre-coated silica gel). FT-IR spectra were recorded in
the range of 4000-400cm-1
on JASCO-4100 spectrometer instrument using KBr
pellets. 1H NMR and
13C NMR spectra recorded using a Bruker AMX 400 FT. HRMS
analysis was obtained from JEOL GC Mate. The X-ray crystallographic diffractions
were determined using a Smart – CCD (Bruker, 2004). The UV-visible absorption
spectra were measured using Shimadzu UV-1800 double beam spectrophotometer.
Fluorescence spectra were measured using Varian-Cary Eclipse fluorescence
spectrophotometer.
5.7.1 GENERAL PROCEDURE FOR THE SYNTHESIS OF TRICYCLIC
QUINOLINE (16a – 16h)
A dry 100ml Erlenmeyer flask was charged with α-tetralone (10 mmol); aromatic
aldehydes (10 mmol); cyanoacetamide (10 mmol); sodium hydroxide (0.5 mol %) and
methanol (15 ml). The reaction mixture was stirred at room temperature for 30-60
min. The reaction was monitored by TLC and after the completion of reaction, the
mixture was neutralised using 0.1 N HCl and extracted into DCM (3 X 20 ml). The
crude reaction mixture was purified by column chromatography on silica gel using
ethyl acetate/hexane as the eluents.
5.7.2 GENERAL PROCEDURE FOR THE SYNTHESIS OF TETRACYCLIC
QUINOLINE (17a – 17l)
A dry 100ml Erlenmeyer flask was charged with α-tetralone (10 mmol); aromatic
aldehydes (10 mmol); cyanoacetamide (20 mmol); sodium hydroxide (0.5 mol %) and
methanol (15 ml). The reaction mixture was stirred at room temperature for 30-60
min. The reaction was monitored by TLC and after the completion of reaction, the
mixture was neutralised using 0.1 N HCl and extracted with DCM (3 X 20 ml). The
crude reaction mixture was purified by column chromatography on silica gel using
ethyl acetate/hexane as the eluents.
-
175
5.7.3 X-RAY CRYSTALLOGRAPHY
Single crystals of data collection quality for 17b were grown from a mixture of
ethanol and THF (1:1) Data were collected on an Oxford Diffraction Xcalibur Eos
Gemini diffractometer (Oxford Diffraction, 2010). The structure was solved by
applying the direct phase-determination technique using SHELXS-97, and refined by
full-matrix least square on F2 using SHLEXL-97 (Sheldrick, 2008). Structural
calculations were performed with WinGX suit of programs (version 1.85.05)
(Farrugia, 1999). Hydrogens were stereochemically fixed and refined with the riding
options. Amide NH and water hydrogens were isotropically refined. The N-H and O-
H distance in the final cycle of refinement was 0.91(3)-0.92(3) Å & 0.81(3)-0.82(3)
Å, respectively. Distances with rest of the hydrogen atoms are: aromatic/sp2 C—H =
0.93 Å, methine C—H = 0.98 Å, and Uiso = 1.2 Ueq(parent). Essential crystal data
are listed in Table 5.6. Crystallographic data for the structures in this paper have been
deposited with the Cambridge Crystallographic Data Centre having accession
numbers, CCDC 956630. Copies of the data can be obtained, free of charge, on
application to CCDC, 12 Union Road, Cambridge CB2 1EZ, UK (fax: +44 (0)1223
336033 or email: [email protected]).
Table 5.6 Crystal data of compound 17b
17b
Crystal Data
Empirical formula 2(C21 H17 F3 N2 O2), 3(H2 O)
Molecular weight 826.78
Morphology Colorless, block
Crystallizing solvent CH3CN : THF (1:1)
Crystal size (mm) 0.20x0.15x0.10
Cell Parameters
a(Å) 10.0111(6)
b(Å) 11.8131(7)
c(Å) 16.4284(9)
(º) 84.685(2)
β(º) 84.790(3)
(º) 83.889(3)
V (Å3) 1923.39(19)
Cell measuring reflection 9329
θ-range (º) 2.38-27.93
-
176
Crystal system Triclinic
Space group P1bar
Z/Z’ 4/2
Dx(cal.) (g/cm3) 1.428
μ (mm-1
) 0.117
Absorption correction multi-scan
F(000) 860
Data Collection
Radiation MoKα
Temperature (0K) 295(2)
θ-range (º) 1.74 – 28.71
Index ranges -12
Related Documents