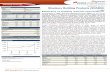
ICICI Securities Ltd. | Retail Equity Research August 13, 2015 Annual Update Steeplechase run continues amid new hurdles Pharma stocks had an eventful FY15 with handsome gains on the back of a re-rating (contrary to our expectation), few M&A deals and scores of compliance issues involving the USFDA. In this report, we have tried to summarise events that influenced stock returns in FY15. We have also highlighted major events to watch in FY16 that may influence the returns for each company. The sector maintained its outperformance over the Nifty as well as broader indices in FY15. After remaining a laggard till the first quarter of FY15, pharma stocks staged a comeback in the remaining fiscal on the back of rejuvenated buying, contrary to our expectations. There are two notable aspects to this shift - 1) Consolidation in the cyclical space after the swift rally on hopes of progressive steps towards ground changing reforms by the newly elected government and 2) consensus beating numbers by most pharma players in H1FY15 on the back of US traction and a recovery in domestic formulations. The earnings momentum was lost in the second half, however, with the slowdown in approvals in the US, increased instances of adverse USFDA scrutiny and currency headwinds in the emerging markets. On the regulatory front, some players from the universe faced increased USFDA scrutiny with Ipca receiving as many as three import alerts. Established players such as Sun, DRL, Lupin and Cadila also received Form 483 observations. The fiscal also witnessed Sun’s acquisition of Ranbaxy in an all-stock transaction and scores of other M&A deals. Notable among them were the Strides-Shasun merger and Aurobindo’s acquisition of US based Natrol. On the financial front (ex-Sun), the I-Direct healthcare universe turnover grew ~15% YoY to | 95469 crore whereas the EBITDA and adjusted PAT grew 9% and 8% to | 20630 crore and | 12111 crore, respectively. Overall EBITDA margins were at 21.6%, declining 130 bps YoY due to higher remedial expenses, incremental R&D spend, currency fluctuation in the emerging markets, high base and absence of high value product launches in the US. Aggregate US sales (ex-Sun) for the select pack of the universe grew ~24% YoY in FY15. With very few approvals and absence of big ticket launches, the growth was mainly driven by the base business. Aggregated domestic formulations (ex-Sun) for the select pack of the universe grew ~15% YoY. As per the secondary sales data released by AIOCD, IPM, as a whole, grew ~13% in FY15 vs. 6.2% growth recorded in FY14. Healthcare Sector Sector View Outperformer Stocks Performance (% returns as on March 31, 2015) Company 1Y 2Y 3Y 5Y FY16 (YTD) Strides Arcolab 254 264 442 846 22 Ajanta Pharma 211 623 1958 5267 64 Natco Pharma 165 396 507 1543 42 Indoco Remedies 161 541 603 631 21 Aurobindo Pharma 140 748 957 563 22 Torrent Pharma. 124 248 288 365 19 Lupin 116 223 286 536 25 Cipla 87 89 137 117 0 Sun Pharma.Inds. 78 152 264 487 4 Cadila Health. 71 139 136 229 11 CNX Pharma 68 116 155 220 9 Apollo Hospitals 50 65 118 288 14 Glenmark Pharma. 40 71 157 197 30 Dr Reddy's Labs 37 100 102 184 6 CNX 500 34 57 65 62 2 Divi's Lab. 32 86 143 180 6 Nifty 27 49 60 62 1 Biocon 12 78 108 79 10 Jubilant Life -2 -6 -18 -50 43 Unichem Labs. -10 25 65 30 -23 Ipca Labs. -25 21 93 145 -7 YTD: till 10 August, 2015 Currency Movement 70 80 90 100 110 120 130 140 Mar-14 May-14 Jul-14 Sep-14 Nov-14 Jan-15 Mar-15 USDINR GBPINR EUROINR INRJPY Quotes have been indexed to 100 with base as March 28, 2014 [ Price movement 100 120 140 160 180 200 220 240 Apr-14 Jun-14 Aug-14 Oct-14 Dec-14 Feb-15 CNX Pharma Nifty Research Analyst Siddhant Khandekar [email protected] Mitesh Shah [email protected] Nandan Kamat [email protected]

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

ICICI Securities Ltd. | Retail Equity Research
August 13, 2015
Annual Update
Steeplechase run continues amid new hurdles Pharma stocks had an eventful FY15 with handsome gains on the back of a re-rating (contrary to our expectation), few M&A deals and scores of compliance issues involving the USFDA. In this report, we have tried to summarise events that influenced stock returns in FY15. We have also highlighted major events to watch in FY16 that may influence the returns for each company. The sector maintained its outperformance over the Nifty as well as broader indices in FY15. After remaining a laggard till the first quarter of FY15, pharma stocks staged a comeback in the remaining fiscal on the back of rejuvenated buying, contrary to our expectations. There are two notable aspects to this shift - 1) Consolidation in the cyclical space after the swift rally on hopes of progressive steps towards ground changing reforms by the newly elected government and 2) consensus beating numbers by most pharma players in H1FY15 on the back of US traction and a recovery in domestic formulations. The earnings momentum was lost in the second half, however, with the slowdown in approvals in the US, increased instances of adverse USFDA scrutiny and currency headwinds in the emerging markets. On the regulatory front, some players from the universe faced increased USFDA scrutiny with Ipca receiving as many as three import alerts. Established players such as Sun, DRL, Lupin and Cadila also received Form 483 observations. The fiscal also witnessed Sun’s acquisition of Ranbaxy in an all-stock transaction and scores of other M&A deals. Notable among them were the Strides-Shasun merger and Aurobindo’s acquisition of US based Natrol. On the financial front (ex-Sun), the I-Direct healthcare universe turnover grew ~15% YoY to | 95469 crore whereas the EBITDA and adjusted PAT grew 9% and 8% to | 20630 crore and | 12111 crore, respectively. Overall EBITDA margins were at 21.6%, declining 130 bps YoY due to higher remedial expenses, incremental R&D spend, currency fluctuation in the emerging markets, high base and absence of high value product launches in the US. Aggregate US sales (ex-Sun) for the select pack of the universe grew ~24% YoY in FY15. With very few approvals and absence of big ticket launches, the growth was mainly driven by the base business. Aggregated domestic formulations (ex-Sun) for the select pack of the universe grew ~15% YoY. As per the secondary sales data released by AIOCD, IPM, as a whole, grew ~13% in FY15 vs. 6.2% growth recorded in FY14.
Healthcare SectorSector View
Outperformer
Stocks Performance (% returns as on March 31, 2015) Company 1Y 2Y 3Y 5Y FY16 (YTD)
Strides Arcolab 254 264 442 846 22
Ajanta Pharma 211 623 1958 5267 64
Natco Pharma 165 396 507 1543 42
Indoco Remedies 161 541 603 631 21
Aurobindo Pharma 140 748 957 563 22
Torrent Pharma. 124 248 288 365 19
Lupin 116 223 286 536 25
Cipla 87 89 137 117 0
Sun Pharma.Inds. 78 152 264 487 4
Cadila Health. 71 139 136 229 11
CNX Pharma 68 116 155 220 9
Apollo Hospitals 50 65 118 288 14
Glenmark Pharma. 40 71 157 197 30
Dr Reddy's Labs 37 100 102 184 6
CNX 500 34 57 65 62 2
Divi's Lab. 32 86 143 180 6
Nifty 27 49 60 62 1
Biocon 12 78 108 79 10
Jubilant Life -2 -6 -18 -50 43
Unichem Labs. -10 25 65 30 -23
Ipca Labs. -25 21 93 145 -7YTD: till 10 August, 2015 Currency Movement
70
80
90
100
110
120
130
140
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
Mar
-15
USDINR GBPINR EUROINR INRJPY
Quotes have been indexed to 100 with base as March 28, 2014 [
Price movement
100120140160180200220240
Apr-1
4
Jun-
14
Aug-
14
Oct-1
4
Dec-
14
Feb-
15
CNX Pharma Nifty
Research Analyst
Siddhant Khandekar [email protected] Mitesh Shah [email protected] Nandan Kamat [email protected]

ICICI Securities Ltd. | Retail Equity Research
Page 2
After re-rating, product approvals, execution key for sustainability
The benchmark CNX Pharma gained 68% in FY15, reflecting a re-rating wave across the sector. Note that in FY09-14, prices were driven mainly by strong earnings momentum. FY15 witnessed adjustment of forward PE multiple from a historical range of ~18-20x to 23x. This, we believe, was a culmination of conviction based on earning consistency, adaptability to the changes, conducive business environment, especially in the US and resilience to structural challenges both in India and abroad. We have seen a marked improvement in balance sheets for most players attributable to an improvement in performance and consistent generation of free cash flows. Hence, higher valuations and higher premium vis-à-vis the benchmark has become a ‘’new normal’’ phenomenon.
In FY16, we have seen some consolidation in the pharma space as after a relentless run-up in FY15, which drove some stocks way ahead of valuations (higher adjusted), prices have started adjusting to their intrinsic values. The CNX Pharma index has corrected ~10% from the peak. Going ahead, the expected slowdown in Q1 and, to some extent, Q2 numbers is likely to keep the rally in check even as the focus shifts to the USFDA approvals pace and the consolidation exercise.
Major areas of concerns, going ahead, include increased and sudden scrutiny of the USFDA that is likely to keep most pharma players on the tenterhooks. Although managements of most companies have sounded confident regarding the cGMP compliance the ball will always be in the USFDA’s court as the agency tries to balance between expedition of approvals to achieve its own target of ~90% clearance of the approvals backlog by September 2017 and maintaining the quality standards adherence. Another developing concern is the sharp depreciation of some currencies in some emerging economies. These markets comprise ~26% of the overall universe turnover and have witnessed 15-40% depreciation vis-à-vis the US$, thus converting into translation losses for most players. In the near future, no respite is expected in the currency issues of these economies.
To conclude, US and Indian formulations remain the main growth drivers for the sector on the back of a strong pipeline and incremental product launches. In general, healthy operating margins, relatively low leverage and strong return ratios are some of the other attributes for most pharma players. Lupin and DRL are our top picks from the large cap pharma space whereas Cadila, Aurobindo, Torrent and Ajanta are our picks from the midcap pharma space.
Exhibit 1: One year forward PE of CNX pharma and Sensex
0
5
10
15
20
25
30
35
Mar
-06
Oct-0
6
May
-07
Dec-
07
Jul-0
8
Feb-
09
Sep-
09
Apr-1
0
Nov
-10
Jun-
11
Jan-
12
Aug-
12
Mar
-13
Oct-1
3
May
-14
Dec-
14
x
CNX Pharma Sensex
81.7% Premium
Source: Bloomberg, ICICIdirect.com Research
Exhibit 2: CNX Pharma forward PE; scaling up
18.0
23.8
20.519.318.8
20.8
15.0
17.0
19.0
21.0
23.0
25.0
FY10 FY11 FY12 FY13 FY14 FY15
Average PE
Source: Bloomberg, ICICIdirect.com Research

ICICI Securities Ltd. | Retail Equity Research
Page 3
Exhibit 3: Domestic performance (primary-reported by companies) ICICIdirect.com coverage companies quarter wise
8
1714
2124
17
29
15 13
39
2014 14 14
19 2018
47
-4
915
1114
713 14 14
40
-5
9
2016 15 17 16 15
5
55
5
-20
-10
0
10
20
30
40
50
60
Cadila Cipla Dr Reddy's Glenmark Indoco Ipca Labs Lupin Sun Pharma Torrent Pharma Unichem Labs
(%)
Q1FY15 Q2FY15 Q3FY15 Q4FY15
Source: Company, ICICIdirect.com Research
Exhibit 4: Domestic performance (secondary market) ICICIdirect.com coverage companies
10 9 10
1516 16
25
15
6
1110
21
1416
2018
14 13 13 14
7
118
15
6
1214
15
20
-3
6
20
17
24
16
2523
16
27
12
-5
0
5
10
15
20
25
30
Cadila Cipla Dr Reddy's Glenmark Indoco Ipca Labs Lupin Sun Pharma Torrent Pharma Unichem Labs
(%)
Q1FY15 Q2FY15 Q3FY15 Q4FY15
Source: AIOCD data base, ICICIdirect.com Research

ICICI Securities Ltd. | Retail Equity Research
Page 4
Exhibit 5: Growth trends in Indian formulations market in FY15 (secondary market)
7.3
11.213.0
9.6 10.4
19.9
6.8
11.413.9
12.7
18.6
20.9
0
5
10
15
20
25
Apr-1
4
May
-14
Jun-
14
Jul-1
4
Aug-
14
Sep-
14
Oct-1
4
Nov
-14
Dec-
14
Jan-
15
Feb-
15
Mar
-15
IPM Growth (%)
Source: AIOCD Data base
Exhibit 6: Growth in key therapeutic segments in FY15
9.414.0
16.7 14.8 15.320.7
13.410.0
5.4 3.56.2
11.5
21.1
6.2 6.0
47.1
28.7
39.8
25.1
05
101520253035404550
Anti-
Infe
ctiv
es
Card
iac
Gast
roIn
test
inal
Vita
min
s
Resp
irato
ry
Anti
Diab
etic
Pain
CNS
Derm
a
Gyna
ecol
ogic
al
Vacc
ines
Horm
ones
Anti-
Neo
plas
tics
Opht
hal
Othe
rs
Bloo
d Re
late
d
Urol
ogy
Anti
Mal
aria
ls
Sex
Stim
ulan
ts
(%)
[
Source: AIOCD Data base
Exhibit 7: Leading brands in Indian formulation market Brand Company API Therapy FY15 FY14 Gr.(%)
Augmentin Glaxosmithkline PAmoxycillin + Clavulanic Acid Anti-Infectives 325.8 259.0 25.8
Becosules Pfizer Ltd Vitamin B Complex Vitamins / Minerals 249.1 227.3 9.6
Corex Pfizer Ltd Chlorpheniramine + Codeine Respiratory 231.2 229.9 0.6
Clavam Alkem Laboratori Amoxycillin + Clavulanic Acid Anti-Infectives 226.3 207.7 9.0
Lantus Sanofi India Ltd. Glargine Anti Diabetic 225.9 171.7 31.5
Volini Ranbaxy LaboratoDiclofenac Pain / Analgesics 211.0 223.8 -5.7
Spasmo ProxyvWockhardt Ltd Dicyclomine + Paracetamol + Tra Gastro Intestinal 190.7 95.6 99.5
Galvus Met Novartis India LtdVildagliptin + Metformin Anti Diabetic 190.5 141.7 34.5
Skinlite Zydus Cadila Hydroquinone + Mometasone + T Derma 185.6 147.1 26.2
Taxim Alkem Laboratori Cefotaxime Anti-Infectives 184.3 151.2 21.8
Manforce Mankind Pharma Sildenafil Sex Stimulants / R 183.6 183.3 0.2
Taxim O Alkem Laboratori Cefixime Anti-Infectives 177.4 156.6 13.3
Pan Alkem Laboratori Pantoprazole Gastro Intestinal 174.1 138.7 25.6
Foracort Cipla Ltd. Formoteral + Budesonide Respiratory 174.1 141.8 22.7
Aciloc Cadila PharmaceuRanitidine Gastro Intestinal 173.0 158.0 9.5
Moxikind Cv Mankind Pharma Amoxycillin + Clavulanic Acid Anti-Infectives 168.9 150.6 12.2[
Source: AIOCD data base, ICICIdirect.com Research
Note: Quotes in all stock Performance Graphs are indexed
to 100 with a base as on 28 March 2013
Revenues EBITDA Profit Apollo Hospitals 20.6 19.6 19.4Aurobindo Pharma 27.7 25.5 22.7Biocon 5.5 8.0 6.5Cadila Healthcare 18.6 16.6 17.9Cipla 15.1 9.8 2.0Divi's Labs 26.6 21.7 20.3Dr Reddy's Labs 16.1 41.4 83.4Glenmark Pharma 21.7 14.6 7.4Indoco Remedies 16.2 23.2 14.0Ipca laboratories 14.5 9.5 4.7Jubilant 8.9 9.8 -6.2Lupin 21.3 29.9 28.7Natco Pharma 12.6 18.1 16.7Torrent Pharma 19.4 19.4 28.1Unichem Laboratories 10.2 -9.1 -11.8
Financial CAGR in FY10-15
Revenues EBITDA Profit Ajanta Pharma 28.9 52.9 62.8Apollo Hospitals 17.9 12.6 14.8Aurobindo Pharma 37.4 60.6 NABiocon 13.8 10.1 5.8Cadila Healthcare 18.2 17.5 20.6Cipla 17.2 9.1 1.5Divi's Labs 18.5 17.9 16.5Dr Reddy's Labs 15.1 13.9 15.7Glenmark Pharma 18.0 13.0 0.2Indoco Remedies 14.6 28.7 20.3Ipca laboratories 9.3 0.4 -2.3Jubilant 10.5 -5.0 2.9Lupin 21.5 22.5 20.0Natco Pharma 16.5 26.1 20.7Torrent Pharma 19.7 26.5 40.7Unichem Laboratories 11.3 -3.5 -2.7
Financial CAGR in FY12-15
Revenues EBITDA Profit Ajanta Pharma 19.0 17.6 17.8Apollo Hospitals 20.5 29.1 34.6Aurobindo Pharma 14.7 18.6 24.9Biocon 9.9 13.8 15.5Cadila Healthcare 20.8 26.2 30.5Cipla 16.8 31.1 40.9Divi's Labs 18.2 94.0 93.3Dr Reddy's Labs 12.7 19.1 21.1Glenmark Pharma 17.8 33.0 64.0Indoco Remedies 26.5 34.5 52.3Ipca laboratories 16.3 24.1 38.1Jubilant 10.5 NA NALupin 17.7 19.1 19.0Natco Pharma 25.0 30.1 46.5Torrent Pharma 20.3 28.8 23.7Unichem Laboratories 16.9 56.7 72.1
Expected CAGR in FY15-17E
Source: Company, icicidirect.com Research

ICICI Securities Ltd. | Retail Equity Research
Page 5
Exhibit 8: Trends in R&D spend
3.2 4.
3
7.5
6.8
4.5 5.
5
5.4
5.4
5.4
6.8
6.1 6.
6
9.4
11.8
11.9
12.0
3.1
4.7
6.5
6.5
9.0 9.
4 10.0
9.5
7.3 8.
3
7.4
7.4 8.
2 8.6
10.0
10.0
6.3
5.2
5.2 6.
0
6.1
7.3 8.
0
6.3
0
2
4
6
8
10
12
14
FY10 FY11 FY12 FY13 FY14 FY15E FY16E FY17E(%
of r
even
ues)
Biocon Dr Reddy's Labs Glenmark Pharma Lupin Sun Pharma
Source: Company, ICICIdirect.com Research
Exhibit 9: US market performance of select companies in FY15 (growth %)
40
56
17
1
16
37
0
10
20
30
40
50
60
Aurobindo Cadila Dr Reddy's Glenmark Lupin Sun Pharma TorrentPharma
(%)
Source: Company, ICICIdirect.com Research

ICICI Securities Ltd. | Retail Equity Research
Page 6
Apollo Hospitals
Profitability of new hospitals key for overall margin improvement
FY15 Highlights • Revenues grew 18% YoY in FY15 driven by 13% growth in
hospital segment and 31% growth in the pharmacy segment • However, higher growth in the low margin pharmacy business
led to a contraction in EBITDA margin by 114 ps YoY • Apollo has acquired Nova Specialty Hospitals, which owns a
chain of 11 facilities across eight cities, and will add to Apollo's network of 45 modular operation theatres. Also, it acquired Hetero Med Solutions, which added 320 pharmacy stores to the company’s pharmacy business from Q1FY16
• In addition to this, it has tied up with Sanofi India to launch 50 Apollo Sugar Clinics
• The company has added only 510 beds in FY15 against planned beds addition of 1000. This was due to a delay in construction work and clearances
• Inpatient average length of stay improved to 4.43 days in FY15 vs. 4.54 days in FY14) while average revenue per occupied bed improved to | 25381 per day vs. | 23684 per day
• Newly commissioned hospitals reported a | 7.5 crore loss at the EBITDA level affecting overall margins. On the other hand, hospitals that were commissioned in FY13 (Vanagaram & Jayanagar) reported EBITDA of | 6.8 crore during the year
• It added 242 stores and closed 52 stores taking total net addition to 190 stores. As on March 31 2014, total stores count was 1822
What to Watch • The company is planning to add 875 beds in FY16 including
among others- North Bangalore 180, Chennai Main 30, Navi Mumbai 350, Indore 65 and Vizag 250
• We expect the company to add ~415 standalone pharmacy stores including addition of 320 stores of Hetero
• We expect robust revenue growth of ~ 24% YoY in FY16E due to volume increase on new hospitals and pharmacy stores addition
• Even on the EBITDA margins front, we expect at least 150 bps improvements as the pressure on margins due to 1) newly commissioned hospitals and 2) higher growth in the low margin pharmacy space is likely to be outweighed by incremental contribution from earlier hospitals and pharmacies
Ajanta Pharma
FMCG attributes in pharma mould justify high valuation
FY15 Highlights • The company has maintained its strong growth trajectory in
FY15. Revenues grew 23% YoY driven by both exports (23% growth) and the domestic formulations (25% growth).
• The growth was driven by 74 new product launches in India and other exports markets including 12 for the first time in India.
• EBITDA margins also improved ~300 bps to 34% on the back of an improved product mix
• The global field force strength has gone up from 450 to 572 in FY15
• R&D spend was ~5% of revenues in FY15 • Total 25 ANDAs have been filed in the US market, out of which
two have been approved. Sales in FY15 accounted for | 4 crore
Stock Performance
80
100
120
140
160
180
200
220
240
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
Mar
-15
CNX Pharma Apollo Hospitals Nifty
Stock Performance [
60160260360460560660760860960
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
Mar
-15
CNX Pharma Ajanta PharmaNifty

ICICI Securities Ltd. | Retail Equity Research
Page 7
What to watch • The company has guided for 32-34% EBITDA margins for FY16
with 16-18% revenue growth. In the domestic market, it expects ~25% YoY growth while in exports APL expects 14-15% growth on a higher base
• The company plans to file at least six ANDAs in FY16 • Capex guidance for FY16 - | 180-200 crore • We expect higher US traction on the back of recent approval for
three respiratory products from the USFDA Aurobindo Pharma
Robust product basket bodes well for future
FY15 Highlights • The company registered yet another strong yearly performance
backed by strong US growth (42% YoY) besides Actavis European acquisition. Revenues increased 50% YoY in FY15
• Despite the loss making Actavis integration, EBIDTA margins were at ~21% mainly on the back of robust US traction
• The company has also forayed into the US nutritional supplement segment by acquiring US based bankrupted company Natrol Inc
• During the year, the company filed 40 ANDAs in the US and received approvals for eight. Cumulatively, Aurobindo has filed 376 ANDAs, of which 166 are final approvals, 27 tentative approvals and is awaiting approvals for 183 ANDAs. Also, 43 injectable filings are under review
• Gross debt stood at US$714 million (~98% forex debt) in FY15 with cash of ~US$75 million
What to watch • The company expects a bottomline break-even at the Actavis
European business in FY16. It also expects to transfer 50% of Actavis products to India in the next 12-18 months
• Actavis sales were US$400 million in FY15 and are expected to grow at mid-single digits
• It expects gross margins to remain at current level of ~54% • We expect initial margin pressure, on account of Actavis and
Natrol, to ease further on the back of incremental high margin US launches
• We expect continued traction in the US, backed by robust product pipeline (183 pending approvals) and niche launches.
Exhibit 10: Notable Para IV Filings Brand Name Indicator Generic Name NDA holder ANDA Filer Date
Abilify Depression aripiprazole Otsuka Pharma Aurobindo 23-May-14
Aloxi nausea and vomiting palonosetron hydrochloride Roche Aurobindo 15-Apr-13
Angiomax blood clots bivalirudin The Medicines Compan Aurobindo 11-Apr-13
Boniva osteoporosis biphosphonic acid Roche Aurobindo 22-Jan-13
Allegra-D seasonal allergies fexofenadine hydrochloride Sanofi-Aventis U.S. LLC Aurolife Pharma LLC 5-Nov-10 [
Source: Bloomberg, ICICIdirect.com Research
Stock Performance
0100200300400500600700800900
1000
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
Mar
-15
CNX Pharma Aurobindo Pharma Nifty

ICICI Securities Ltd. | Retail Equity Research
Page 8
Biocon
Unlocking of Syngene key event to watch for FY16
FY15 Highlights • The company initiated steps for the Syngene listing (which was
eventually concluded recently). In September 2014, Biocon bought back the 7.69% stake held by GE Equity International Mauritius in Syngene at | 2800 crore. In January 2015, the company sold a 10% stake in Syngene to Silver Leaf Oak at a valuation of | 3800 crore
• Its insulin Glargine was approved by COFEPRIS, the Mexican health authority, through its partner PiSA Farmacéutica (Pisa). The combined market for Insulin Glargine in Mexico is estimated to be in excess of US$40 million
• Operationally, the bio-pharmaceutical segment continues to struggle for one reason or the other. Even the branded formulations segment is witnessing a structural slowdown due to portfolio restructuring
• The revenues grew just 7% YoY in FY15 mainly due to portfolio rationalisation in the branded business and capacity constraints/pricing pressure in its bio pharmaceutical business
• Margins also contracted ~147 bps mainly due to increase in R&D
What to watch • The unlocking of Syngene and the management’s commentary
towards utilisation of funds from unlocking • The company expects five biosimilars in Phase III clinical trials by
the end of FY16 • The Malaysian insulin facility is likely to start shipments to
emerging markets from FY17 • The company plans to foray into ~| 330 crore Indian disposable
insulin device segment in FY16
Cadila Healthcare
US growth trajectory key for overall growth
FY15 Highlights • Cadila’s cumulative ANDAs stood at 260, of which, it has
received approvals for 99. In FY15, the company launched nine products, received eight approvals and filed 38 ANDAs.
• Revenue was increased 20% YoY mainly on the back of a steep US growth trajectory driven by aggressive filings and launches in the last few quarters. Margins expanded sharply by ~360 bps in FY15 mainly due to a better product mix
• The company launched a biosimilar drug of Adalimumab in India for the treatment of auto immune disorders. The drug will be marketed under the brand name 'Exemptia' at a cost that will be one-fifth of the innovator product Humira by US based AbbVie. It expects to clock sales of | 100-150 crore in three to five years.
What to watch • The management has given a sales guidance of | 10,000 crore
with EBITDA margin of 21% • It expects to file 40 ANDAs with the USFDA and launch ~20
products in FY16 including niche launches like gAsacol HT and gPrevacid
Stock Performance
80
130
180
230
280
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
Mar
-15
CNX Pharma Biocon Nifty
Stock Performance
80
130
180
230
280
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
Mar
-15
CNX Pharma Cadila Healthcare Nifty

ICICI Securities Ltd. | Retail Equity Research
Page 9
Exhibit 11: Notable Para IV Filings Brand Name Indicator Generic Name NDA holder Filing Date
Exelon dementia rivastigmine Novartis 27-Aug-13
Daliresp Chronic Obstructive PulmRoflumilast AstraZeneca 15-May-15
Nexium gastroesophegal reflux di esomeprazole magnesium AZ 12-Aug-13
Abilify Depression aripiprazole Otsuka Pharma 30-May-08
LIVALO high cholesterol Pitavastatin Calcium Kowa Pharma 17-Apr-14
Optivar seasonal allergic rhinitis azelastine hydrochloride Meda Pharmaceuticals 22-Mar-10
Strattera attention-deficit hyperact atomoxetine hydrochloride Eli Lilly and Co. 27-Oct-10 [
Source: Bloomberg, ICICIdirect.com Research
Cipla
Next 18-24 months crucial for scalability
FY15 Highlights • Cipla was the first company to launch Salmeterol Fluticasone
inhaler (Advair) in the European markets • The company has started supplying Nexium formulations to
Teva, which the latter launched under exclusivity in the US. Cipla has cost plus profit sharing arrangement with Teva for this product
• It has launched generic version of Gilead’s hepatitis C drug Sovaldi (Sofosbuvir) in India under the brand name ‘Hepcvir’. It has also entered into an In-licensing agreement with Gilead for manufacture and distribution of Sofosbuvir, Ledipasvir and GS-5816 in 91 countries for the treatment of hepatitis C
• The company has launched its own products (Amlodipine, Meloxicam, Lamotrigine, Topiramate, Valacyclovir and Doxycycline) in the US
• In its endeavour to de-risk the business model by focusing more on front-end it has built its own front-end presence in 17 new markets including Iran, Sri Lanka, Myanmar, Morocco and Algeria.
• Cipla won the Global Fund ARV tender worth US$189 million to be executed over three years starting from January 1, 2015.
• On the financial front, Cipla struggled in FY15 on account of apparent margin pressure, most of which was self inflicted. The dent on margins was on mainly on account of 1) higher R&D spends, 2) higher global level recruitments, and 3) expenses related to transformation from back-end model to front-end model.
• The company’s ANDAs stood at 147 of which it is awaiting 68 approvals (including tentative approvals). It has launched 50 products in the US.
What to watch • The company expects to launch five to six own label generic
products in the US every year for the next three years • Expect strong cash flows to continue from Nexium supply to
Teva in H1FY16 • Cipla has won first gSerroflo (inhaler) tender in Germany. The
company expects to file all tenders for gSerroflo across EU markets in next 18 months.
• The company has done out-licensing deal with Salix for Rifaximin complexes. It expects milestone payment and profit sharing post launch
• Cipla expects respiratory launches in the US market post FY18
Stock Performance
80
130
180
230
280
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
Mar
-15
CNX Pharma Cipla Nifty

ICICI Securities Ltd. | Retail Equity Research
Page 10
• The company expects to start European filing for paediatrics vaccine from FY16 in a partnership with Serum Institute. It plans to commercialise these vaccines in India in the next couple of months
• It expects R&D spend to be in the range of 6-8% for the next three years and capex at ~8% of sales (6% in FY15). Higher capex will be mainly for capacity addition in FY16 across segments
Exhibit 12: Notable Para IV Filings Brand Name Indicator Generic Name NDA holder ANDA Filer Date
Emtriva HIV emtricitabine Gilead Sciences Cipla 20-Aug-12
Pataday allergic or inflammatory eye co olopatadine hydrocholric solutioAlcon Research Cipla 29-Jan-14
Prezista HIV darunavir Tibotec Inc. Cipla 13-Aug-14
Sustiva HIV rivastigmine tartrate Merck Cipla 27-Jun-13[
Source: Bloomberg, ICICIdirect.com Research
Divi’s Laboratories
Capacity additions to hold key
FY15 Highlights • Carotenoids business witnessed robust growth of 38% in FY15. • The company’s operations were affected for a brief period during
October, 2014 on account of cyclone Hudhud in Vishakhapatnam • During the year, the company has capitalized Fixed Assets
amounted to | 230 crore • Revenue growth of 23% was on account strong traction in the
API business. However, it affected overall margins, which declined ~270 bps due to high growth in low margin API business
What to Watch • Carotenoids business is expected to garner sales of ~| 220 crore • Capex for | 500 crore in the next two years for facility at
Kakinada. The facility will be used for both APIs and CS • DSN capacity utilisation likely to peak by FY16
Dr Reddy’s Laboratories
US, India remain strong; to compensate for slowdown in others
FY15 Highlights • The company received nine USFDA 483 observations for its
Srikakulam API facility in Andhra Pradesh • The company has filed three new drug applications (NDAs) DFD-
01, DFD-09 and DFN-11 with USFDA. These proprietary products are focused on developing therapies in dermatology and neurology
• It acquired Novartis‘ Habitrol brand (an over-the-counter nicotine replacement therapy transdermal patch) in the US
• Till date, the company is awaiting 68 ANDA approvals, which includes 43 Para IV filings comprising 13 products with FTF status. Total ANDA filings stood at 220. Global cumulative DMF filings have reached 735
• The company has initiated the acquisition of select rands of UCB India in India, Nepal, Sri Lanka and Maldives for €118 million (| 800 crore) to strengthen its presence in the dermatology, respiratory and paediatric segments. The transaction includes
Stock Performance
80
130
180
230
280
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
Mar
-15
CNX Pharma Divi's Labs Nifty
Stock Performance
80
130
180
230
280
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
Mar
-15
CNX Pharma Dr Reddy's Nifty

ICICI Securities Ltd. | Retail Equity Research
Page 11
approximately 350 employees engaged in India operations. The revenues of the acquired business are ~| 150 crore
• It launched Valganciclovir tablets (anti-viral), a therapeutic equivalent generic version of Valcyte tablets under exclusivity in the US. Annual sales of this drug were ~US$440 million
• It entered into an agreement with Hetero Drugs under which Dr Reddy's has been licensed to distribute and market Sofosbuvir tablets (gSovaldi) in 400 mg strength in India. The drug, to be sold under the Resof brand, is indicated for the treatment of chronic Hepatitis C
• On the financial front, lack of major US launches and problems in Russia and Venezuela (macroeconomic and currency related) affected FY15 growth (~12%). The EBITDA margins contracted by 160 bps due to higher R&D spend and forex losses
What to Watch • Clearance for Srikakulam API facility from the USFDA will be
keenly watched • Sharp currency depreciation in Russia and Europe had dented
the company’s FY15 growth. We expect the Russia, Europe business to continue to pose challenges for being lumpy & volatile
• We expect US and India to together hold the key for DRL’s overall growth
• The company expects four or five meaningful USFDA approvals in FY16
• The company expects highest ever R&D cost (~12% of sales) in FY16 to support its innovative pipeline and complex ANDA filings

ICICI Securities Ltd. | Retail Equity Research
Page 12
Exhibit 13: Notable Para IV Filings Brand Name Indicator Generic Name NDA holder ANDA Filer Date
Aloxi prevention of acute nausea and palonosetron hydrochloride Roche Dr. Reddy 8-Jul-11
Angiomax anticoagulant for patients underbivalirudin Medicis Pharma Dr. Reddy 28-Apr-11
Boniva osteoporosis ibandronic acid; ibandronate so Hoffmann-La Roche Inc.Dr. Reddy 28-Oct-10
Boniva treatment of osteoporosis] ibandronate sodium Hoffmann-La Roche Inc.Dr. Reddy 18-Jul-08
Copaxone multiple sclerosis glatiramer acetate Teva Pharma Dr. Reddy 11-Sep-14
Dexilant erosive esophagitis, heartburn dexlansoprazole Takeda Pharma Dr. Reddy 31-Mar-11
Diprivan delirium, agitation prpofol Fresenius Kabi Dr. Reddy 25-May-13
Effient acute coronary syndrome prasugrel hydrochloride Eli Lilly Dr. Reddy 12-Mar-14
Folotyn Lymphoma Pralatrexate Allos Therapeutics Dr. Reddy 19-Jun-14
Gleevec leukemia imatinib mesylate Novartis Dr. Reddy 6-Feb-14
Intermezzo insomnia zolpidem tartrate Purdue Pharma Dr. Reddy 2-Apr-13
Ixempra breast cancer epothilone Bristol-Myers Squibb Dr. Reddy 21-Dec-12
Jevtana Kit prostate cancer Cabazitaxel Sanofi-Aventis Dr. Reddy 6-Apr-15
Kuvan Phenylketonuria Sapropterin Dihydrochloride Biomarin Pharma Dr. Reddy 17-Nov-14
Mozobil non-Hodgkin's lymphoma plerixafor Sanofi Dr. Reddy 29-Aug-13
Mucinex bronchial congestion pseudoephedrine hydrochloride Reckitt Benckiser Inc. Dr. Reddy 26-Jun-15
Nexium treatment of acid reflux] esomeprazole sodium AstraZeneca AB Dr. Reddy 17-Jan-08
Pristiq depression desvenlafaxine Pfizer Dr. Reddy 4-Jun-13
Reclast osteoporosis and Paget's Disea zolendronic acid Novartis Pharma Dr. Reddy 20-Feb-13
Treanda chronic lymphocytic leukemia bendamustine Cephalon Inc. Dr. Reddy 20-Dec-13
Uloric febuxostat hyperuricemia Takeda Dr. Reddy 30-Oct-13
Velcade cancer bortezomib Millennium Dr. Reddy 25-Jun-15
Zegerid gastroesophageal reflux diseas omeprazole/sodium bicarbonateSantarus, Inc. Dr. Reddy 15-Aug-12 [
Source: Bloomberg, ICICIdirect.com Research
Glenmark Pharma
Upbeat management guidance based on expected US traction
FY15 Highlights • The company’s US growth was muted on the back of a
slowdown in product approvals while domestic growth remained strong owing to market share gains across therapies
• The RoW business was impacted due to currency devaluation and subdued business environment in Russia
• Revenues grew ~11% YoY. EBITDA margins contracted 337 bps in FY15 due to an adverse product mix, forex losses and higher R&D expenses
• Total USFDA filings as on date are 170. So far, the company has received approvals for 95. From the pending ANDAs, 33 are Para IV applications
• Dahej and Ankaleshwar facilities were successfully inspected by the USFDA in FY15
• The Delhi High Court has restrained Glenmark from manufacturing and marketing anti-diabetes drugs Zita and Zita-Met due to infringement of MSD’s patent. The drug contributes ~| 94 crore (5% of domestic sales and 1.4% of overall sales) to the company.
• Glenmark’s US subsidiary firm will pay US$25 million under a settlement reached with the Texas State’s Attorney General over the pricing investigation case under the Texas Medicaid Fraud Prevention Act
Stock Performance
80
130
180
230
280
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
Mar
-15
CNX Pharma Glenmark Pharma Nifty

ICICI Securities Ltd. | Retail Equity Research
Page 13
• Glenmark has drawn up plans to set up a new manufacturing facility at Monroe Corporate Center, North Carolina, US. This facility will be spread over a 100,000 sq ft plot. Glenmark will first begin work on an oral solid unit and, thereafter, set up manufacturing units for injectables and topicals
• Gross debt and cash stood at ~| 3800 crore and ~| 768 crore respectively in FY15
What to watch
• The management expects revenue to grow at 18-20% in FY16 with EBITDA of ~ | 1750 crore
• It expects US to grow at ~20% on the back of 10-12 products approvals. It also expects sales growth of more than 20% in the European market for the next three years on account of the launches, new approvals and specialty products
• It expects its employee cost to increase 15% in FY16 • GRT 17536 is likely to be out licensed. Its phase 2b will start in
Q3FY16 and would be completed by the end of CY16 • Net debt is expected at ~| 2400 crore for FY16. The company
has given net debt/EBITDA guidance of 1.2-1.5x. Exhibit 14: Notable Para IV Filings Brand Name Indicator Generic Name NDA holder ANDA Filer Date
Alimta Cancer pemetrexed disodium Eli Lilly Glenmark 23-Jan-14
Bystolic Hypertension nebivolol Forest Lab Glenmark 13-Mar-12
Crestor high cholesterol rosuvastatin calcium AstraZeneca Pharmace Glenmark 26-Apr-10
Effient acute coronary syndrome prasugrel hydrochloride Eli Lilly Glenmark 12-Mar-14
Epiduo acne benzoyl peroxide Galderma Lab Glenmark 6-May-15
Finacea rosacea azelaic acid Bayer Glenmark 14-Mar-13
Multaq prevention of cardiovascular hodronedarone hydrochloride Sanofi Glenmark 6-Mar-14
Savella fibromyalgia milnacipran hydrochloride Forest Lab Glenmark 6-Feb-14
Treanda chronic lymphocytic leukemia bendamustine Cephalon Inc. Glenmark 26-Dec-13
Velcade cancer bortezomib Millennium Glenmark 10-Sep-14
Vimpat epilepsy lacosamide UCB Pharma Glenmark 10-Jul-13
Jevtana Kit prostate cancer Cabazitaxel Sanofi-Aventis Glenmark 6-Apr-15
Onglyza type II diabetes Saxagliptin Astrazeneca Glenmark 23-May-14 [
Source: Bloomberg, ICICIdirect.com Research
Indoco Remedies
Domestic growth, Watson deal main catalysts for earnings
FY15 Highlights • The visibility improved substantially after the approvals - EIR for
plant II and first-time approval for plant III. • The growth in the regulated markets was driven by growth in the
US, UK and South Africa. German Metformin tenders also contributed to growth in the regulated markets.
• The company has filed 28 ANDAs with the USFDA and received approval for 11 products. Of the 28 ANDAs, 16 were filed under the Watson deal
• Under the Aspen deal, the company registered | 20 crore of revenues in FY15. A delay in shipments was mainly due to a delay in product approvals in South Africa
Stock Performance
80130180230280330380430480530580630
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
Mar
-15
CNX Pharma Indoco Remedies Nifty

ICICI Securities Ltd. | Retail Equity Research
Page 14
• With the acquisition of contract research organisation (CRO) business from Piramal the company has made its maiden foray in the CRO business
What to watch
• Indoco has guided for 20% revenue growth (excluding Watson revenues) with overall margins in the range of 20-21%. It expects 18-20% growth in the domestic market and18-20% growth in European markets.
• The company expects to file five to six products with the USFDA in FY16 on its own and four products under the Watson deal.
• Regarding Watson execution, we expect the company to generate | 90 crore in FY16.
• It expects capex of | 125 crore in FY16 mainly for capacity addition.
Ipca Laboratories
Remedial measures likely to delay normal functioning
FY15 Highlights • The USFDA has put an import alert on two of Ipca’s facilities in
Pithampur (Madhya Pradesh) and Piparia (Silvassa) as they failed to maintain good manufacturing practices. Ipca’s Ratlam facility has already been under import alert since January
• The Ratlam facility also received observations from WHO Geneva subsequent to which the dispatches for institutional tender business were halted for some time. This adversely affected the tender business in FY15
• The company acquired a manufacturing facility from Alpa Laboratories in Pithampur, Indore, for | 72 crore. Alpa owns a wide range of products including a wide variety of over the counter (OTC) drugs in various finished dosage forms for both human and veterinary use. The unit has been approved by the WHO
• Overall financials were affected adversely on account of these cGMP issues. Revenues declined ~4% YoY whereas EBITDA margins contracted 830 bps to 17%
• The Ratlam facility received the cGMP certificate from the UKMHRA. UKMHRA also inspected the Pithampur facility recently without any major observations
What to watch • The company has given a revenue guidance of 7-8% (including
US shipments) with EBITDA margins expected to be around 17-18%. Domestic formulations growth expected to be 12% in FY16
• Normalisation of institutional business is expected from H2FY16 • Updates on remedial measures post receiving the compliance
enhancement plan from consultants, the company will approach the USFDA for re-inspection
[
Stock Performance
80
100
120
140
160
180
200
220
240
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
Mar
-15
CNX Pharma Ipca labs Nifty

ICICI Securities Ltd. | Retail Equity Research
Page 15
Jubilant Life Sciences
Sustainability of pharma important for overall improvement
FY15 Highlights • The company continued to struggle on both fronts - pharma
segments and life science ingredients (LSI) segment. The impact was more severe on EBITDA margins. While revenues were flat YoY, EBITDA margins plunged ~510 bps YoY to 12.6%
• The pharma segment was affected by a decline in the contract manufacturing business (owing to the warning letter at US based Spokane facility) and decline in finished dosage formulations (generics business) owing to slower approvals from the USFDA.
• The LSI segment was affected due to regulatory changes in China and pricing pressure in some sub-segments
• Till FY15, cumulative ANDA filings stood at 72 including 38 approvals. Globally, it has launched 48 products including more than 20 products in the US, 29 in Europe and 25 in RoW markets. The company has 38 pending ANDAs till date.
• The company acquired the remaining stake (82%) in Cadista Holding for ~| 170 crore
• No respite was on the gross debt level as well, which increased to | 4790 crore from | 4390 crore in FY14
What to watch • After the cGMP clearance of Spokane facility by the USFDA, we
expect some recovery in the CMO business (comprises 12% revenues on a normalized basis) in FY16
• The generics business is expected to witness higher traction on account of incremental ANDA approvals and launches
• The LSI segment is expected to see some traction from nutritional products on account of volume improvement
• The company has guided at ~| 350 crore of capex for FY16. The capex has been earmarked for debottlenecking in the pharma segment
• Overall, with a likely improvement in the pharma segment, we expect a recovery in EBITDA margins and on the debt front
Lupin
Gavis acquisition optically bold move; to provide major thrust for US growth
FY15 Highlights • The company registered ~13% YoY revenues growth driven by
~16% growth in the US and ~20% growth in domestic formulations. The Japanese business, on the other hand, grew just ~2% YoY
• An improved product mix and operational efficiency resulted into 250 bps improvement in EBITDA margins to 28.7%
• On the cGMP front, Lupin’s manufacturing facility at Pithampur, near Indore had received six USFDA observations (483s). The facility was audited by USFDA in January, 2015. Despite observations, the company has been continuously receiving USFDA product approval and site-transfer approvals from this facility
• The company acquired a Mexican company Laboratories Grin, which owns ophthalmic portfolio in the Mexican market
• Lupin launched InspiraChamber, an anti-static valved holding chamber (VHC) in the US under a licensing agreement with
Stock Performance
20
70
120
170
220
270
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
Mar
-15
CNX Pharma Jubilant Life Sciences Nifty
[
Stock Performance
80
130
180
230
280
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
Mar
-15
CNX Pharma Lupin Nifty

ICICI Securities Ltd. | Retail Equity Research
Page 16
InspiRX Inc. The agreement grants Lupin exclusive rights to promote, distribute and market InspiraChamber VHC in the US
• Lupin forayed into the Canadian market by entering into an agreement with Salix Pharma to market GI drugs
• The product pipeline in the US (210 ANDAs filed, 111 approved and 77 launches) includes some limited competition products and Para IV/FTF opportunities
• Lupin has entered into a long term strategic partnership with Merck for implementation of Merck’s general medicines portfolio expansion initiative in emerging markets
• Lupin forays into Brazilian market with the acquisition of a company called Medquimica. This company registered sales of US$31 million in CY14
What to watch • The company expects revenue growth to be a tad below the
earlier guidance of 10-15% with EBITDA margins at 26-28% • Integration of US based Gavis Pharma, which it acquired recently
and raising of debt for the same • Resolution of cGMP Form 483 observations, which the company
received for the Goa facility • Lupin expects 20 ANDA filings in FY16 and 15-20 product
approvals • It expects to receive dermatology ANDA approvals post FY17
and injectable and respiratory approvals post FY18 • The company expects to get gNexium approval by H2FY16 • Lupin and Celon Pharma SA (Celon) will jointly develop a
fluticasone/salmeterol dry powder inhaler (DPI) product, which is a generic version of GSK's Advair Diskus. Lupin will be responsible for commercialisation of the product. Celon will supply the product to Lupin for its commercialisation in the US, Canada, Mexico, and other key markets

ICICI Securities Ltd. | Retail Equity Research
Page 17
Exhibit 15: Notable Para IV Filings Brand Name Indicator Generic Name NDA holder Date
Axiron testosterone hypogonadism Eli Lilly 6-Jul-15
Celebrex pain and inflammation benzenesulfonamide Pfizer 5-Mar-13
Detrol LA overactive bladder tolterodine tartrate Pfizer 21-Mar-13
Lyrica diabetic peripheral neuropathy; pregabalin Pfizer Inc. 29-Apr-09
Multaq prevention of cardiovascular hodronedarone hydrochloride Sanofi 21-May-15
Namenda treatment of Alzheimer’s Diseasmemantine hydrochloride Forest Laboratories, Inc 28-Jan-08
Natazia oral contraceptive estradiol valerate and dienoges Bayer 28-Nov-12
Nexium acid reflux esomeprazole magnesium AstraZeneca AB 21-Oct-09
Nuvigil excessive sleepiness associate armodafinil Cephalon Inc., Cephalon 16-Mar-10
Paxil CR Anxiety Paroxetine Hydrochloride Apotex Inc. 1-May-15
Pennsaid osteoarthritis diclofenac Novo 14-Jan-15
Phoslyra end stage renal disease calcium acetate Fresenius Medical 27-Feb-14
Prevacid gastroesophageal reflux lansoprazole Takeda Pharma 29-Nov-12
Prezista HIV darunavir Tibotec Inc. 15-Nov-10
Prolensa postoperative eye inflammationbromfenac Bausch & Lomb Inc 31-Jan-14
Quartette Contraceptive Ethinyl Estradiol; Levonorgestre Teva Pharma 4-Jun-14
Renagel renal disease sevelamer hydrochloride Genzyme Corp. 5-Mar-09
Renvela chronic kidney disease sevelamer carbonate Genzyme Corp. 14-May-09
Roxane Calcium ARenal Disease NA Roxane Lab 10-Feb-15
Safyral birth control drospirenone Merck 21-May-15
Savella fibromyalgia milnacipran Forest Lab 23-Sep-13
Seroquel XR schizophrenia, bipolar disorder, quetiapine fumarate Astrazeneca 5-Nov-12
Toviaz overactive bladder fesoterodine fumarate Pfizer 28-Jun-13
TravatanZ elevated intraocular pressure travoprost ophthalmic solution Alcon Research 20-Jul-15
Truvada HIV tenofovir disoproxil fumarate/emGilead Sciences 16-Aug-12
Uloric gout febuxostat Takeda 12-Feb-14
Vimovo osteoarthritis, rheumatoid arthr naproxen / esomeprazole magn Astrazeneca 25-Jul-11
Viread HIV tenofovir disoproxil fumarate Gilead Sciences 20-Aug-12
Welchol high cholesterol and type 2 diabcolesevelam hydrochloride Daiichi Sankyo Inc. 30-Jul-10
Zorvolex Anti-Inflammatory Diclofenac Iroko Pharmaceuticals 7-Jan-15
Nuvigil excessive sleepiness associate armodafinil Cephalon Inc. 2-Nov-11
Revlimid myelodysplastic syndrome; mu lenalidomide Celgene Corp. 8-Oct-10
Tamiflu influenza oseltamivir phosphate Roche 15-Mar-11
Treanda chronic lymphocytic leukemia bendamustine Cephalon Inc. 27-May-14
Apriso ulcerative colitis mesalamine Salix Pharmaceuticals I 18-Feb-14
Exelon dementia rivastigmine Novartis 3-Apr-13
Lyrica diabetic peripheral neuropathy; pregabalin Pfizer 7-Jan-11
Phoslo kidney disease calcium acetate Fresenius Medical 20-Feb-15
Suprep laxative sodium / potassium / magnesiu Braintree Laboratories, 9-Mar-11
Aloxi prevention of acute nausea and palonosetron hydrochloride Roche 13-Feb-15
Phoslo kidney disease calcium acetate Fresenius Medical 17-Jul-15[
Source: Bloomberg, ICICIdirect.com Research; Included Subsidiaries filings

ICICI Securities Ltd. | Retail Equity Research
Page 18
Natco Pharmaceuticals
Copaxone, gSovaldi, domestic oncology and a unique product pipeline
FY15 Highlights • The company tied up with Gilead Sciences to launch Hepatitis C
drug gSovaldi in 91 countries • Revenues increased 12% YoY mainly due to 26% growth in the
domestic oncology segment and 29% growth in the API segment. However, due to the absence of Venezuelan tenders, export formulation growth was muted
• EBITDA expanded ~160 bps YoY to 26% mainly due to lower raw material cost led by a better product mix.
• US ANDAs filings stood at 35 including 19 pending approvals. In FY15, it filed five ANDAs and received two approvals
• Out of the eight companies having Sovaldi tie up with Gilead Sciences for 91 emerging markets, only Natco and Hetero have Sovaldi API manufacturing approvals
What to Watch • We expect the company to launch gCopaxone 20 mg (US$3
billion) in Q3FY16. Sandoz has launched gCopaxone 20 mg in the US and gained ~28% market share (Source: Bloomberg)
• The company also expects to receive USFDA approvals for gEntocort (US$400 million) & gTracleer (US$585 million) in FY16
• Due to first mover and low cost competitive advantage (backed by own API), we expect the company to rake in ~| 120 core in FY16E and ~| 200 crore in FY17E from Sovaldi
• The management expects | 350-400 crore from domestic formulation sales (including more than | 100 crore sales from Sovaldi)
• It plans to file six to seven ANDAs in FY16. Sun Pharmaceuticals
Short term pains to remain but long term story in tact
FY15 Highlights • In a landmark deal, the company acquired Ranbaxy in a shares
swap deal. Under the terms of the agreed deal, Ranbaxy shareholders had received 0.8 Sun Pharma share for each share of Ranbaxy. The deal was closed on April 6, 2015
• The company’s Halol facility received 23 USFDA observations. This facility has contributed 55-60% of the company’s US sales (ex-Taro) in FY14
• Due to Ranbaxy’s acquisition and supply constraint in the US due to USFDA observations for its Halol Facility, FY15 revenue was muted while EBITDA margins contracted to 29.4% in FY15 from 44.7% in FY14
• Taro registered ~14% YoY growth in revenues driven by price hikes even as volumes declined. Operating profit margins improved 440 bps to 61.1% on the back of improved product mix
• The company acquired the opiates business of GlaxoSmithKline (GSK) in Australia including manufacturing sites in Latrobe (Tasmania) and Port Fairy (Victoria) and its portfolio of opiates products (for Analgesics) with annual sales of ~US$70 million
• Sun and Merck entered into an exclusive worldwide licensing agreement for Merck’s investigational therapeutic antibody candidate, Tildrakizumab, (MK-3222), which is currently being evaluated in phase III registration trials for the treatment of
Stock Performance
80
130
180
230
280
330
380
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
CNX Pharma Natco PharmaNifty
Stock Performance
80
130
180
230
280
330
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
CNX Pharma Sun Pharma Nifty

ICICI Securities Ltd. | Retail Equity Research
Page 19
chronic plaque psoriasis, a skin ailment. Under the terms of the agreement, Sun Pharma will acquire the worldwide rights to Tildrakizumab for use in all human indications from Merck in exchange for an upfront payment of US$80 million
• The company has settled the patent litigation case of Gleevec (Imatinib Mesylate), which is used for the treatment of chronic myeloid leukaemia, with Novartis in the US. Under the settlement agreement, Sun Pharma’s subsidiary may launch its generic version in the US market on February 1, 2016
• The company closed its manufacturing plant in Detroit in an effort to trim a few of its manufacturing facilities in the US. The plant is operated by Sun Pharma’s subsidiary Caraco Laboratories. The plant is one of eight manufacturing facilities that the company owns in the US
• The US product basket remains robust - 597 ANDAs filed, 438 approvals received; some niche launches include Lipodox/ Doxil, Doxycycline, Nystatin, etc. besides complex/limited competition products and plain vanilla generics
What to Watch
• USFDA clearance of Halol and Karkhadi manufacturing facilitations in FY16
• We expect FY16 numbers to be subdued due to Ranbaxy consolidation and remedial measures at Halol and other plants
• Launch of gGleevec in Q4FY16

ICICI Securities Ltd. | Retail Equity Research
Page 20
Exhibit 16: Notable Para IV Filings Brand Name Indicator Generic Name NDA holder ANDA Filer Date
Reclast osteoporosis and Paget's zolendronic acid Novartis Pharma Caraco Pharma (Sun) 20-Feb-13
Antara hypercholesterolemia; dy fenofibrate Lupin Atlantis Holdings Ranbaxy (Sun) 4-Aug-10
Atelvia post-menopausal osteoporisedronate sodium Warner Chilcott Compa Ranbaxy (Sun) 26-Apr-12
Gleevec leukemia imatinib mesylate Novartis Ranbaxy (Sun) 29-Dec-14
Namenda XR Alzheimer's disease memantine Teva Ranbaxy (Sun) 30-May-14
Opana ER moderate to severe pain oxymorphone Endo Pharm Ranbaxy (Sun) 3-Dec-13
Ortho TRI-Cyclen LO oral contraceptive ethinyl estradiol; norgestimate) Purdue Pharma Ranbaxy (Sun) 8-Apr-11
Oxecta moderate to severe pain oxycodone hydrochloride Acura Pharma Ranbaxy (Sun) 29-Apr-13
Savella fibromyalgia milnacipran Forest Lab Ranbaxy (Sun) 23-Sep-13
Uloric gout febuxostat Takeda Ranbaxy (Sun) 12-Feb-14
Vascepa hypertriglyceridemia icosapent ethyl Amarin Pharma Ranbaxy (Sun) 20-Jun-14
Vimpat epilepsy lacosamide UCB Pharma Ranbaxy (Sun) 10-Jul-13
Xyrem narcolepsy paracetamol Jazz Pharma Ranbaxy (Sun) 2-Oct-14
Alimta lung cancer; mesotheliompemetrexed disodium Eli Lilly Sun Pharma 13-Sep-13
Allegra seasonal allergies fexofenadine hydrochloride Aventis PharmaceuticalSun Pharma 9-Oct-09
Allegra-D seasonal allergies fexofenadine hydrochloride / pseuAventis PharmaceuticalSun Pharma 23-Jan-09
AMPYRA multiple sclerosis Dalfampridine Alkermes Pharma Sun Pharma 18-May-15
Angiomax anticoagulant for coronar bivalirudin Medicis Pharma Sun Pharma 21-Nov-11
Crestor high cholesterol rosuvastatin calcium AstraZeneca Pharmace Sun Pharma 26-Apr-10
Effient acute coronary syndromeprasugrel hydrochloride Eli Lilly Sun Pharma 12-Mar-14
Focalin Attention Deficit Hyperac dexmethylphenidate hydrochloridCelgene Corp. Sun Pharma 1-Jul-10
Gabitril partial seizures tiagabine hydrochloride Cephalon Inc. Sun Pharma 21-Sep-11
GanirelixAcetate IVF ganirelix acetate Merck Sun Pharma 27-Aug-12
Gleevec leukemia imatinib mesylate Novartis Sun Pharma 6-Feb-14
Glumetza type 2 diabetes mellitus metformin hydrochloride Depomed, Inc. Sun Pharma 20-Jun-11
Lyrica diabetic peripheral neuroppregabalin Pfizer Inc. Sun Pharma 29-Apr-09
Multaq cardiovascular hospitalizadronedarone hydrochloride Sanofi Sun Pharma 6-Mar-14
Namenda Alzheimer's disease memantine hydrochloride Forest Sun Pharma 14-Feb-14
Nexium acid reflux esomeprazole sodium AstraZeneca AB Sun Pharma 26-Feb-10
Onglyza type II diabetes Saxagliptin Astrazeneca Sun Pharma 3-Jun-14
Reclast osteoporosis and Paget's zolendronic acid Novartis Pharma Sun Pharma 14-Jul-12
Ryzolt chronic pain relief tramadol hydrochloride Purdue Pharma ProductSun Pharma 5-Nov-09
Taclonex psoriasis vulgaris calcipotriene / betamethasone di Cancer Research Tech. Sun Pharma 15-Sep-10
Treanda chronic lymphocytic leukebendamustine Cephalon Inc. Sun Pharma 26-Dec-13
Treximet migraine sumatriptan / naproxen sodium Pozen Inc. Sun Pharma 26-Nov-11
Velcade cancer bortezomib Millennium Sun Pharma 25-Jun-15
Vimpat epilepsy lacosamide UCB Pharma Sun Pharma 10-Jul-13
Yaz contraception; acne; premdrospirenone / ethinyl estradiol Bayer Sun Pharma 12-Jan-11
Zemplar secondary hyperthyroidis paricalcitol AbbVie Inc Sun Pharma 24-Jan-13
Acanya severe acne benzoyl peroxide Dow Pharma Taro Pharma (Sun) 13-Aug-14
Naftin Fungal Infection Naftifine Hydrochloride Merz Pharma Taro Pharma (Sun) 14-May-15
Suprep colon cleansing sodium sulfate Braintree Lab Taro Pharma (Sun) 9-Oct-14 [
Source: Bloomberg, ICICIdirect.com Research; *Included Subsidiaries filings

ICICI Securities Ltd. | Retail Equity Research
Page 21
Torrent Pharma
US, Brazil, India key catalysts for future
FY15 Highlights • Revenues grew 11% mainly due to consolidation of Elder’s
domestic portfolio. The US remained in good shape despite the exclusivity sunset of gCymbalta
• Acquired dermatology arm of Encore Group’s Zyg Pharma. The company has USFDA approved facility in Pithampur (Indore). Torrent expects to start monetising its portfolio within two years
• In the US, Torrent owns a healthy product pipeline (67 filed ANDAs, 48 approvals). It has received six ANDA approvals during the year
• The company has also entered into an exclusive licensing agreement with Reliance Life Sciences for marketing three biosimilars in India
• The company has completed the acquisition of minocycline tablets and capsules from Sun Pharma in April 2015
What to watch • Expects gNexium and gDetrol approval from USFDA in FY16. It
has already launched gAbilify in April 2015. Overall, it is planning 15-18 ANDA filings in the US and eight to 10 product launches
• Commercialisation of the Dahej facility, which was approved by the USFDA will start by the end of FY16
• The company expects the Elder portfolio to be EPS accretive by the second year of acquisition i.e. in FY16
• Expects double digit constant currency growth in Brazil in FY16 • The MR strength is expected to come down by 300 to 3000 by
the end of FY16 in accordance with the rationalisation of the Elder portfolio
• FY16 R&D spend is expected to be more than 6% of sales. The company also plans to add ~400 scientists to its existing R&D team of 600 scientists
Exhibit 17: Notable Para IV Filings Brand Name Indicator Generic Name NDA holder ANDA Filer Date
Bystolic Hypertension nebivolol Forest Lab Torrent Pharma 13-Mar-12
Crestor high cholesterol rosuvastatin calcium AstraZeneca Pharmace Torrent Pharma 8-Jul-10
Daliresp Chronic Obstructive PulmRoflumilast AstraZeneca Torrent Pharma 15-May-15
Detrol LA overactive bladder tolterodine tartrate Pfizer Torrent Pharma 25-Jul-11
Nexium gastroesophegal reflux di esomeprazole magnesium AZ Torrent Pharma 26-Jan-12
Viagra erectile dysfunction pyrazolopyrimidinones Pfizer Torrent Pharma 17-Mar-14[
Source: Bloomberg, ICICIdirect.com Research
Unichem Laboratories
Domestic business likely to crawl back to normal
FY15 Highlights • Revenues of the company grew just ~7% on the back of
formulation exports. Domestic formulations, on the other hand, continued to remain under pressure due to ongoing rationalisation at the distribution level. EBITDA margins dropped to a record low level at ~9% due to pressure in domestic formulations, which fetches high margins
• Domestic formulations struggled during FY15 as the company initiated a portfolio realignment exercise as Unichem had
Stock Performance
80
130
180
230
280
330
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
Mar
-15
CNX Pharma Torrent PharmaNifty
Stock Performance
60
110
160
210
260
Mar
-14
May
-14
Jul-1
4
Sep-
14
Nov
-14
Jan-
15
Mar
-15
CNX Pharma Unichem LabsNifty

ICICI Securities Ltd. | Retail Equity Research
Page 22
initiated conversion from a distribution-driven model to C&F driven model for better working capital management. According to the management’s own admission, ~50% of the domestic portfolio is mature and declining. Similarly, close to 20% of the domestic portfolio is under NLEM
• NLEM impact on two of its major drugs i.e. Losar and Trika led to a | 10.8 crore loss in FY15
• Total ANDA filing till FY15 stood at 34 of which, it has launched 12 products and is awaiting approvals for 17 ANDAs (including two tentative approvals)
• R&D spend for FY15 was 5% of revenues
What to watch • The company has guided for 9-10% YoY growth in FY16 in
domestic formulation sales • The company has guided for | 150 crore capex mainly for
debottlenecking at its Pithampur (API) and Goa plant • It plans to file five to six ANDAs in the US • It is expecting four to six approvals in Brazil

ICICI Securities Ltd. | Retail Equity Research
Page 23
Exhibit 18: USFDA approvals for companies under I-Direct coverage in FY15 Date Company Drug Name Therapeutic Area Innovator company Generic Version of Market Size
Jun-14 Alembic Pharms Lamotrigine tablets Anti-epileptic GlaxoSmithKline Lamictal NA
May-14 Aurobindo Pharma Olanzapine tablets CNS Eli Lilly Zyprexa US$ 3.2 billion
Jun-14 Aurobindo Pharma Divalproex Sodium ER tablets CNS Abbvie Depakote ER US$ 194 million
Sep-14 Aurobindo Pharma Amoxicillin Anti-Bacterial Dr Reddy's Labs Amoxil US$19 million
Jan-15 Aurobindo Pharma Valsartan CVS Novartis Diovan US$ 1.8 billion
Jan-15 Cadila Healthcare Calcium Acetate Blood phosphate Cypress Pharm Eliphos NA
Apr-14 Caraco* Furosemide tablets CVS Sanofi Aventis Lasix US$ 70 million
Nov-14 Cipla Finasteride Baldness Merck Propecia US$ 130 million
Apr-14 Dr Reddys Labs Eszopiclone tablets Insomnia Sepracor Lunesta US $ 787 million
Jun-14 Dr. Reddys Labs Paricalcitol capsules Hormonal Abbvie Zemplar US$ 110 million
Oct-14 Dr. Reddy's Labs Sirolimus Oral Tablet 1mg, 2mg Anti-Infective PF Prism CV Rapamune US$ 206 million
Nov-14 Dr. Reddy's Labs Valganciclovir Hydrochloride Anti-Infective Roche Valcyte US$ 388 million
Nov-14 Dr. Reddy's Labs Fexofenadine HCI And Pseudoephedrine HCI Anti-Allergy Sanofi Allegra-D NA
Feb-15 Glenmark Ethyl Estradiol; Levonorgestrel (Ashlyna) Oral Contraceptive Teva Seasonique US$ 161 million
Apr-14 Glenmark Generics Eszopiclone tablets Insomnia Sepracor Lunesta US $ 787 million
Jul-14 Glenmark Generics Telmisartan tablets CVS Boehringer Ingelheim PharMicardis US$ 250 million
Jul-14 Glenmark Generics Fluocinonide cream Anti-Inflamatory Medicis Vanos US$ 103 million
Nov-14 Jubilant Zolmitriptan CNS IPR Zomig US$ 149 million
Dec-14 Jubilant Rizatriptan Benzoate Anti-Migraine Merck Maxalt US$ 193 million
Feb-15 Jubilant Life Sciences Irbesartan Tablets CNS Sanofi-Aventis Avapro US$ 50 million
Jan-15 Jubilent Life Valsartan CVS Novartis Diovan US$ 1.8 billion
Apr-14 Lupin Pioglitazone Hydrochloride Anti-Diabetic Takeda Actos US$ 3 billion
May-14 Lupin Drospirenone; Ethinyl Estradiol tablets Oral Contraceptive Bayer Healthcare Yaz US$ 347 million
Jun-14 Lupin Levonorgestrel OTC tablets Oral Contraceptive Teva Plan B One stop NA
Dec-14 Lupin Lamivudine HIV ViV Healthcare Epivir NA
Jan-15 Lupin Valsartan CVS Novartis Diovan US$ 1.8 billion
Jan-15 Lupin Vancomycin Hydrochloride Diarrhoea ANI Pharmaceuticals Vancocin US$ 164 million
Feb-15 Lupin Bimatoprost Opthalmic Allergan Lumigan US$ 600 million
Mar-15 Lupin Amlodipine and Valsartan CNS Novartis Exforge US$ 415 million
Sep-14 Lupin Ltd. Moxifloxacin Hydrochloride Solution/Drops Opthalmic Alcon Pharms Moxeza/Vigamox NA
Oct-14 Lupin Ltd. Celecoxib Oral Capsule 50mg, 100mg, 200mg, 400mg Anti-Inflammatory GD Searle Celebrex US$ 2.44 billion
Oct-14 Natco Pharma Carisoprodol Tablet 350mg CNS Meda Pharms Soma NA
Jun-14 Orchid Healthcare Eszopiclone tablets Insomnia Sepracor Lunesta US $ 787 million
Jul-14 Strides Acrolab Tacrolimus Capsule Anti-Infective Astellas Protopic US$ 676 million
Dec-14 Strides Acrolab Calcitriol Oral 0.25MCG, 0.50MCG Vitamins Validus Pharms Rocaltrol US$ 50 million
Jun-14 Strides Acrolabs Pramipexole Dihydrochloride tablets CNS Boehringer Ingelheim PharMirapex NA
Jun-14 Strides Acrolabs Methoxsalen capsules Anti-psoriasis Dow Pharma Oxsoralen-Ultra US$ 13.6 million
Jun-14 Strides Acrolabs Imiquimod cream Skin Diseases Medicis Aldara NA
Aug-14 Strides Acrolabs Buspirone Hydrochloride Tablets Anxiety Bristol Myers Squibb Buspar NA
Apr-14 Sun Pharma Oxaliplatin injection Anti-Cancer Sanofi Eloxatin US$1.4billion
Source: USFDA website, ICICIdirect.com Research; * Subsidiary of Sun Pharma

ICICI Securities Ltd. | Retail Equity Research
Page 24
Exhibit 19: USFDA approvals for companies under I-Direct coverage in FY15 Date Company Drug Name Therapeutic Area Innovator company Generic Version of Market Size
Jun-14 Sun Pharma Risedronate Sodium tablets (75 mg and 150 mg) Osteoporosis Warner Chilcott Actonel US$ 180 million
Jul-14 Sun Pharma Clonidine Hydrochloride Tablets Hypertension Boehringer Ingelheim PharCatapres US$ 72 million
Aug-14 Sun Pharma Clonazepam Tablets CNS Roche Clonazeepam NA
Sep-14 Sun Pharma Opcicon One Step Anti-Contraceptive Bayer Healthcare Mirena US$81.1 million
Nov-14 Sun Pharma Zolmitriptan CNS IPR Zomig US$ 149 million
Dec-14 Sun Pharma Bupropion Hydrochloride 150mg Tablet Anti-depressant GSK Wellbutrin US$ 268 million
Jan-15 Sun Pharma fexofenadine HCL and pseudoephedrine HCL Anti-allergic Sanofi Allegra-D (OTC) US$ 49.8 million
Apr-14 Sun Pharma Global Niacin tablets CVS Abbvie Niaspan US$ 1.2 billion
Apr-14 Taro Pharma Phenytoin Anti-Epileptic Parke Davis Dilantin US$ 125 million
Jul-14 Taro Pharma Fluocinonide cream Anti-Inflamatory Medicis Vanos US$ 103 million
Oct-14 Taro Pharma Desonide Dermatology Galderma Labs Desowen NA
Nov-14 Taro Pharma Diclofenac Sodium Anti-Inflammatory Nuvo Res Pennsaid US$ 13.3 million
Jul-14 Torrent Pharma Telmisartan tablets CVS Boehringer Ingelheim PharMicardis US$ 250 million
Jan-15 Torrent Pharma Valsartan CVS Novartis Diovan US$ 1.8 billion
Mar-15 Torrent Pharma Amlodipine and Valsartan CNS Novartis Exforge US$ 415 million
Apr-14 Torrent Pharms Moxifloxacin Hydrochloride tablets Anti-Infective Bayer Avelox US$ 195 million
Jul-14 Zydus Pharma Etomidate Injection Anesthesia Hospira Amidate US$ 6.5 million
Aug-14 Zydus Pharma Acyclovir Tablets Anti-Infective Delcor Asset Zovirax US$55.8 million
Aug-14 Zydus Pharma Telmisartan Tablets CVS Boehringer Ingelheim PharMicardis US$ 250 million
Source: USFDA, ICICIdirect.com Research; * subsidiaries of Sun Pharma

ICICI Securities Ltd. | Retail Equity Research
Page 25
Exhibit 20: Tentative USFDA approvals received by companies under I-Direct Universe in FY15 Date Company Drug Name Therapeutic Area Innovator company Generic Version of Market Size
Apr-14 Alembic Memantine Hydrochloride tablets CNS Forest Labs Nameda NA
Mar-15 Aurobindo Pharma Lacosamide Epilepsy USB Inc. Vimpat US$ 365 million
Mar-15 Cadila Ritonavir HIV Abbvie Norvir US$ 400 million
May-14 Cipla Zoledronic Acid injections Anti-Cancer Novartis Zometa US$ 731 million
May-14 Dr Reddy's Valganciclovir tablets Anti-infective Roche Valcyte US$ 390 million
Apr-14 Jubilant Life Memantine Hydrochloride tablets CNS Forest Labs Nameda NA
May-14 Lupin Norethindrone Acetate;Ethinyl Estradiol;Ferrous Fumarate tab Oral Contraceptive Warner Chloctt Loestrin 24 FE US$ 390 million
Feb-15 Lupin Tenofovir Disoproxil Fumarate HIV Gilead Sciences Viread US$ 1 billion
Oct-14 Natco Pharma Armodafinil 50mg, 150mg, 250mg Tablets Wakefulness Cephalon Nuvigil US$ 457 million
Jan-15 Stride Arcolab Rilpivirine HIV Janssen Prods Edurant NA
Jun-14 Sun Pharma Risedronate Sodium tablets (5 mg, 30mg and 35 mg) Paget’s disease of the boWarner Chilcott Actonel & Atelvia US$ 942 million
Jul-14 Sun Pharma Zoledronic Acid Injection Osteoporosis Novartis Reclast US$ 355 million
Apr-14 Sun Pharma Global Fexofenadine; Pseudoephedrine Hydrochlorides ER tablets Anti-Biotic Sanofi Aventis Allegra D 12 US$ 214 million
Apr-14 Sun Pharma Global Oxaliplatin injection (200mg / 40ml) Anti-Cancer Sanofi Eloxatin US$ 104 million
Jan-15 Taro Pharma Desloratadine Anti-allergic Merck Clarinex NA
Sep-14 Torrent Pharma Amlodipine and Valsartan CVS Novartis Exforge NA
Oct-14 Torrent Pharma Amlodipine; Valsartan; Hydrochlorothaizide Oral Tablets CVS Novartis Exforge NA
Sep-14 Zydus Pharma Glipizide Anti-Diabetic Pfizer Glucotrol US$90.1 million
Source: Company, ICICIdirect.com Research

ICICI Securities Ltd. | Retail Equity Research
Page 26
Exhibit 21: Patent litigations - Indian companies vs. innovators Month Innovator ANDA Filer Brand Name API Used for Apr-14 Kowa Aurobindo Pharma Livalo Pitavastatin CVSApr-14 Medicines co Aurobindo Pharma Angiomax Bivalirudin CVSApr-14 Kowa Aurobindo Pharma Livalo Pitavastatin CVSMay-14 Senju Pharmaceuticals Aurobindo Pharma Zymaxid Gatifloxacin ConjunctivitisMay-14 Celgene Corp. Natco Pharma Revlimid Lenalidomide Multiple MyelomaMay-14 Otsuka Pharmaceutical Co. Alembic Pharma Abilify Aripiprazole CNSMay-14 Purdue Pharmaceutical Products LP Dr Reddy's Intermezzo Zolpidem InsomniaMay-14 Otsuka Pharmaceutical Co. Cadila Healthcare Abilify Aripiprazole CNSMay-14 Otsuka Pharmaceutical Co. Aurobindo Pharma Abilify Aripiprazole CNSMay-14 AstraZeneca AB Aurobindo Pharma Onlyza Saxagliptin Type 2 DiabetesMay-14 AstraZeneca AB Glenmark Generics Onlyza Saxagliptin Type 2 DiabetesMay-14 AstraZeneca AB Wockhardt Onlyza Saxagliptin Type 2 DiabetesMay-14 Otsuka Pharmaceutical Co. Wockhardt Abilify Aripiprazole CNSMay-14 Forest Laboratories Inc. Ranbaxy Labs Namenda XR Memantine CNSJun-14 AstraZeneca Pharmaceuticals LP Sun Pharma Global FZE; Onglyza Saxagliptin DiabetesJun-14 Bristol-Myers Squibb Co. Aurobindo Pharma Reyataz Atazanavir HIVJun-14 Teva Women's Health Lupin Quartette Levonorgestre ContraceptiveJun-14 Forest Laboratories Inc Aurobindo Pharma Namenda XR Memantine CNSJun-14 Senju Pharma Lupin Pharma Inc. Prolensa Bromfenac Ocular InflammationJul-14 Pfizer Inc Aurobindo Pharma Tygacil Tygecycline InfectionJul-14 GlaxoSmithKline LLC Glenmark Generics Inc. Coreg Carvedilol High Blood PressureJul-14 Allos Therapuetics Inc Dr Reddy's Folotyn Pralatrexate LymphomaJul-14 Acorda Therapeutics Inc.; Aurobindo Pharma Ampyara Dalfampridine Multiple SyrosysJul-14 Alcon Pharmaceuticals Ltd. Aurobindo Pharma Vigamox Moxifloxacin Eye InfectionJul-14 Gilead Sciences Inc. Lupin Ltd. Atripla Efavirenz/Emtricitabine/Tenofovir HIVJul-14 AbbVie Inc. Aurobindo Pharma Norvir Ritonavir HIVJul-14 Alkermes Pharma Ltd. Sun Pharmaceuticals Focalin XR Dexmethylphenidate ADHDJul-14 Otsuka Pharma Co. Torrent Pharma Abilify Aripiprazole CNSJul-14 Astrazeneca AB Cadila Healthcare Nexium Esomeprazole Acid RefluxAug-14 Janssen Products LP Cipla Ltd. Prezista Darunavir HIVAug-14 Forest Labs Lupin Ltd. Namenda Memantine Hydrochloride CNSAug-14 Senju Pharma Ltd. Lupin Ltd. Prolensa Bromfenac Ocular InflammationAug-14 Novartis Pharma Dr. Reddy's Labs Gleevec Imatinib Mesylate Organ RejectionSep-14 Millenium Pharmaceuticals Glenmark Pharma Valcade Bortezomib Multiple MylomaSep-14 Teva Pharma Dr. Reddy's Labs Copaxon Glatiramer Acetate Multiple SclerosisSep-14 Reckitt Benckiser LLC Aurobindo Pharma Mucinex Guaifenesin MucusSep-14 Cephalon Inc. Dr. Reddy's Labs Treanda Bendamustine LeukemiaSep-14 Cephalon Inc. Sun Pharma Treanda Bendamustine LeukemiaOct-14 Novartis Pharma Dr. Reddy's Labs Gleevec Imatinib LeukemiaOct-14 Braintree Laboratory Inc. Taro Pharma Suprep Sodium Sulphate Bowel PreparationOct-14 Cephalon Inc. Wockhardt Treanda Bendamustine LeukemiaNov-14 Biomarin Pharma Dr. Reddy's Labs Kuvan Sapropterin Dihydrochloride phenylketonuriaNov-14 Otsuka Pharma Cadila Healthcare Abilify Aripiprazole CNSNov-14 Otsuka Pharma Alembic Pharma Abilify Aripiprazole CNS
Jan-15 iCeutica Lupin Ltd. Zorvolex Diclofenac Anti-Inflammatory
Jan-15 Senju Pharma Lupin Ltd. Prolensa Bromfenac Eye Inflammation
Jan-15 Sheonogi & Co. Aurobindo Pharma Doribax Doripenem Infection
Jan-15 Teva Pharma Dr. Reddy's Labs Copaxone Glatiramer Multiple Sclerosis
Feb-15 Roxane Laboratories Lupin Ltd. Roxane Calcium Acetate CCalcium Acetate Serum phosphorous Reduction
Feb-15 Cephalon Inc. Glenmark Generics Treanda Bendamustine Leukemia
Feb-15 Cephalon Inc. Dr. Reddy's Labs Treanda Bendamustine Leukemia
Mar-15 Otsuka Pharma Indoco Remedies Abilify Aripiprazole CNS
Source: Bloomberg, ICICIdirect.com Research

ICICI Securities Ltd. | Retail Equity Research
Page 27
ICICIdirect.com coverage universe (Healthcare) I-Direct CMP TP Rating M CapCode (|) (|) (| Cr) FY15 FY16E FY17E FY15 FY16E FY17E FY15 FY16E FY17E FY15 FY16E FY17E FY15 FY16E FY17E
Ajanta Pharma AJAPHA 1711 1488 BUY 14481.0 38.3 45.6 53.1 33.1 27.8 23.9 7.8 6.5 5.3 50.6 48.7 46.4 41.9 38.5 35.9
Apollo Hospitals APOHOS 1428 1454 BUY 20111.9 460.2 540.5 0.0 3.1 2.6 0.0 ### ### 0.0 6.1 8.4 9.6 0.0 0.0 0.0
Aurobindo Pharma AURPHA 761 840 BUY 45461.6 28.0 36.7 42.0 27.9 20.5 17.9 10.1 7.8 6.3 23.5 25.5 25.0 30.3 29.9 26.1
Biocon BIOCON 462.6 469 HOLD 9313.0 20.4 23.0 28.0 18.8 20.4 16.7 6.9 5.4 4.1 10.4 11.8 13.3 12.5 12.7 13.9
Cadila Healthcare CADHEA 1914 1913 BUY 38798.8 56.2 75.1 95.6 20.8 15.6 12.2 14.7 11.2 8.6 21.3 24.6 26.6 26.4 27.3 26.8
Cipla CIPLA 719.2 650 HOLD 57442.1 14.9 22.3 29.5 30.1 20.1 15.2 3.4 2.8 2.2 13.2 17.6 19.9 10.8 14.3 16.2
Dr Reddy's Labs DRREDD 4224 4600 BUY 72652.6 130.6 159.9 191.6 21.6 17.7 14.7 13.9 11.2 9.2 17.0 19.3 20.6 19.9 20.1 19.8
Glenmark Pharma GLEPHA 1060 1000 HOLD 29695.5 16.5 33.7 47.5 38.6 18.9 13.4 16.5 11.6 8.7 12.6 23.7 29.5 14.3 24.0 26.7
Indoco Remedies INDREM 342 385 BUY 3119.3 9.0 12.6 19.2 24.3 17.4 11.3 12.4 10.0 7.1 19.8 22.3 28.0 16.0 19.0 23.4
Ipca Laboratories IPCLAB 760 665 HOLD 9587.3 20.2 25.0 41.5 18.7 17.0 11.6 3.2 2.9 2.4 11.5 11.0 16.6 11.5 11.0 16.6
Jubilant Life Sciences VAMORG 313 168 HOLD 4506.1 -3.7 13.2 23.6 0.0 11.8 6.6 9.5 7.2 5.3 5.7 8.9 12.1 -2.4 8.2 13.4
Lupin LUPIN 1643 1885 BUY 76305.9 53.6 55.7 75.4 30.7 29.5 21.8 -0.4 0.8 0.2 31.1 29.2 30.4 25.3 22.8 23.6
Natco Pharma NATPHA 2471 2200 HOLD 8272.5 31.2 46.4 67.1 23.0 15.5 10.7 15.9 12.2 9.2 15.6 19.6 23.0 12.4 16.6 19.8
Sun Pharma SUNPHA 841.8 850 HOLD 202314.1 19.8 20.5 32.6 44.2 46.9 25.5 24.6 24.0 16.6 19.0 18.3 22.8 20.8 18.5 23.6
Torrent Pharma TORPHA 1494 1660 BUY 25248.0 44.4 94.4 79.0 32.0 15.0 18.0 13.6 5.1 6.6 19.8 44.9 30.3 30.2 44.4 29.0
Unichem Laboratories UNILAB 240.8 214 HOLD 2250.2 8.3 14.8 21.4 24.8 13.9 9.6 17.9 9.4 6.5 8.4 17.1 21.5 8.7 14.5 18.5
RoNW (%)Company
EPS (|) PE(x) EV/EBITDA (x) RoCE (%)

ICICI Securities Ltd. | Retail Equity Research
Page 28
RATING RATIONALE ICICIdirect.com endeavours to provide objective opinions and recommendations. ICICIdirect.com assigns ratings to its stocks according to their notional target price vs. current market price and then categorises them as Strong Buy, Buy, Hold and Sell. The performance horizon is two years unless specified and the notional target price is defined as the analysts' valuation for a stock. Strong Buy: >15%/20% for large caps/midcaps, respectively, with high conviction; Buy: >10%/15% for large caps/midcaps, respectively; Hold: Up to +/-10%; Sell: -10% or more;
Pankaj Pandey Head – Research [email protected]
ICICIdirect.com Research Desk, ICICI Securities Limited, 1st Floor, Akruti Trade Centre, Road No 7, MIDC, Andheri (East) Mumbai – 400 093

ICICI Securities Ltd. | Retail Equity Research
Page 29
ANALYST CERTIFICATION We /I, Siddhant Khandekar, CA INTER and Mitesh Shah, MS (finance), Nandan Kamat MBA Research Analysts, authors and the names subscribed to this report, hereby certify that all of the views expressed in this research report accurately reflect our views about the subject issuer(s) or securities. We also certify that no part of our compensation was, is, or will be directly or indirectly related to the specific recommendation(s) or view(s) in this report.
Terms & conditions and other disclosures: ICICI Securities Limited is a SEBI registered Research Analyst having registration no. INH000000990. ICICI Securities Limited (ICICI Securities) is a full-service, integrated investment banking and is, inter alia, engaged in the business of stock brokering and distribution of financial products. ICICI Securities is a wholly-owned subsidiary of ICICI Bank which is India’s largest private sector bank and has its various subsidiaries engaged in businesses of housing finance, asset management, life insurance, general insurance, venture capital fund management, etc. (“associates”), the details in respect of which are available on www.icicibank.com. ICICI Securities is one of the leading merchant bankers/ underwriters of securities and participate in virtually all securities trading markets in India. We and our associates might have investment banking and other business relationship with a significant percentage of companies covered by our Investment Research Department. ICICI Securities generally prohibits its analysts, persons reporting to analysts and their relatives from maintaining a financial interest in the securities or derivatives of any companies that the analysts cover. The information and opinions in this report have been prepared by ICICI Securities and are subject to change without any notice. The report and information contained herein is strictly confidential and meant solely for the selected recipient and may not be altered in any way, transmitted to, copied or distributed, in part or in whole, to any other person or to the media or reproduced in any form, without prior written consent of ICICI Securities. While we would endeavour to update the information herein on a reasonable basis, ICICI Securities is under no obligation to update or keep the information current. Also, there may be regulatory, compliance or other reasons that may prevent ICICI Securities from doing so. Non-rated securities indicate that rating on a particular security has been suspended temporarily and such suspension is in compliance with applicable regulations and/or ICICI Securities policies, in circumstances where ICICI Securities might be acting in an advisory capacity to this company, or in certain other circumstances. This report is based on information obtained from public sources and sources believed to be reliable, but no independent verification has been made nor is its accuracy or completeness guaranteed. This report and information herein is solely for informational purpose and shall not be used or considered as an offer document or solicitation of offer to buy or sell or subscribe for securities or other financial instruments. Though disseminated to all the customers simultaneously, not all customers may receive this report at the same time. ICICI Securities will not treat recipients as customers by virtue of their receiving this report. Nothing in this report constitutes investment, legal, accounting and tax advice or a representation that any investment or strategy is suitable or appropriate to your specific circumstances. The securities discussed and opinions expressed in this report may not be suitable for all investors, who must make their own investment decisions, based on their own investment objectives, financial positions and needs of specific recipient. This may not be taken in substitution for the exercise of independent judgment by any recipient. The recipient should independently evaluate the investment risks. The value and return on investment may vary because of changes in interest rates, foreign exchange rates or any other reason. ICICI Securities accepts no liabilities whatsoever for any loss or damage of any kind arising out of the use of this report. Past performance is not necessarily a guide to future performance. Investors are advised to see Risk Disclosure Document to understand the risks associated before investing in the securities markets. Actual results may differ materially from those set forth in projections. Forward-looking statements are not predictions and may be subject to change without notice. ICICI Securities or its associates might have managed or co-managed public offering of securities for the subject company or might have been mandated by the subject company for any other assignment in the past twelve months. ICICI Securities or its associates might have received any compensation from the companies mentioned in the report during the period preceding twelve months from the date of this report for services in respect of managing or co-managing public offerings, corporate finance, investment banking or merchant banking, brokerage services or other advisory service in a merger or specific transaction. ICICI Securities or its associates might have received any compensation for products or services other than investment banking or merchant banking or brokerage services from the companies mentioned in the report in the past twelve months. ICICI Securities encourages independence in research report preparation and strives to minimize conflict in preparation of research report. ICICI Securities or its analysts did not receive any compensation or other benefits from the companies mentioned in the report or third party in connection with preparation of the research report. Accordingly, neither ICICI Securities nor Research Analysts have any material conflict of interest at the time of publication of this report. It is confirmed that Siddhant Khandekar, CA INTER and Mitesh Shah, MS (finance), Nandan Kamat MBA, Research Analysts of this report have not received any compensation from the companies mentioned in the report in the preceding twelve months. Compensation of our Research Analysts is not based on any specific merchant banking, investment banking or brokerage service transactions. ICICI Securities or its subsidiaries collectively or Research Analysts do not own 1% or more of the equity securities of the Company mentioned in the report as of the last day of the month preceding the publication of the research report. Since associates of ICICI Securities are engaged in various financial service businesses, they might have financial interests or beneficial ownership in various companies including the subject company/companies mentioned in this report. It is confirmed that Siddhant Khandekar, CA INTER and Mitesh Shah, MS (finance), Nandan Kamat MBA, Research Analysts do not serve as an officer, director or employee of the companies mentioned in the report. ICICI Securities may have issued other reports that are inconsistent with and reach different conclusion from the information presented in this report. Neither the Research Analysts nor ICICI Securities have been engaged in market making activity for the companies mentioned in the report. We submit that no material disciplinary action has been taken on ICICI Securities by any Regulatory Authority impacting Equity Research Analysis activities. This report is not directed or intended for distribution to, or use by, any person or entity who is a citizen or resident of or located in any locality, state, country or other jurisdiction, where such distribution, publication, availability or use would be contrary to law, regulation or which would subject ICICI Securities and affiliates to any registration or licensing requirement within such jurisdiction. The securities described herein may or may not be eligible for sale in all jurisdictions or to certain category of investors. Persons in whose possession this document may come are required to inform themselves of and to observe such restriction.
Related Documents