The community structure, and ecology, in and around a Sabellaria alveolata biogenic reef. Abstract Cei-Bach is a semi sheltered bay within the Cardigan Bay Special Area of Conservation (SAC) in West Wales, and designated for its honeycomb worm (Sabellaria alveolata) biogenic reef habitat, which provides a biodiverse substratum on an otherwise scouring benthos. The study objectives were to make a rapid and effective assessment of the community structure and ecology with limited resources. GIS was used to measure the reef extent and environmental gradients for direct comparison with taxa response. With some 52 species of macro-epifauna identified, there was much noise in the data and challenges in identifying the key players shaping the community. Indirect ordination techniques of Cluster Analysis and Principal Components Analysis (PCA) were used with MVSP to resolve three clear community assemblages and their defining species. This enabled direct ordination with five environmental variables of shore position, stability, salinity, turbulence and submersion through Canonical Correspondence Analysis (CCA); whilst mitigating the characteristic “horseshoe” effect when resolving noisy data, or rare taxa, with Correspondence Analysis. The results showed significant heterogeneity in the community structure and higher biodiversity within the reef extent. The reef was effectively “framed” by limiting factors of transition from an intertidal environment to the North; desiccation to the south, and east; excessive seston and salinity reducing inundation of freshwater to the west, where the honeycomb worm was competitively excluded by functioning guilds of Ulva sp. The study found that the assemblages were defined most strongly by shore position, substrate stability and salinity, and highlighted the challenges of effective environmental variable selection in direct ordination. SID 0917866 Undergraduate Project Abstract

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

The community structure, and ecology, in and around a
Sabellaria alveolata biogenic reef.
Abstract
Cei-Bach is a semi sheltered bay within the Cardigan Bay Special Area of Conservation
(SAC) in West Wales, and designated for its honeycomb worm (Sabellaria alveolata)
biogenic reef habitat, which provides a biodiverse substratum on an otherwise scouring
benthos. The study objectives were to make a rapid and effective assessment of the
community structure and ecology with limited resources. GIS was used to measure the reef
extent and environmental gradients for direct comparison with taxa response.
With some 52 species of macro-epifauna identified, there was much noise in the data and
challenges in identifying the key players shaping the community. Indirect ordination
techniques of Cluster Analysis and Principal Components Analysis (PCA) were used with
MVSP to resolve three clear community assemblages and their defining species. This
enabled direct ordination with five environmental variables of shore position, stability,
salinity, turbulence and submersion through Canonical Correspondence Analysis (CCA);
whilst mitigating the characteristic “horseshoe” effect when resolving noisy data, or rare taxa,
with Correspondence Analysis.
The results showed significant heterogeneity in the community structure and higher
biodiversity within the reef extent. The reef was effectively “framed” by limiting factors of
transition from an intertidal environment to the North; desiccation to the south, and east;
excessive seston and salinity reducing inundation of freshwater to the west, where the
honeycomb worm was competitively excluded by functioning guilds of Ulva sp. The study
found that the assemblages were defined most strongly by shore position, substrate stability
and salinity, and highlighted the challenges of effective environmental variable selection in
direct ordination.
SID 0917866 Undergraduate Project Abstract

TITLE
The community structure, and ecology, in and around a Sabellaria alveolata biogenic reef.
By 0917866, B.Sc. (Hons.) Marine Biology, Ecology and Conservation,
Anglia Ruskin University.
TABLE OF CONTENTS
Page
2. Introduction
5. Methods and results
9. Ordination methods flow chart
40. Discussion
46. Conclusions
47. Critique
48. Acknowledgements
48. References
57. Appendices
1. Species list
2. Raw data summary
3. CD ROM – Project data
1

INTRODUCTION
The objectives of this study are to gain an understanding of the defining communities within
a distinct coastal ecosystem, and how biotic and abiotic influences shape these. The study
focuses on the polychaete, Sabellaria alveolata (Linnaeus, 1767), at the intertidal area of Cei
Bach in Cardigan Bay, West Wales (Fig. 1). S. alveolata, the honeycomb worm, is an
important ecosystem engineer, and UK Biodiversity Action Plan (BAP) species, as it creates
littoral biogenic reef substrate which facilitates increased biodiversity (Maddock, 2008).
Cei – Bach is a semi-sheltered sandy bay with deposited terminal moraine, left by retreating
glaciers some 11000 -18000 years ago (Crampton, 1965; CCW, 2013). The resultant
boulder and cobble shore area, at the interface of the bay and River Llethi, provides a refuge
of stability for settling epifauna and flora within an otherwise moving and scouring
environment (Geograph, 2013; Little et al, 2009). This S. alveolata habitat is part of the
Special Area of Conservation (SAC) designation and towards the northern limit of the
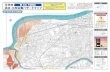
polychaetes’ geographic range (Fig. 1; Desroy et al, 2011; Moore, 2009).
N
Cardigan Bay SAC
Cei Bach
(Image: Moore, 2009)Image: NBN, 2012
Figure 1. The location of the study area at Cei Bach, within the Cardigan Bay Special Area
of Conservation designation and, (right) UK distribution of S. alveolata (Cardigan Bay SAC,
2008; NBN, 2012).
2

Sabellariidae recruit sand grains through mucus secretion to create thick walled tubes, at
right angles to the substratum, which aggregate and cement to each other like cells in a
honeycomb (Fig. 2; Ruppert et al, 2004).
Figure 2. S. alveolata reef formation (left) and uncased worm (right; Image: Moore, 2009)
S. alveolata uses ciliated radioles to sort the sand grains into suitable sizes for tube building
and suspension feeding (Hayward & Ryland, 1995). Unsuitable particles are rejected, and
particle size, availability and distribution affect clearance and particulate retention efficiency
(Dubois et al, 2003 & 2005). Fresh water outfalls alter the hydro-sedimentary system which
affects the duration of seston suspension, particle distribution, feeding and settlement
patterns of S. alveolata and other dispersing taxa (Aller & Cochran, 1976; Aller et al, 1980;
Dubois et al, 2009; Pawlik, 1988).
My study examines the influence of autochthonous, endogenous and allochthonous
environmental factors on the successful settlement distribution of S. alveolata, and taxa
assemblage patterns within the terminal moraine (Little et al, 2009). The theory of the study
animal, and other assemblage defining taxa, such as Mytilus, as a foundation species is
considered, within the distinct community assemblages (Albrecht, 1998; Little et al, 2009). I
further theorise that assemblage influencing factors will include, shore position and
3

desiccation, substrate stability, salinity, turbulence, submersion, particle deposition and size,
competition and facilitation (Dubois et al, 2006; Pawlik, 1988). Anthropogenic pollution will
be considered; specifically River Llethi eutrophication from agricultural run-off and pollution
risk from the Llanina long sea treated sewage outfall emanating from the study area (Dodds,
2002 & 2006; EAW, 2012).
The hypotheses are that the distribution of S. alveolata will be heterogeneous within the
sample frame; that distinct community assemblages will be defined by key taxa and
associated definable areas (zones); I also hypothesise that there will be distinct taxa and
assemblage response to environmental and endogenous factors.
As budget, equipment and human resources were limited, simple GIS methods were
combined with traditional ecological census techniques, and a number of proxies adopted to
measure environmental variables. Ecological and environmental data were collected over a
six week period in May and June 2012. Basic descriptive statistics were calculated before
analysis with indirect and direct ordination techniques to infer community structure and
response of the key taxa and assemblages, to environmental factors. The key ordination
outcomes were tested for statistical significance (Anderson, 2001). The methods and results
are presented in a combined format and a flow diagram is presented on page 9,
summarising the ordination process and methods, prior to significance testing (Table 2;
Ridley et al, 2010).
I discuss the distribution of the community assemblages within the boulder and cobble
intertidal area, before focusing on the three assemblage defining taxa, S. alveolata, Mytilus
edulis and Enteromorpha intestinalis. The ecology of these species and their response to the
most correlated endogenous and exogenous environmental factors, interspecific
competition, association and facilitation is then discussed.
4

METHODS AND RESULTS
A pre-site survey visit was made in March 2012 to consider experimental design and
adaptations from methodologies in Joint Nature Conservation Committee (JNCC) guidance
and the Countryside Council for Wales (CCW) commissioned intertidal surveys (Allen et al,
2002; Boyes & Allen, 2008; Davies, 2001a,b; Davies et al, 2001; JNCC, 2004; Moore, 2009).
Methods were designed to enable one surveyor, with a safety observer, to commence top-
down surveys two hours before low tides. Lower shore surveying effort was enabled by
synchronisation of lower shore sampling with Spring tides. Best practice was adopted from
the JNCC handbook for monitoring of Annex 1 habitats, to ensure non-invasive and non-
destructive methods, and no core samples were taken (Davies, 2001a; Davies et al, 2001;
JNCC, 2004 & 2012; UKMPA Centre, 2012).
Five pre-survey visits were made to ensure consistent identification of fauna and flora and
the consistent assessment of live S. alveolata worm numbers, adopting the “lipped porch
diagnosis” method from Boyes & Allen p. 21 (2008; Moore, 2009). The extent of the S.
alveolata reef was assessed in accordance with Hendrick & Foster-Smith (2006) criteria for
“reefiness”, from which a clear ecotone could be identified as the reef boundary (Park,
2008). The reef boundary (Fig. 3; Zone SQ) was tracked with Garmin Map 62 handheld
device, accurate to 10m with 95% confidence, with map datum WGS84 setting (Garmin,
2012; GPS Information, 2013). The zones neighbouring the reef in the boulder and cobble
intertidal shore area, were tracked similarly; Zone NQ to the west and Zone MQ to the south-
east (Fig. 3).
Tracks and waypoints were downloaded, with GPS Utility, into Quantum GIS and zipped to
polygons for each reef zone (GPS Utility, 2012; QGIS, 2010). The sample frame was
groundtruthed, with Cei-Bach landmarks, on OS Map TL38 downloaded from Digimap (2012;
Fig. 3; QGIS, 2010).
5

Moore (2009) made criticism of the original Boyes & Allen (2008) survey which used 2 x 2m
quadrats and a very low sampling frequency. Random stratified sampling was also
considered for lower, upper and middle shore zones but rejected in favour of random cluster
sampling, as GPS logging of each random quadrat allows stratification of the data to be
reconsidered after the survey (Bolam et al, 2011). By using gridded 0.5m x 0.5m quadrats
with 25 equal cells, sampling frequency, accuracy and statistical power was increased
(Ausden & Drake, 2006; Kent, 2012; Moore, 2009).
Moore (2009), randomly located quadrat sites along transects by way of Microsoft Excel
random numbers and “closed eyes” in placing the quadrats upon the stratum as no other
cheap and simple method was available. Whilst GPS locations of each sample were logged
by Moore (2009), it was not effective to copy the same locations as the extent and location of
the reef is dynamic (Lancaster & Savage, 2008).
To reduce potential bias, Quantum was used to generate random samples in the zone
polygons; 12 in Zone MQ, 24 in Zone SQ and 22 in Zone NQ, in accordance with the
sampling effort guidance of Davies (2001a); based on the reef extent survey in May 2007
(Davies et al, 2001; Greenwood & Robinson, 2006; JNCC, 2004 & 2012; QGIS, 2010). The
random generated waypoints were uploaded to the GPS unit and the “Find” function used to
locate each random sample point, within the accuracy tolerance of the unit. To maximise
precision, the quadrat was placed touching the surveyors toes immediately upon “0m”
distance to sample appearing on the unit, and the actual sample waypoint captured
(Greenwood & Robinson, 2006).
A data card was designed to enable consistent collection and logical MVSP, Excel and
SPSS data input. The number of live S. alveolata worms, identification and
presence/absence of epifauna and flora, and coverage (%) of S. alveolata, algae, Mytilus,
Cirrepedia and standing water (submersion), were recorded. Dominant substrate type was
categorised as sand, granule, pebble, cobble or boulder as a measure of environmental
6

substrate stability (Wentworth scale, in Little, 2000). One sand sample was collected when
available, at every other quadrat and stored in zipped labelled polythene bags for laboratory
data analysis of sediment size and sorting (Wentworth, 1922).
GPS tracks were logged for; the centre of the River Llethi channel, as a proxy for salinity i.e.
increasing distance from freshwater influence, and the centre of the high water jetsam
strandline as a datum for shore position. The waypoint was logged at a substantial boulder
marking the low water extent of the freshwater channel outflow as a proxy for turbulence
influence (Fig. 3 (a, b, c); Aller & Cochran, 1976; Aller et al, 1980; Dubois et al, 2009). The
Quantum “ruler” function was used to measure euclidean distance from the tracks and
waypoint to each actual sample waypoint (QGIS, 2010).
N
a
b
b
MQ
SQ
NQ
KeyNQ: Western Zone SQ: Sabellaria reef extent zone MQ: Mytilus zonea. Centre of freshwater channelb. Centre of high water markc. Boulder datum marking extent of freshwater channel at low water
11
3
3
c
Cei Bach
Figure 3. Map of the Cei Bach intertidal boulder and cobble shore area indicating the S.
alveolata reef zone (SQ), two neighbouring zones (NQ & MQ), and three environmental
factor markers (a,b,c; Image: QGIS, 2010).
The community data matrix was exported from Microsoft Excel 2007, to MVSP (Kovach,
1999) and SPSS (IBM, 2011). A species list is attached and the raw data summary shows
rarity and clumping amongst many of the 52 taxa present (Appendix 1 & 2).
7

Microsoft Excel and SPSS were used to calculate the estimated coverage (%) of S.
alveolata, algae, M. edulis, and Cirrepedia in the three intertidal zones, which is illustrated in
figure 4 and detailed in table 1. Zone MQ has the highest proportion of bare shore and the
least in the Sabellaria zone (SQ). There is low algal coverage in the Mytilus zone (MQ),
highest coverage in the Sabellaria zone (SQ) and intermediate coverage in Zone NQ.
Mytilus is absent from the Sabellaria zone (SQ) and Sabellaria rare in the Mytilus zone (MQ);
with Zone NQ, intermediate for both taxa (Table 1). There is Cirrepedia coverage in Zone
NQ but rarity in Zone MQ & SQ (Fig. 4; Table 1).
N
a
b
b
MQ
SQNQ
Key1. S. alveolata 2. Algae 3. M. edulis 4. Cirrepedia 5. Bare shore
a. Centre of freshwater channel b. Centre of high water markc. Boulder datum marking extent of freshwater channel at low water
NQ: Western Zone SQ: Sabellaria reef zone MQ: Mytilus zone
5
5
51
1
3
2
2
3
4
4 2c
Figure 4. Map of the Cei Bach intertidal boulder and cobble shore area illustrating substrate
coverage (%) by S. alveolata, algae, M. edulis and Cirrepedia (Table 1; Image: QGIS, 2010).
Table 1. Coverage (%) of the boulder and cobble shore intertidal zones, by S. alveolata,
algae, M. edulis and Cirrepedia (Fig. 4; standard error in parenthesis).
Zone S. alveolata Algae Mytilus Cirrepedia Bare shore nMQ 0.02 (+/‐ 0.01) 5.12 (+/‐ 0.98) 11.84 (+/‐ 0.99) 0.48 (+/‐ 0.10) 82.54 300SQ 30.93 (+/‐ 1.47) 45.46 (+/‐ 1.68) 0 0.87 (+/‐ 0.16) 22.74 600NQ 4.97 (+/‐ 0.84) 19.79 (+/‐ 1.55) 4.86 (+/‐ 0.72) 7.67 (+/‐ 0.83) 62.71 550
1450
8

The process and methods for subsequent ordinations are summarised in Table 2 (Ridley et
al, 2010).
9

Table 2. Summary of ordination methods
Key: H20 = standing water
All taxa + H20 presence/absence (binary) MVSP
Cluster UPGMASorensen’s
Taxa
Analysis PCAKaisers
Figs. 6 & 7Output
Distillation to 11 taxa + H2OTaxa
PCA KaisersMVSP Grouping Function (by zones)Analysis
Fig. 8Output
Distillation to 7 taxa + H2OTaxa
Cluster UPGMASorensen’s
PCAKaisers
Analysis
Cluster UPGMASimple Matching
Analysis
Figs. 9 & 10 (a & b)Output
7 taxa - H2O RemovedTaxa
Cluster UPGMASimple MatchingAnalysis
Fig. 11 (a & b)Output
Analysis
PCAKaisers
Direct Ordination – CCA (MVSP)
11 key taxa Excl. H20 absence/presence binaryTaxa
EnvironmentalVariables
CCA Hills algorithm Scaling by SpeciesAnalysis
Figs. 12, 13 (a & b), 14 (a, b, c); Table 4Output
Analysis CCA MVSP – Grouping function by zone (11sp.)
Output Figs. 15 & 16
Significance testing
Analysis
All Log10 transformed: 1. Shore position, 2.Stability, 3. Salinity, 4. Turbulence, 5. Submersion
10

Live S. alveolata density was highly skewed overall, rare in Zone MQ and only occasional in
Zone NQ: Zone median densities were (worms per m2); All zones, 0, 0 – 16, n = 1450: Zone
MQ, 0, 0 – 0, n = 300: Zone SQ, 24, 0 – 72, n = 600: Zone NQ, 0, 0 – 0, n = 550. SPSS was
used to test S. alveolata density difference between intertidal zones; confirming significantly
higher density in Zone SQ, with many extremes and outliers: Kruskal-Wallis X22 = 461.75, n1
= 300, n2 = 600, n3 = 550, P < 0.001 (Figs. 4 & 5).
S. alveo
lata den
sity per m
2
n = 1450
Figure 5. Box plot showing the significantly higher density of live S. alveolata worms in Zone
SQ, as illustrated in figure 4 (Kruskal-Wallis test).
11

MVSP was used to undertake indirect ordination through cluster analysis and Principal
Components Analysis (PCA) to infer community composition. Direct ordination by Canonical
Correspondence Analysis (CCA) was used to compare how well the extractions are
explained by environmental factors, “species-environment correlation” (Palmer, 1993; ter
Braak, 1986 & 1994).These techniques were used successfully to explain biological
assemblages and distribution of taxa, in aquatic communities, along environmental gradients
by ter Braak & Verdonschot (1995).
All samples within the reef (SQ), abutting zone to the west (NQ) and abutting zone to the
southeast (MQ) were tested by taxa and submersion proxy presence for inference of
community structure (Fig. 3; Lesperance, 1990). Cluster analysis was utilised with UPGMA
logarithm and Sorensen’s coefficient, in view of clumping and taxa rarity within the dataset
(App. 1; Krebs, 1998). Figure 6 illustrates Sorensen’s coefficient extracting two distinct
clusters of 13 taxa (A & B), nesting the 3 strongest clusters; S. alveolata and Fucus
vesiculosus (Ss 0.38), M. edulis and Littorina littorea (Ss 0.38), E. intestinalis and standing
water (Ss 0.35). Cluster C infers rare or insignificant taxa (Fig. 6; App. 1).
12

KEY
N1, N2, N3 = Cluster no
des
A = Cluster group
of
G. umbilicalis, C. m
ontagu
i, Po
rifera, F. spiralis; ne
sting
N2, L. littorea & M
. edu
lis
B= Cluster grou
p of
C. ru
pestris, C. officiana
lis, O
. pinn
atifida
& Ulva; nestin
g N3, E. intestina
lis & stand
ing
water and
N1, S. a
lveo
lata & F. vesiculosus
C= Cluster grou
p of
Rare or po
tentially insignificant taxa
(App
. 1)
Figu
re6.
Clu
ster
ana
lysi
s de
ndog
ram
of a
ll 52
sam
pled
taxa
indi
catin
g gr
oupi
ng o
f rar
e or
insi
gnifi
cant
taxa
(C) a
nd 1
3 ta
xa in
two
pote
ntia
lly
sign
ifica
nt g
roup
s; A
(6 s
p.) &
B (7
sp.
)
13

C
A B
n = 14
50N1 N2
N3N3
14

The cluster dendogram was copied to Microsoft Word and compared with the data output of
PCA to consider removal of insignificant or rare taxa and/or grouping of related taxa to
reduce noise (Gauch, 1982; Palmer, n.d.). PCA (Kaisers rule) indicated 41 insignificant taxa,
now including Porifera and Fucus spiralis, and distillation to 11 significant taxa driving
39.72% of the variance extracted by the first three axes, 1 (16.7%), 2 (12.0%) & 3 (11.0%).
The three strongest component loadings of axis 1 were examined to assess relative species
importance in the extracted axis 1 (Kovach, 2011). The component loadings were compared
with the cluster results and dendogram (Fig. 6). M. edulis (-0.52), E. intestinalis (0.46) and S.
alveolata (0.34), were strongest and congruent with the cluster groupings and used to define
the assemblage groupings A, B & C in figure 7 & subsequent figs. The insignificant taxa are
distributed around the cross hairs of axes 1 and 2, with axis 1 acting as a vector separating
groups B & C from group A and axis 2; group B from C (Fig. 7).
Me
Pd
EpCm
Gw
H20
Sa
Bw
KEY
Me Mytilus edulisEp Littorina littoreaCm Chthamalus montaguiH2O Standing water @ low tideGw Enteromorpha intestinalisPd Osmundia pinnatifidaBw Fucus vesiculosusSa Sabellaria alveolataPt Gibbula umbilicalisCo Corallina officinalisCr Cladophera rupestrisUl Ulva lactuca
Pt
Cr Ul
Co
C
B
A
Assemblage Groupings
CBA
Enteromorpha group
Mytilus group
Sabellaria group
n = 1450
Figure 7. All sampled taxa PCA indicating key groupings of 11 significant taxa and standing
water environmental factor with 39.72% of the variance extracted in axes 1 (16.7%); 2
(12.0%); 3 (11.0%). M. edulis, E. intestinalis and S. alveolata can be seen clearly as furthest
neighbours in the scatter plot, due to the highest variance component loadings, and are
used to define the three species assemblages, A, B & C respectively.
15

Porifera and F. spiralis were removed to refine PCA analysis with 49.81% of variance now
extracted by the first three axes, 1 (21.0%), 2 (15.2%) & 3 (13.6%) and M. edulis, E.
intestinalis & S. alveolata axis 1 component loadings of -0.53, 0.47 & 0.34, respectively (Fig.
8; Kovach, 2011).
The samples were grouped by the three study zones (MQ, SQ, NQ) using MVSP grouping
function and combined with euclidean bi-plots to test different taxa and grouping response
and vectoring with the study zones (Duigan & Kovach, 1991; Kovach, 2011). Axis 1 acted as
a presence vector confirming taxa group A (M. edulis, L. littorea and Chthamalus montagui)
to the left of the bi-plot, where Mytilus zone MQ dominates, and groups B & C to the right
where Sabellaria zone SQ dominates; Taxa / group response to the western area NQ is not
clear and considered later in a test of association (Fig. 8; ter Braak, 1987 & 1994).
Figure 8 illustrates short vectors and the lower significance of Gibbula umbilicalis,
Cladophera rupestris, Corallina officinalis and Ulva in driving variance and these were
removed for subsequent and final refinement of PCA analysis with and without the
submersion proxy (Palmer, n.d.; Pusceddu et al, 2011). Of the significant responses, E.
intestinalis appears diametrically opposed to L. littorea and C. montagui, and S. alveolata
very directionally similar to Osmundia pinnatifida and F. vesiculosus, which are identically
orientated.
16

X= (M
Q)M
ytilus zone
= (SQ)Sab
ellaria zone
= (NQ) Zon
e to W
est
A = M
ytilusgrou
pB = Enteromorph
agrou
pC = Sabellariagrou
p
Figu
re 8. PCA
Euclidean bi‐plot of 11 significant taxa and standing
water, ind
icating three key species assemblages (A,B,C) and
sho
wing
Axis 1 actin
g as a presence vector fo
r the samples of zon
e SQ
to th
e right and zone
MQ to
the left, w
ith m
ore evenly distributed
zon
e NQ.
49.81%
of the
variance was extracted
in the first th
ree axes; 1
(21%
); 2
(15.2%
); 3 (1
3.6%
). The short environm
ental vector arrows illustrate
low taxa respo
nse of G. umbilicalis, C. rup
estris, C. officinalis & Ulva,
which were removed
for subseque
nt and
final cluster and
PCA
analysis.
17

A
C
B
n = 14
50
18

To further resolve the whole community dendogram, the seven taxa contributing most
strongly to the PCA assemblage directional vectors were used in cluster analysis with
Sorensen’s coefficient (Figs. 6 & 9). Fig. 9 illustrates the three clusters (A (Ss 0.32 ); B (Ss
0.35 ); C (Ss 0.27)) & extracted, with nested clusters within A & C. Group A; L. littorea & M.
edulis (Ss0.38) and Group C; S. alveolata & F. vesiculosus (Ss 0.39).
A
B
C
N4
N2
N1
N5
N3
n = 1450
N1 – 5 = Cluster nodes
Figure 9. Cluster analysis and suggested assemblages (A,B,C) of seven most significant
taxa and submersion proxy (H2O; Sorensen’s coefficient).
To understand taxa response, inferred by absence, to environmental factors such as salinity,
desiccation, standing water (submersion), or competitive exclusion, I used simple matching
coefficient cluster analysis (Little et al, 2009). Jaccards coefficient was rejected as the taxa
are not evenly distributed through the dataset (Lesperance, 1990). Simple matching
coefficient shows strong confidences in splitting group A (Sm 0.69) from B (Sm 0.68) & C (Sm
0.75), linking them by the submersion proxy, A(Sm 0.62) & B(Sm 0.63) and inferring nested
clusters of C. montagui & M. edulis (E, Sm 0.74) within A and O. pinnatifida and F.
vesiculosus (D, Sm 0.83) within C (Fig. 10(a)).
19

The simple matching clusters are corroborated by PCA with 59.0% of variance extracted in
the first three axes, 1 (24.6%), 2 (18.5%) & 3 (15.9%), illustrated by groups A, B & C in figure
10(b).
(a)
n = 1450A
B
E
C
D
N4N3
N5
N2 N1
N7N6
KEY: N1 – 7 Cluster nodes
KEY:Me Mytilus edulisEp Littorina littoreaCm Chthamalus montaguiH2O Standing water @ low tideGw Enteromorpha intestinalisPd Osmundia pinnatifidaBw Fucus vesiculosusSa Sabellaria alveolata
Me
Pd
Gw
H20
Cm Ep
Sa
Bw
AB
C
Assemblage Groupings
Mytilus group
Enteromorpha group
Sabellaria group
ABC
n = 1450(b)
20

Figure 10. Cluster and corroborative PCA analysis of seven most significant taxa and
submersion proxy, with 59% of the variance extracted in axes 1 (24.6%); 2 (18.5%); 3
(15.9%).
With the submersion proxy removed, the final taxa assemblage cluster and PCA analyses
show high congruence (Fig. 11(a & b)). Cluster analysis shows, group A M. edulis similarity
with C. montagui (Sm 0.74) and L. littorea (Sm 0.69); group B O. pinnatifida and F.
vesiculosus similarity (Sm 0.83) and S. alveolata (Sm 0.75), connected to group C (E.
intestinalis) (Sm 0.68; Fig. 11(a)). PCA corroborates groups A, B & C with 63.5% of variance
extracted in the first three axes, 1 (28.4%), 2 (20.4%) & 3 (14.7%) and the three group
defining taxa contributing most strongly to axis 1 with component loadings of -0.54, 0.46 &
0.44 for M. edulis, E. intestinalis and S. alveolata respectively (Fig. 11(b)). Axis 1 acts as a
vector separating groups B & C from A, and axis 2, sub group Ai & C from E. intestinalis and
M. edulis (Fig. 11(b)).
21

n = 1450
KEY: N1, N2, N3, N4, N5 = cluster nodesN1N2N4
N3N5
(a)
A
C
D
B
E
n = 1450
Bw
Me
Cm
Ep
Pd
Gw
Sa
KEY:Me Mytilus edulisEp Littorina littoreaCm Chthamalus montaguiGw Enteromorpha intestinalisPd Osmundia pinnatifidaBw Fucus vesiculosusSa Sabellaria alveolata
Assemblage Groupings
ABC
Mytilus group (incl. Ai nested group)
Enteromorpha group
Sabellaria group
A
B
C
Ai
(b) Figure 11. Cluster dendogram and PCA scatter plot of seven most significant taxa and
assemblage groupings, with 63.5% of the variance extracted in axes 1 (28.4%); 2 (20.4%); 3
(14.7%). The submersion proxy was removed in this final cluster analysis and PCA.
22

CCA is suitable for contingency data such as taxa absence/presence and was used to
directly identify taxa response to the environmental variables measured in the study
(Kovach, 2011; Palmer, 1993; ter Braak, 1986). By including only the 11 significant taxa
identified by PCA as driving variance, the Correspondence Analysis (CA) tendency of
distortion by outliers is avoided and CCA “downweighting” of rare species unnecessary (Fig.
7; Gauch, 1982; Kovach, 2011; ter Braak, 1994). MVSP CCA is not affected by the instability
problems described by Oksanen & Minchin (1997; Kovach, 2011).
Hills algorithm and scaling by species was used in each CCA (ter Braak, 1986 & 1994).
Substrate type data was collected in nominal classes drawn from the Wentworth scale (in
Little, 2000), as a measure of stability. For CCA analysis, these nominal classes were
changed to the lowest size value for each class and log transformed, in view of the non-
linear nature of the classes, as discussed by Palmer (1993; Table 3). As species response
curves could be affected by non-transformation of the other environmental data, all
environmental variables were Log transformed using Microsoft Excel Log10 function (Palmer,
1993). Where standing water coverage was zero %; 0.001% was substituted to avoid infinity
values.
Table 3. Log transformed values of Wentworth scale particle classes
Nominal Value Min. size (mm) Log 10Boulder 256 2.41Cobble 64 1.81Pebble 4 0.60Granules 2 0.30Sand 0.063 ‐1.20
Eigenvalues (Ev) are stated for the axes to indicate ordination quality and the variance in the
taxa presence data; cumulative constrained percentage (Cc) to indicate how much of the
variance is explained by the constraining environmental variables; and
species/environmental correlation (Se r) as a measure of how well the environmental
variables explain the taxa community composition (ter Braak, 1986, 1994; ter Braak &
Verdonschot, 1995).
23

Correlations with the ordination axes of species or environmental variables, are not
published in tables or text as they can be derived from the bi-plot figures (head of the arrow
for environmental variables) e.g. Shore position (HW; 1) in fig. 12 is correlated -1.3 with axis
1 and +0.3 with axis 2, which derives the arrow length from source, at the cross hairs of axes
1 and 2 (Leps & Smilauer, 2003; ter Braak, 1994).
The MVSP graphing function was used to create bi-plots of the CCA which were illustrated in
Microsoft PowerPoint to consider corroboration of cluster and PCA assemblages and
defining taxa. Environmental variable arrows were given conspicuous extensions and
perpendicular lines drawn from taxa centroids to illustrate inferred taxa response ranking (ter
Braak, 1987). To avoid overcrowding in interpretation, separate illustrations of the bi-plots
were illustrated for each key environmental variable (Snoeijs & Prentice, 1989).
Five environmental variables were considered, with the 11 taxa; 1. Shore position and
associated desiccation (HW) i.e. distance from high water; 2. Stability (Substrate) i.e. macro-
particle size class (Table 3); 3. Salinity proxy (FW) i.e. distance from centre of freshwater
channel; 4. Turbulence proxy i.e. low water point of freshwater channel; 5. Submersion (SW
H20%) i.e. coverage by standing water at low tide (Figs. 11 & 12). Sand particle size was not
included in CCA analysis as an environmental factor, as sand samples were not collected, or
available, from all quadrat samples. Sand grain size is considered later.
The CCA direct ordination shows 68% of variance in the taxa presence data extracted in the
first two axes and supports the refined cluster and PCA extracted community assemblages
(Figs. 7 & 11): Ev; Axes 1 (0.53), 2 (0.15). 77.6% of variance was explained by
environmental constraint: Cc; Axes 1 (60.5), 2 (17.1; Fig. 12). Axis 1 acts strongly to explain
taxa community composition and the separation of groups B & C from A, by shore position
(1; HW) and relative stability (3): Se r; Axes 1 (0.83), 2 (0.55), (Fig. 12; ter Braak 1994; ter
Braak & Verdonschot, 1995).
24

CCA axis 2 correlates more weakly with species assemblage but vectors environmental
variance in the proxies for increasing salinity (4) and reducing turbulence (5; Fig. 12). Ulva is
associated with group B (E. intestinalis) and C. officinalis with submersion (2) and group C
(S. alveolata) in axis 2, which is a transposition from their weak axis 2 vectoring in PCA &
cluster analysis (Figs. 7 & 12).
Cm
Ul
BwPd
Gw
Sa
Cr
CoPt
MeEp
n = 1235Axis 1 r2 = 0.69Axis 2 r2 = 0.30
1
54
3
2
AB
C
KEYMe Mytilus edulisEp Littorina littoreaCm Chthamalus montaguiGw Enteromorpha intestinalisPd Osmundia pinnatifidaBw Fucus vesiculosusSa Sabellaria alveolataPt Gibbula umbilicalisCo Corallina officinalisCr Cladophera rupestrisUl Ulva lactuca
Assemblage groupings A = Mytilus group
B = Enteromorpha group
C = Sabellaria group
Figure 12. CCA bi-plot of 11 key taxa, their group assemblages (A,B,C), and response to
environmental factors of 1. (HW) Gradient towards low water 2. (SW) Gradient of increased
submersion, 3. (Substrate) Gradient towards increased abiotic stability, 4. (FW) Gradient
towards increased salinity and, 5. (Bldr) Gradient towards reduced turbulence / mixing.
Variance is extracted 68% in the first two axes; 1 (53%); 2 (15%) and 77.6% explained by
environmental constraint; 1 (60.5%); 2 (17.1).
25

Figure 13 illustrates the ranking of taxa response to environmental variables of, (a) gradient
from high water to low water (position on shore) and, (b) gradient from unstable to stable
available abiotic substrate; vectored most strongly by axis 1 variance extraction.
Cm
Ul
BwPd
Gw
Sa
Cr
CoPt
MeEpKEY
Me Mytilus edulisEp Littorina littoreaCm Chthamalus montaguiGw Enteromorpha intestinalisPd Osmundia pinnatifidaBw Fucus vesiculosusSa Sabellaria alveolataPt Gibbula umbilicalisCo Corallina officinalisCr Cladophera rupestrisUl Ulva lactuca
n = 1235Axis 1 r2 = 0.69Axis 2 r2 = 0.30
1
54
3
2
Vector extension illustrations
Perpendicular to environmental vector
Low Water
High Water
(a) Shore Position
Cm
Ul
BwPd
Gw
Sa
Cr
CoPt
MeEp
1
54
3
2
Vector extension illustrations
Perpendicular to environmental vector
(b) Stability
Axis 2
Unstable
Stable
n = 1235Axis 1 r2 = 0.69Axis 2 r2 = 0.30
Figure 13. Ranking of taxa response to environmental variables of, (a) Shore position and
associated desiccation gradient from high to low water and, (b) Stability gradient from
unstable to stable abiotic substrate. Axis 1 is acting as the key vector with 53% variance
extracted and 15% in axis 2. 77.6% of variance is explained by environmental constraint,
extracted in the first 2 axes; 1 (60.5%); 2 (17.1%).
26

Figure 14 illustrates the CCA ranking of taxa response to the environmental gradients of; (a)
submersion, vectored by axis 1 & 2 and, (b) salinity and, (c) turbulence / mixing, both
vectored by axis 2.
Cm
Ul
BwPd
Gw
Sa
Cr
CoPt
MeEp
Vector extension illustrations
Perpendicular to environmental vector
(a) SubmersionAxis 2
Submerged
Dessicatedn = 1235Axis 1 r2 = 0.69Axis 2 r2 = 0.30
Cm
Ul
BwPd
Gw
Sa
Cr
CoPt
MeEp
Vector extension illustrations
Perpendicular to environmental vector
(b) Salinity
Axis 2
Seawater salinity
Freshwater n = 1235Axis 1 r2 = 0.69Axis 2 r2 = 0.30
Cm
Ul
BwPd
Gw
Sa
Cr
CoPt
MeEp
Vector extension illustrations
Perpendicular to environmental vector
(c) Turbulence / mixing
Axis 2
LessMixed by river
Turbulent / Mixed n = 1235
Axis 1 r2 = 0.69Axis 2 r2 = 0.30
Figure 14. CCA ranking of taxa response to environmental variables (a) submersion,
vectored by axis 1 and 2, (b) salinity proxy and, (c) turbulence / mixing proxy, both vectored
by the more weakly correlated axis 2. Variance is extracted 68% by axes 1 and 2; 1 (53%); 2
27

(15%) and 77.6% explained by environmental constraint; 1 (60.5%); 2 (17.1%).
28

The CCA taxa response rankings are summarised in table 4.
Table 4. Summary CCA ranking of taxa response to environmental variables, shown in
figure 13 & 14.
Axis 1 Axis 2
Rank From High Water Group Stable Group Dessicated Group Freshwater Group Mixed GroupTowards Low Water Unstable Submersed Saline Unmixed
1 M. edulis A M. edulis A M. edulis A M. edulis A M. edulis A2 C. montagui A C. montagui A G. umbilicalis A C. montagui A C. montagui A3 G. umbilicalis A G. umbilicalus A C. montagui A G. umbilicalis A L.littorea A4 L. littorea A L. littorea A L.littorea A L. littorea A G. umbilicalis A5 C. officinalis C C. officinalis C C. officinalis C C. officinalis C E. intestinalis B6 S. alveolata C S. alveolata C S. alveolata C S. alveolata C S. alveolata C7 E. intestinalis B E. intestinalis B E. intestinalis B E. intestinalis B Ulva B8 O. pinnatifida C O. pinnatifida C C. rupestris C O. pinnatifida C O. pinnatifida C9 F. vesiculosus C F. vesiculosus C F. vesiculosus C F. vesiculosus C F. vesiculocus C10 C. rupestris C C. rupestris C O. pinnatifida C C. rupestris C C. officinalis C11 Ulva B Ulva B Ulva B Ulva B C. rupestris C
Fig. PPP (a) PPP (b) NNN (a) NNN (b) NNN (c)= DIFFERENCE IN GROUP RANKING
The MVSP group function was used to express the above CCA analysis and the sample
sites to consider vectoring of reef zones, which corroborated the PCA euclidean bi-plot (Figs.
8 & 15).
29

Figu
re 1
5. C
CA
com
bine
d bi
-plo
t usi
ng M
VS
P gr
oupi
ng fu
nctio
n to
illus
trate
axi
s 1
actin
g as
a v
ecto
r for
the
Myt
ilus
reef
zon
es (M
Q) t
o
the
right
, S. a
lveo
lata
(SQ
) to
the
left,
and
NQ
zon
e m
ore
even
ly
dist
ribut
ed, c
orro
bora
ting
PC
A re
solu
tion
(Fig
. 8).
68%
of v
aria
nce
extra
cted
by a
xes;
1 (5
3%);
2 (1
5%) a
nd 7
7.6%
exp
lain
ed b
y
envi
ronm
enta
l con
stra
int;
1 (6
0.5%
); 2
(17.
1%).
X= (M
Q)M
ytilus zone
= (SQ)Sab
ellaria
zon
e
= (NQ) Zon
e to W
est
30

M. ed
ulis
S. alv
eolat
a
E. int
estina
lis
Axis 2
n = 12
35
31

To increase the strength of the environmental constraints and reduce the effect of inter-
correlation between the salinity proxy, turbulence proxy and submersion, the latter two were
removed and CCA repeated with only the three assemblage defining species (ter Braak,
1994).
The revised CCA ordination shows variance extraction reduced from 68% (Fig. 12) to 57% of
variance in the taxa presence data extracted in the first two axes. This supports the refined
cluster and PCA extracted dispersion of the three assemblages by these species (Figs. 11 &
16); Variance extracted by Axes; 1 (45%); 2 (12%). 100% of variance is explained by
environmental constraint: Cc; Axes; 1 (78.65%), 2 (21.35%; Fig. 16). Axis 1 acts as a vector
in the separation of E. intestinalis and S. alveolata from M. edulis, i.e. shore position (HW):
Se r; Axes 1 (0.68), 2 (0.41) (Fig. 16).
KEY
Vector extension illustrations
Perpendicular to environmental vector
n = 954Axis 1 r2 = 0.47Axis 2 r2 = 0.17
Figure 16. CCA bi-plot of three assemblage defining taxa and three environment variables,
(HW) gradient of shore position towards low water, (Substrate) gradient of increasing abiotic
stability, (FW) proxy for gradient of increasing salinity by reducing exposure to freshwater.
57% of the variance is extracted by axis1 (45%) and 2 (12%). 100% of the variance was
explained by environmental constraint: Axes; 1 (78.65%); 2 (21.35%).
32

The environmental factors inferred by CCA were tested for significance. Shore position and
the proxies for salinity and turbulence were euclidean distance measurements (m) from the
quadrats. Each sub cell of any given quadrat was allocated the same distance value on the
environmental gradient, because the distance between the furthest cells in a quadrat was
within the GPS accuracy specification (Garmin, 2012). As quantitative abundance data were
not available, occurrence density was calculated from sub sample presence within each
quadrat as a rapid assessment proxy for abundance (Kent, 2012; Ramsay, 2006).
The covariance between taxa occurrence density and the environmental variables of shore
position, salinity proxy and turbulence proxy was tested by non parametric Spearman
correlation (SPSS); as the taxa distribution was skewed (Hawkins, 2009). This confirmed
strong significance, corroborating the CCA inference of the opposing taxa response of S.
alveolata (Sa) and M. edulis (Me), to the shore position gradient, with the latter showing
increasing occurrence density towards the more desiccated environment higher on the shore
(Figs. 13(a) & 17(a, b) : Sa, rs = 0.399, N = 58, P = 0.002; Me, rs = -0.601, N = 58, P <
0.001.
Distance to high water (m)(a)
S. alveo
lata occurrence de
nsity pe
r m
2
N = 58r2 = 0.16
(b)
N = 58r2 = 0.36
M. edu
lis occurrence de
nsity pe
r m2
Distance to high water (m)
Figure 17. The significant and opposed Spearman correlations of S. alveolata (a) and M.
edulis (b) to shore position with M. edulis occurring in greater density in more desiccated
conditions closer to high water; corroborating CCA inference of taxa ranking (Fig. 13(a)).
33

The Spearman correlation suggests the inferred CCA covariance of E. intestinalis and shore
position is non-significant: rs = 0.230, N = 58, P = 0.083 (Figs. 13(a); 18)
Distance to high water (m)
E. intestinalis occurrence de
nsity pe
r m
2N = 58r2 = 0.05
Figure 18. Non-significant relationship between E. intestinalis and shore position gradient
(Fig. 13(a)).
S. alveolata showed significant increasing occurrence density as the distance from the
freshwater channel increased, corroborating the CCA inferred response to reduced salinity
(Fig. 14(b)): rs = 0.439, N = 58, P = 0.001 (Fig. 19).
S. alveo
lata occurrence de
nsity pe
r m
2
N = 58r2 = 0.19
Distance to freshwater channel (m)
Figure 19. Significant increasing S. alveolata occurrence density as the distance from the
freshwater channel increases, corroborating CCA taxa response (Fig. 14(b)).
34

Covariance with distance from the freshwater channel and E. intestinalis (1) and M. edulis
(2) occurrence density was insignificant, corroborating the E. intestinalis position at the
source point of this environmental variable in CCA figure 14(b) and inferring weak taxa
response to salinity gradient Fig. 20(a,b): (1) rs = -0.137, N = 58, P = 0.304; (2) rs = 0.167, N
= 58, P = 0.167.
N = 58r2 = 0.02
Distance to freshwater channel (m)
E. intestinalis occurrence de
nsity pe
r m
2
(a)
M. edu
lis occurrence de
nsity
per m
2
N = 58r2 = 0.03
Distance to freshwater channel (m)(b)
Figure 20. Insignificant covariance of E. intestinalis (a) and M. edulis (b) occurrence density
with distance from the freshwater channel (Fig. 14(b)).
Spearman correlation confirmed similarly significant covariance between S. alveolata (1) and
M. edulis (2; Fig. 21(a, b)) i.e. increased occurrence density with distance increase from the
turbulence proxy which supports the close perpendiculars either side of axis 2 in the CCA
taxa response ranking illustrated in figure 21(c): (1) rs = 0.285, N = 58, P = 0.03; (2) rs =
0.279, N = 58, P = 0.03 (Fig. 21).
Distance to turbulence proxy datum (m)(a)
S. alveo
lata occurrence de
nsity pe
r m
2
N = 58r2 = 0.08
Distance to turbulence proxy datum (m)(b)
M. edulis occurren
ce den
sity per m
2
N = 58r2 = 0.08
Figure 21. Significant increase in occurrence density of S. alveolata (a) and M. edulis (b)
with increasing distance from the turbulence proxy datum, corroborating CCA taxa response
(Fig. 14(c)).
35

E. intestinalis showed non-significant covariance with the turbulence proxy: rs = -0.115, N =
58, P = 0.39 (Fig. 14(c) & 22).
Distance to turbulence proxy datum (m)
N = 58r2 = 0.01
E. intestinalis occurrence de
nsity pe
r m
2
Figure 22. Non-significant covariance between E. intestinalis and the turbulence proxy (Fig.
14(c)).
Unlike the three environmental variables tested by correlation, the data for stability and
submersion were collected to sub sample level. Therefore, a more expeditious method was
used in testing significance. Stability was tested for association with the three zones and
submersion for difference between the zones; and the zones tested for taxa association.
To consider stability association with the zones, the frequency distribution of samples by
Wentworth scale category (Table 3; Little, 2000) was tested with two-way Chi square
(SPSS). There was only one granule sample (Zone SQ), representing 0.07% of the total
sample, and this was removed to maintain the integrity of the test (Hawkins, 2009).
Association was significant and corroborated CCA axis 1 vectoring of M. edulis (dominant in
Zone MQ; Fig. 27), with more stable substrate, away from S. alveolata (dominant in Zone
SQ; Fig. 27); X26 = 210.13, N = 1449, P < 0.001 (Figs. 12 & 13(b)).
The median and upper quartile substrate classifications were pebble and cobble,
respectively, in all zones; but in Zone SQ & NQ sand was the lower quartile value compared
to more stable cobble in Zone MQ (all values mm): Zone MQ; 3.98, 3.98 – 64.57, n = 300:
Zone SQ; 3.98, 0.063 – 64.57, n = 600: Zone NQ; 3.98, 0.063 – 64.57, n = 550 (Fig. 12).
36

Whilst there are boulders in Zone SQ and NQ, instability is derived from the higher sand
fraction of Zone SQ & NQ. One-way Chi square (SPSS) testing of Zones SQ & NQ only,
show significant heterogeneity, due to the higher sand and boulder content of Zone NQ; X23
= 47.39, N = 1149, P < 0.001 (Fig. 13(b) & 23).
n= 300 n= 550n= 599*
Sand SandSand
Pebble PebblePebble
Cobble Cobble Cobble
Boulder Boulder
% Substra
te Type
Zone
Stability
* 1 sample removed (granule) from statistical test; representing 0.07% of total sample Figure 23. Sample distribution by zone and substrate type, illustrating the stability gradient
and corroborating CCA taxa response of M. edulis and S. alveolata inferred by zone
association (Fig. 13(b) & 27).
A Kruskal – Wallis non-parametric test was used to test the significance of difference in the
submersion extent of samples in each zone, in view of skewed data; before testing taxa
association with zones. The submersion median was; 0, 0 – 0.10, N = 1450 and there was
no significant difference between zones; X22 = 2.359, n1 = 300 , n2 = 600, n3 = 550, P =
0.307 (Fig. 14 (a) & 24).
n = 300 n = 600 n = 550
Zone
Subm
ersion
exten
t decim
al
Figure 24. Submersion extent by zone showing non – significant difference between zones
and highly skewed data with many extremes and outliers (14(a)).
37

The sand samples were prepared for analysis to consider grain size distribution at each
zone and associated taxa. Each sample of 70g – 270g was dried in a Thermo Scientific
Heratherm oven at 500C, on low fan, for 4 days and de-aggregated with a stainless steel
spatula. The samples were dry-sieved using a Fritch Analysette Type 03.502 Ro-Tap shaker
at amplitude seven for five minutes duration per sample (Wentworth, 1922). Sieve size
selection is shown in table 5. The sand fractions were weighed and cumulative mass
retained by each fraction analysed using Microsoft Excel (Fig. 25).
Table 5. Sieve selection and phi value for Ro-Tap analysis of sand samples
Sieve No Size (qm) Phi value1 500 1.002 355 1.493 250 2.004 180 2.475 125 3.006 P 3.99
The median phi value at 50% cumulative distribution was 2 (medium sand) in all three zones
and indicates that all three zones were well sorted, with the samples from Zone SQ & NQ
slightly skewed towards smaller particles at the upper quartile (Fig. 25; Wentworth, 1922).
The sample size was too small, and expected cell values too low, to statistically test
independence of the distribution by zone.
Phi value
Larger SmallerGrain size Larger SmallerGrain size
Phi value
Figure 25. Median % of Phi fraction mass (left) and cumulative % of fraction mass (right);
illustrating sample particle size distribution by intertidal zone. (MQ, n = 6; SQ, n = 11, NQ, n
= 11).
38

Two way Chi-square (SPSS) was used to test the association between the 11 key taxa and
the three intertidal zones, inferred by PCA & CCA (Figs. 4, 8 & 15). Significant association
between the taxa and distinct zones was confirmed; X220 = 1516.32, N = 2518, P < 0.001
(Fig. 26). Zone MQ showed highest presence frequency of M. edulis, L. littorea and G.
umbilicalus: Zone SQ; S. alveolata, E. intestinalis and F. vesiculosus: Zone NQ; C.
montagui, E. intestinalis and L. littorea (Fig. 26). The higher frequency of C. montagui in
Zone NQ, was also significant (one way Chi-Square (SPSS)); X22 = 63.86, N = 268, P <
0.001 (Figs. 4, 8, 15 & 26). Zones MQ and SQ assemblages in figure 26 are similar to those
inferred by the final cluster analysis resolution (Fig. 11).
Taxa:1. E. intestinalis 2. S. alveolata 3. M. edulis 4. F. vesiculosus 5. Ulva 6. O. pinnatifida
7. C. officinalis 8. C. rupestris 9. L. littorea 10. G. umbilicalis 11. C. montagui
Presen
ce cou
nt
n = 2518
1 2 3 4 5 6 7 8 9 10 11 1 2 3 4 5 6 7 8 9 10 11 1 2 3 4 5 6 7 8 9 10 110
Zone MQ Zone NQZone SQ
Figure 26. Presence frequency of the 11 key taxa at the three intertidal zones, illustrating
heterogeneity in community structure. “N” is greater than total sample size (1450) as
numerous taxa may be present in each sample (Figs. 4, 8 & 15).
39

Association between the zones and the three assemblage defining taxa, E. intestinalis, M.
edulis and S. alveolata, was tested by two way Chi square (SPSS). This showed significant
association; X24 = 783.76, N = 1078, P < 0.001 (Figs. 4, 8, 15 & 27). This supports the PCA
opposed presence vectoring of S. alveolata to M. edulis (Zones MQ & SQ); but not Zone
NQ, as E. intestinalis exhibits high presence frequency in both Zone SQ & NQ, the latter of
which has intermediate presence of the three taxa, when compared to Zones MQ & SQ
(Figs. 4, 8, 15 & 27).
KEY:
1. E. intestinalis
2. S. alveolata
3. M. edulis
Presen
ce cou
nt
1
1
1
2
2
2
3
3
3
n = 1078
Figure 27. Significant association between zones MQ & SQ with M. edulis and S. alveolata
and high E. intestinalis presence in zones SQ & the intermediate zone, NQ, corroborating
PCA & CCA inferences (Figs. 4, 8 & 15).
40

As CCA inferred shore position as the environmental factor most correlated with taxa
response, shore position was tested for difference by zone (Fig. 13(a)). As the distances
were normally distributed, parametric testing was suitable and most powerful (Hawkins,
2009). This was significant (One-way Anova (SPSS)); F2, 1447 = 836.47, P< 0.001, N = 1450
and corroborates the taxa response ranking to shore position, inferred by CCA, of S.
alveolata and M. edulis (by zone association; Figs. 13(a) & 27). Mean distance (m) from high
water for Zone MQ, associated with Mytilus, was; 54.72, +/- 0.45, n = 300: Zone SQ,
associated with Sabellaria; 119.02, +/- 1.13, n = 600: Zone NQ, intermediate taxa
association; 104.05, +/- 0.91, n = 550 (Fig. 28).
N = 1450CI = 95%
HW Distan
ce to
high water (m
)
Figure 28. Significant difference in mean distance from high water in the samples of the
three intertidal zones (One-way Anova; Figs. 13(a) & 27).
41

DISCUSSION
The boulder and cobble shore at Cei Bach exhibited distinct community assemblage
patterns within the reef, to the west and to the south-east, as hypothesised (Fig. 26). Cluster
analysis and PCA were highly corroborative in distilling the overall community of 52 taxa to
the seven key species that were driving the greatest variance, and assemblage distinction
(App. 1; Figs. 6 & 11(a & b)). It was also confirmed that two zones could be associated with
assemblage group defining taxa; Zone SQ with S. alveolata and Zone MQ with M. edulis;
but, the defining taxa of the third assemblage, E. intestinalis, could not be distinctly zone
associated as it was present in Zone MQ and populated zones SQ and NQ with high
frequency, inferring a broader fundamental niche than two other taxa (Figs. 11(a & b), 27;
Begon et al, 2006).
The EU Habitat Directive designation of the S. alveolata biotope recognises the high
biodiversity supported by this ecosystem engineer, and the higher taxa presence frequency
in Zone SQ, compared to both other zones, and higher species richness than the Mytilus
zone, indicates higher biodiversity in the reef (Figs. 4 & 26; Table 1; Maddock, 2008). In view
of the limitations of the abundance proxy, more refined data are required to formally quantify
this proposition, e.g. Shannon-Wiener index (Espinosa & Guerra-Garcia, 2005; Ramsay,
2006). Similar species richness in zones SQ and NQ, lower bare shore coverage in zone SQ
and intermediate taxa frequency in Zone NQ, suggest a biodiversity gradient decreasing
through zones, SQ, NQ and MQ (Figs. 4 & 26; Table 1; Park, 2008).
PCA inferred similar axis 1 vectoring of group B (E. intestinalis) and C (S. alveolata), driven
by common presence in zones SQ and NQ, and similar lower shore position (Figs. 8, 27 &
28). CCA corroborated this as a strong response to the shore position gradient and vectored
group A (M. edulis) opposite groups B and C, which are closer to low water resulting in
increased periods of submersion; identical resolution to the simple matching cluster
dendogram (Fig. 11, 15 & 28; Leps & Smilauer, 2003). Response to the CCA submersion
42

gradient illustrated in figure 14(a) is not discussed further as it is highly intercorrelated with
shore position and proximity to the freshwater channel, and non-significant (Fig. 24; Leps &
Smilauer, 2003). However, this was resolved in cluster analysis and corroborated with PCA
by treating standing water as a species, for analysis purposes and confirms the association
of E. intestinalis, O. pinnatifida, F. vesiculosus and S. alveolata with higher submersion (Fig.
10).
The broad fundamental niche explanation of substantial E. intestinalis presence in zones SQ
and NQ is supported by its competitive ability, nutrient uptake efficiency, euryhalinity and
high tolerance of nutrient load (Fig. 27; Zhang, 2012). Superior net primary productivity of E.
intestinalis, similar to terrestrial grass as discussed by Silvertown et al (2006), enables it to
characteristically outcompete other taxa when nitrate and light is available in salt, fresh or
brackish conditions (Hutchinson, 1960; MarLIN, 2013; Smith et al, 2006; Zhang et al 2012).
Zhang (2012) undertook genetic studies of Ulva linza, a congener of E. intestinalis, and
concluded that early evolution and adaptation of mechanisms including carbon concentrating
proteins, similar to C-4 plants, and heat-shock proteins facilitated rapid radiation and
successful colonization of highly stressed coastal ecosystems. Other studies have shown
strong functional guilds among C-4 terrestrial grass species, and in this study, Ulva lactuca,
demonstrated similar variance to E. intestinalis when environmentally constrained, which
was not as clearly extracted in the earlier PCA analysis (Figs. 7 & 12; Fargione et al, 2003).
The highly efficient net primary productivity and simple thalli solution of E. intestinalis, that
absorbs and processes dissolved nutrients, contrasts with the relatively complex biology and
filter feeding mechanisms of M. edulis and S. alveolata, which suffer impaired feeding
efficiency in high seston concentration and showed reduced occurrence as turbulence
increased (Figs. 14(c); 21(a, b & c); Dubois et al, 2005 & 2009; Seed & Suchanek, 1992;
Zhang, 2012).
E. intestinalis and other Ulva sp. are widely used to indicate fresh water incursion and
eutrophication (Raffaelli et al, 2004; Worm et al, 2000). The Environment Agency (EA)
43

classifies nutrient loading of UK rivers (1 – 6), with class five and six being high and very
high respectively. Agricultural pollution from freshwater run-off can alter community structure
at the intertidal interface but high or very high EA nitrate classification levels are rare in
Welsh rivers, compared to 32% in England, suggesting that the saline diluting effect of the
river may be more important than allochthonous nitrate loading of the study area (EA, 2013a;
Hack et al, 2009; Pusceddu et al, 2011). However, 2008 statistics are the most recent EA
nitrate data published and exceptionally high rainfall levels in 2012 may have increased
agricultural run-off and eutrophication levels (Dodds, 2006; Little et al, 2009; MetOffice,
2013).
The land draining into the Llethi river basin is not designated a Nitrate Vulnerable Zone
under the Nitrate Pollution Prevention Regulations 2008 (as amended 2013), but these
regulations are being phased in with effect from May 2013, revised designations will be
issued shortly and the sensitivity of the S. alveolata reef within the SAC could result in
designation and increased protection from nutrient perturbation (Defra, 2013; EA, 2013b).
Figure 11 illustrates cluster and PCA axis 2 vectoring of group B (E. intestinalis) from C (S.
alveolata) and corroborative CCA infers stronger response to the salinity gradient by S.
alveolata (Figs. 12; 14(b)), despite similar shore position response between the taxa (Fig.
13(a); axis 1). Non-significant covariance with the freshwater channel infers E. intestinalis
euryhalinity and in figure 14(b) the species is located very close to the source of the gradient
arrow of environmental change (Fig. 20(a)). In contrast, S. alveolata occurrence density
reduces closer to the freshwater channel suggesting that the reef extent is limited to the west
by reduced salinity or competitive exclusion (Figs. 4 & 19; MarLIN, 2012; Smith et al, 2006;
Townsend et al, 2008).
It would be difficult to determine the relative impact of E. intestinalis competition and reduced
salinity on the S. alveolata population by inter-seasonal study of the reef boundary alone, as
E. intestinalis dies back in late summer when terrestrial freshwater run-off and flow rates
also decline (EA, 2013a; Worm et al, 2000). Mesocosm exclusion experiments would be an
44

experimental design successfully tested in other investigations (O’ Gorman et al, 2010).
Such further studies could investigate switching between top-down and bottom-up pressures
altering the community structure at that time (Menge, 2000). Also, mobile L. littorea may
migrate further down the shore to the S. alveolata assemblage, with which it is often
associated, to breed and forage fucoids as they start their nutritious growth season (Fig. 11;
Campbell, 2004; MarLIN, 2012).
Mytilus is absent from the reef (SQ), where S. alveolata live worm density is highest, but
dominates Zone MQ, which has very low presence frequency of S. alveolata, algae,
Cirrepedia or other taxa (Figs. 4, 5, 26 & 27; Table 1). M. edulis is euryhaline and more
tolerant of desiccation than many other biogenic reef species, and Kautsky (1982) found
dwarf specimens populating inner Baltic habitats of salinity as low as 4-50/00 (Seed &
Suchanek, 1992). CCA ranked M. edulis response to the reducing salinity gradient far lower
than S. alveolata, and Mytilus covariance with the salinity proxy was non-significant (Figs.
14(b), 19 & 20(b)). Temperature data were not collected in this study, but M. edulis tolerates
the extremes associated with upper shore exposure, by using adapted nucleating agents in
the haemolymph (Bourget, 1983). The environmental stress of the upper shore is within the
broad fundamental niche of M. edulis, reflected in shore position response highly variant to
S. alveolata and E. intestinalis, and the strong axis 1 vectoring of group A from B and C
(Figs. 11 & 13(a); Little et al, 2009; UK Marine, n.d.).
The broad fundamental niche of M. edulis suggests that other factors explain its absence in
Zone SQ, such as substratum stability, particle availability and size, and competition (Figs. 4;
MarLIN, 2012; Seed & Suchanek, 1992). The response to the substrate gradient was
vectored by CCA axis 1, and M. edulis was ranked with increased stability compared to E.
intestinalis and S. alveolata positioned together, with lower stability (Fig. 13(b)). Substratum
stability plays a key role in dispersal of aquatic macroinvertebrates and the frequency
distribution, shown in figures 26 & 27, support Mytilus responding positively to stability and
S. alveolata and E. intestinalis, to the more unstable zones with higher sand fractions (Table.
1; Fig. 23; Elsasser, 2013; Hussain & Pandit, 2012). Adult M. edulis require highly stable
45

substratum for successful settlement and the community structure patterns support this (Fig.
23; Albrecht, 1998; Elsasser, 2013; UK Marine, n.d.).
S. alveolata ecosystem engineering subsequently stabilises mobile substratum within the
optimal niche settlement range of M. edulis (Dubois et al, 2006; Maddock, 2008). However,
studies in Mont St. Michel, France, including Desroy et al (2011), have shown that in optimal
niche conditions the honeycomb worm is able to competitively exclude M. edulis and
Crassostrea gigas; and succession only occurs where the reef is seriously impaired or
damaged by anthropogenic trampling. Dubois et al (2006) found that where the cultivated
bivalve, C. gigas, density was lowest, S. alveolata reef health and associated species
richness was highest, concluding that oysters had greater impact on reef species
assemblages than algal epibionts, and a similar pattern is seen between M. edulis and the
reef in this study (Fig. 26).
As M. edulis presence is frequent either side of the reef, relative specific larval dispersal is
not discussed further, other than that this bi-modal distribution supports the theory of
competitive exclusion of Mytilus by S. alveolata and findings of Desroy et al (2011; Blythe &
Pineda, 2009; Fig. 26). Desroy et al (2011) also found that biodeposits of M. edulis covered
settling sporelings of F. serratus, inhibiting growth and new settlement, which is consistent
with the very low fucoid coverage of Zone MQ (Fig. 4; Table 1; Desroy et al, 2011; Little,
2000). Whilst M. edulis is associated with lower biodiversity in this study, mussels can also
act as a foundation species in other ecological conditions and provide substratum and
facilitation of increased biodiversity; for example Modiolus modilous in Strangford Loch,
Northern Ireland (Fig. 26; Elsasser et al, 2013).
Sand grain availability is critical to tube building macroinvertebrates, such as freshwater
cased caddis flies (Trichoptera) and marine Sabellaria (Dubois et al, 2005; Hussain &
Pandit, 2012). As there was only a small difference in sand particle size between the zones it
is likely that availability is the stronger limiting factor, and figure 23 illustrates significant
46

heterogeneity, and the larger sand fraction of zones SQ and NQ, offering a plausible
explanation for higher occurrence density of S. alveolata in these zones (Fig. 25 & 27).
Grain suspension is also important to the ecology of M. edulis and S. alveolata and studies
have concluded optimal ranges in seston concentration, beyond which clearance rates
decline and fitness is impaired, which is supported by significant density occurrence increase
in both species with distance from the turbulence proxy (Fig. 14(c), 21(a & b); Dubois et al,
2003 & 2009). However, both species do require water flow from currents and wave action
for particulate re-suspension, activation of feeding mechanisms and S. alveolata grain
recruitment (Dubois et al, 2005). Therefore, the simplistic turbulence proxy should be treated
with a caveat of caution.
S. alveolata is often associated with F. serratus which exploits reef substrate for settlement
and development of sporelings, similarly to the settlement and metamorphosis of new
generations of meroplanktonic trochophores of S. alveolata (MarLIN, 2012; Pawlik, 1988). In
this study F. serratus is replaced with F. vesiculosus, which is an indicator of more sheltered
shores (Figs. 11, 12 & 26; Little, 2000). Both S. alveolata and F. vesiculosus are considered
foundation species, providing increased three dimensional substrate and surface area for
other taxa, increasing biodiversity in a similar way to terrestrial forest canopy (Diaz et al,
2012; Little et al, 2009; Maddock, 2008). The relationship between the F. vesiculosus and S.
alveolata is mutualistic, as the fucoid provides a desiccation buffer at low tide without
impairing filter feeding upon tidal inundation as it re-floats vertically (Little et al, 2009).
This study is a “snapshot” resolving and focused upon the assemblage defining taxa of the
boulder and cobble intertidal community, which is dynamic, temporally affected and
vulnerable to extreme perturbations (Bertocci et al, 2012; Lancaster & Savage, 2008;
Wethey et al, 2011).
47

CONCLUSIONS
The indirect and direct ordination techniques used to analyse the boulder and cobble shore
at Cei Bach were effective in supporting all study objectives and hypotheses, other than the
key assemblage defining species, E. intestinalis, was not zone associated due to broad
fundamental niche and competitive superiority in the absence of limiting factors (Zhang,
2012).
Increased biodiversity was associated with the biogenic reef and the results, and literature
review, suggest that its dynamic extent is effectively “framed” by; the open sea to the North;
desiccation to the south; insufficient salinity, high seston levels and competitive exclusion, by
E. intestinalis, to the west and; desiccation, and possibly competitive exclusion by M. edulis,
to the south-east (Fig. 4; Lancaster & Savage, 2008; Maddock, 2008). Therefore, the relative
coverage and abundance of M. edulis, E. intestinalis and S. alveolata could be considered
useful bio-indicators for long term reef health and succession monitoring (Desroy et al, 2011;
Dubois et al, 2006; UK Marine, n.d.). There is evidence of facilitation between S. alveolata
and F. vesiculosus and functional guild relationship between Ulva sp. (Fig. 26; Fargione et
al, 2003; Little et al, 2009).
As we progress from a species and habitat focus towards an ecosystem approach to
conservation, the techniques used in this study may be adapted and improved in assessing
the ongoing ecological status, succession and reference conditions at Cei Bach, and similar
habitats within a landscape and river basin scale, according to the European Water
Framework Directive (EA, 2013a, EAW, 2012; Maddock, 2008; Muxika et al, 2007).
48

CRITIQUE
By combining cluster and PCA, rare taxa and those driving low variance were excluded and
the main criticisms of Correspondence Analysis (CA) mitigated, thus enabling robust CCA
direct ordinations (Gauch, 1982; Palmer, n.d.). However, these taxa should be considered in
further studies, as they are important in complex food webs and community structure
(O’Gorman & Emmerson, 2009; Pusceddu et al, 2011).
For expediency, and as CCA design is suitable for contingency data (presence/absence),
these were used as an efficient alternative to abundance data, but the accuracy of this
surrogate approach has not been tested in this study (Kovach, 2011; Ramsay 2006).
CCA inferred a broader picture of taxa ranking and community response, to multiple
environmental factors, than individual linear regressions (Kovach, 2011). However, CCA
relies on the skills and experience of the ecologist to select the most appropriate
environmental variables and there was, perhaps inevitably, some intercorrelation between
shore position, submersion and proxies for turbulence and salinity which reduced correlation
strength, particularly in axis 2 (Fig. 12; ter Braak, 1986 &1994). This is a challenge for most
studies of complex intertidal communities and more advanced methods continue to be
developed (O’ Gorman et al, 2010).
The salinity gradient proxy of distance from the freshwater channel is relevant only to low
tide conditions, as freshwater stratifies above more dense seawater on tidal inundation, but
remains a good indicator of niche tolerance to daily salinity extremes (Little et al, 2009). The
turbulence proxy is supported by peer reviewed studies of increased seston proximate to
freshwater outfall, but no corroborating measurements were taken in this study (Aller &
Cochran, 1976; Sebens et al, 1997).
49

My study was very ambitious and, with hindsight, I collected too much data of which some
were not salient, increasing data analysis complexity and reducing efficiency. The mantra,
“keep it simple” would have facilitated faster results and I may have spotted omissions and
errors more quickly. For example, temperature data could have been interesting and
coverage data for E. intestinalis would have been useful in the analysis of taxa response of
the three defining species, and as a bio-indicator of eutrophication (Worm et al, 2000;
Zhang, 2012). Selection of alternative software, such as Supervised Multidimensional
Scaling (superMDS) in “R”, may also have reduced my ordination process to fewer steps,
improved efficiency and inferred further interesting variances (Cran, 2013).
With increased funding, GPS sample location accuracy could have been improved to 3 – 5m
with the use of a supplemental Differential Beacon Receiver (GPS Information, 2013).
ACKNOWLEDGEMENTS
I would like to thank, Dr. Philip Pugh (my supervisor), Cardigan Bay Marine Wildlife Centre
(CBMWC) - Laura Mears, Sarah Perry, Steve Hartley, Matt Jones and my other co-
volunteers for their support and encouragement, The Countryside Council for Wales (CCW)
– Paul Brazier, Julia Mackenzie and Jacqueline Bodimead at Anglia Ruskin University (ARU)
and Roger & Bethan Bryan for their hospitality and generous car parking at Cei Bach.
REFERENCES
Albrecht, A.S. 1998. Soft bottom versus hard rock: Community ecology of macroalgae on
intertidal mussel beds in the Wadden Sea. Journal of Experimental Marine Biology and
Ecology: 229: 85–109
Allen, J.H., Billings, I., Cutts, N., Elliott, M. 2002. (Eds). Mapping, Condition & Conservation
Assessment of Honeycomb Worm Sabellaria alveolata Reefs on the Eastern Irish Sea
50

Coast. Unpublished report to English Nature by the Institute of Estuarine and Coastal
Studies, University of Hull. Report: Z122–F–2002
Aller, R.C., Cochran, J.K. 1976. 234Th / 238U disequilibrium in near-shore sediment: Particle
reworking and diagenetic time scales. Earth and Planetary Science Letters. 29(1): 37–50
Aller, R.C., Benningen, L.K., Cochran, J.K. 1980. Tracking particle-associated processes in
nearshore environments by use of 234Th / 238U disequilibrium. Earth and Planetary Science
Letters. 47(2): 161–175
Ausden, M., Drake. M. 2006. (Eds). 5. Invertebrates, in, Sutherland, W.J. Ecological Census
Techniques. (2nd Ed). Cambridge University Press. Cambridge. pp. 214–216
Begon, M., Townsend, C.R., Harper, J.L. 2006. 4th Ed. Ecology – From Individuals to
Ecosystems. Blackwell Publishing. Oxford. pp. 31
Bertocci, I., Araujo, R., Incera, M., Arenas, F., Pereira, R., Abreu, H., Larsen, K., Sousa –
Pinto, I. 2012. Benthic assemblages of rock pools in northern Portugal: seasonal and
between-pool variability. Scientia Marina. 76(4): 781–789
Blythe, J.N., Pineda, J. 2009. Habitat selection at settlement endures in recruitment time
series. Marine Ecology Progress Series. 396: 77-84
Bolam, S., Bremner, J., Forster, R. 2011. Ecological Characterisation of the Intertidal Region
of Hinkley Point, Severn Estuary: Results from the 2008 Field Survey and Assessment of
Risk. Report to Cefas by the Institute of Estuarine and Coastal Studies, University of Hull.
Bourget, E. 1983. Seasonal variations in cold tolerance in intertidal molluscs and their
relation to environmental conditions in the St. Lawrence estuary. Canadian Journal of
Zoology. 61: 1193-1201
Boyes, S., Allen, J.H. 2008. Intertidal monitoring of Sabellaria alveolata reefs in Pen Llyn a’r
Sarnau SAC 2004 / 5. CCW Marine Monitoring Report No. 29
Campbell, A. 2004. 2nd Ed. Seashores and Shallow Seas of Britain and Europe. Hamlyn.
London. pp. 150
Cardigan Bay SAC, 2008. Reefs, available at,
www.cardiganbaysac.org.uk/?s=sabellaria&submit.x=307submit.y=11 [Accessed 04/03/12]
51

CCW. 2013. Llanfihangel Moraine. Available at, http://www.ccw.gov.uk/landscape--
wildlife/protecting-our-landscape/special-landscapes--sites/protected-landscape/sssis/sssi-
sites/llanfihangel-moraine.aspx?lang=en [Accessed, 19/04/13].
Crampton, C.B. 1965. Certain Effects of Glacial Events In The Vale of Glamorgan, South
Wales. Journal of Glaciology. 6(44): 261–266
Cran. 2013. Cran R Project – s Package “superMDS”. Available at, http://cran.r-
project.org/web/packages/superMDS/superMDS.pdf [Accessed 30/04/13]
Davies, J. 2001a. (Eds). 2 Establishing monitoring programmes for marine features, in,
Davies, J., Baxter, J., Bradley, M., Connor, D., Khan, J., Murray, E., Sanderson, W.,
Turnbull, C., Vincent, M. JNCC Marine Monitoring Handbook. pp 27–55, available at, http://jncc.defra.gov.uk/PDF/MMH-mmh_0601.pdf [Accessed 20/03/12]
Davies, J. 2001b. (Eds). 3 Advice on establishing monitoring programmes for Annex 1
habitats, in, Davies, J., Baxter, J., Bradley, M., Connor, D., Khan, J., Murray, E., Sanderson,
W., Turnbull, C., Vincent, M. JNCC Marine Monitoring Handbook. pp 60-69, available at, http://jncc.defra.gov.uk/PDF/MMH-mmh_0601.pdf [Accessed 20/03/12]
Davies, J., Baxter, J., Bradley, M., Connor, D., Khan, J., Murray, E., Sanderson, W.,
Turnbull, C., Vincent, M. (Eds). 2001. JNCC Marine Monitoring Handbook, available at, http://jncc.defra.gov.uk/PDF/MMH-mmh_0601.pdf [Accessed 20/03/12]
Defra. 2013. Magic – Interactive Map – Nitrate Vulnerable Zone & Sensitive Area Search.
Available at, http://magic.defra.gov.uk/website/magic/ [Accessed 02/04/13]
Desroy, N., Dubois, S.F., Fournier, J., Ricquiers, L., Le Mao, P., Guerin, L., Gerla, D.,
Rougerie, M., Legendre, A. 2011. The conservation status of Sabellaria alveolata
(L.)(Polychaeta: Sabellariidae) reefs in Bay of Mont-Saint-Michel. Aquatic Conservation:
Marine and Freshwater Ecosystems. 21: 462–471
Diaz, I.A., Sieving, K.E., Pena-Foxon, M., Armesto, J.J. 2012. A field experiment links forest
structure and biodiversity: epiphytes enhance canopy invertebrates in Chilean forests.
Ecosphere. 3(1): 1-3
Digimap. 2012. Digimap Resource Centre. Available at,
http://digimap.edina.ac.uk/digimap/home [Accessed 10/05/12]
52

Dodds, W.K. 2002. Freshwater ecology: concepts and environmental applications. Academic
Press. San Diego
Dodds, W.K. 2006. Eutrophication and trophic state in rivers and streams. Limnology and
Oceanography. 51: 671–680
Dubois, S., Barille, L., Retiere, C. 2003. Efficiency of particle retention and clearance rate in
polychaete Sabellaria alveolata L. C.R. Biologies.326: 413–421
Dubois, S., Barille, L., Cognie, B., Beninger, P.G. 2005. Particle capture and processing
mechanisms in Sabellaria alveolata (Polychaeta: Sabellariidae). Marine Ecology Progress
Series. 301: 159-171
Dubois, S., Commito, J.A., Olivier, F., Retiere, C. 2006. Effects of epibionts on Sabellaria
alveolata (L.) biogenic reefs and their associated fauna in the Bay of Mont Saint-Michel.
Estuarine, Coastal and Shelf Science. 68: 635–646
Dubois, S., Barille, L., Cognie, B. 2009. Feeding response of the polychaete Sabellaria
alveolata (Sabellariidae) to changes in seston concentration. Journal of Experimental Marine
Biology and Ecology. 376: 94–101
Duigan, C.A., Kovach, W.L. 1991. A study of the distribution and ecology of littoral
freshwater chydorid (Crustacea, Cladocera) communities in Ireland using multivariate
analysis. Journal of Biogeography. 18: 267–280
EA. 2013a. Environment Agency – River Quality - Nutrients. Available at,
http://www.environment-agency.gov.uk/homeandleisure/37813.aspx [Accessed 10/04/13]
EA. 2013b. Environment Agency – Nitrate Vulnerable Zones. Available at,
http://www.environment-agency.gov.uk/homeandleisure/37813.aspx [Accessed 10/04/13]
EAW. 2012. Bathing Water Profile. Available at, http://www.environment-
agency.gov.uk/static/documents/BW_38690_New_Quay_Harbour.pdf [Accessed 01/04/13]
Elsasser, B., Farinas-Franco, J.M., Wilson, C.D., Kregting, L., Roberts, D. 2013. Identifying
optimal sites for natural recovery and restoration of impacted biogenic habitats in a special
area of conservation using hydrodynamic and habitat suitability modelling. Journal of Sea
Research. 77: 11–21
53

Espinosa, F., Guerra-Garcia, J.M. 2005. Algae, macrofaunal assemblages and temperature:
a quantitative approach to intertidal ecosystems of Iceland. Helgoi. Mar. Res. 59: 273–285
Fargione, J., Brown, C.S., Tilman, D. 2003. Community assembly and invasion: An
experimental test of neutral versus niche processes. PNAS. 100(15): 8916-8920
Garmin. 2012. GPS 62 Owner’s Manual. pp. 36. Available at,
http://www8.garmin.com/manuals/GPSMAP62_OwnersManual.pdf [Accessed 15/05/12]
Gauch, H.G. 1982. Noise reduction by eigenvector ordinations. Ecology. 63: 1643–1649
Geograph. 2013. SN4059 : River Llethi near Cei-Bach. Available at,
http://www.geograph.org.uk/photo/2119057 [Accessed 01/02/13]
GPS Information. 2013. Differential GPS. Available at,
http://www.gpsinformation.org/dale/dgps.htm [Accessed, 10/04/13]
GPS Utility. 2012. Available at, http://www.gpsu.co.uk/ [Accessed, 10/03/12]
Greenwood, J.J.D., Robinson, R.A. 2006. (Eds). Principals of Sampling, in, Sutherland, W.J.
Ecological Census Techniques. (2nd Ed). Cambridge University Press. Cambridge. pp 33 –
38; 43–53
Hack, L.A., Tremblay, L.A., Wratten, S.D., Lister, A., Keesing, V. 2009. Benthic meiofauna
community composition at polluted and non-polluted sites in New Zealand intertidal
environments. Marine Pollution Bulletin. 54: 1801-1812
Hawkins, D. 2009. 2nd Ed. Biomeasurement; A Student’s Guide to Biological Statistics.
Oxford University Press. Oxford. pp. 229 & 102
Hayward, P.J., Ryland, J.S. 1995. Handbook of the Marine Fauna of North – West Europe.
Oxford University Press. Oxford pp 246
Hendrick, V.J., Foster-Smith, R.L. 2006. Sabellaria spinulosa reef: a scoring system for
evaluating “reefiness” in the context of the Habitats Directive. Journal of Marine Biology
Association U.K. 86: 665–677
Hussain, Q.A., Pandit, A.K. 2012. Macroinvertebrates in streams: A review of some
ecological factors. International Journal of Fisheries and Aquaculture. 4(7): 114-123
54

Hutchinson, G.E. 1960. On Evolutionary Euryhalinity. American Journal of Science. 258(A): 98-103
IBM, 2011. SPSS Statistics Package. Version 20. Release 20.0.0. IBM Corporation.
JNCC. 2004. Common Standards Monitoring Guidance for Littoral Rock and Inshore
Sublittoral Rock Habitats (version August 2004), available at,
http://jncc.defra.gov.uk/PDF/CSM_marine_rock.pdf [Accessed 30/03/12]
JNCC. 2012. Special Areas of Conservation (SAC), in, JNCC Joint Nature Conservation
Committee, available at, www.jncc.defra.gov.uk/page-23 [Accessed 23/04/12]
Kautsky, N. 1982. Growth and size structure in a Baltic Mytilus edulis population. Marine
Biology. 68: 117-133.
Kent, M. 2012. 2nd Ed. Vegetation Description and Data Analysis – A Practical Approach.
Wiley Blackwell. Oxford. pp. 65–67, 70
Kovach, W.L. 1999. MVSP – A Multivariate Statistical Package for Windows ver. 3.1. Kovach
Computing Services, Pentraeth, Wales
Kovach, W.L. 2011. MVSP – Version 3; Users Manual. Kovach Computing Services,
Pentraeth, Wales
Krebs, J.R. 1998. 2nd Ed. Ecological methodology. Benjamin Cummings. California. pp.405
Lancaster, J., Savage, A. 2008. Sabellaria; How Do You Solve a Problem Like Sabellaria? In
Practice. September: 18–21
Leps, J., Smilauer, P. 2003. Multivariate analysis of Ecological Data using CANOCO.
Cambridge University Press. Cambridge. pp. 37 – 40; 149-166
Lesperance, P.J. 1990. Cluster Analysis of Previously Described Communities From The
Ludlow Of The Welsh Borderland. Palaeontology. 33(1): 209–224
Little, C. 2000. The Biology of Soft Shores and Estuaries. Oxford University Press. Oxford.
pp. 14, 147
55

Little, L., Williams, G.A., Trowbridge, C.D. 2009. 2nd Ed. The Biology of Rocky Shores.
Oxford University Press. Oxford. pp.157, 187 – 206, 228, 235–240.
Maddock, A. 2008. Sabellaria alveolata reefs, in, UK Biodiversity Action Plan Priority Habitat
Descriptions, available at, http://jncc.defra.gov.uk/page-5706 [Accessed 21/04/12]
MarLIN. 2012. Sabellaria alveolata reefs on sand – abraded eulittoral rock, in, Biodiversity
and Conservation, available at,
www.marlin.ac.uk/habitatecology.php?habitatid=351&code=2004 [Accessed 23/04/12]
MarLIN. 2013. Biodiversity and Conservation - Gut Weed – Ulva intestinalis. Available at,
http://www.marlin.ac.uk/speciesinformation.php?speciesID=4540 [Accessed 20/03/13]
Menge, B.A. 2000. Top-down and bottom-up community regulation in marine rocky intertidal
habitats. Journal of Experimental Marine Biology and Ecology. 250: 257-289
MetOffice. 2013. Rainfall station data – Aberporth. Available at,
http://www.metoffice.gov.uk/climate/uk/stationdata/aberporthdata.txt [Accessed 20/04/13]
Moore, J.J. 2009. Intertidal SAC monitoring, Cardigan Bay SAC, May 2007. A report to the
Countryside Council for Wales from Aquatic Survey Monitoring Ltd, Cosheston,
Pembrokeshire. Report no: 57 Muxika, I., Borja, A., Bald, J. 2007. Using historical data, expert judgement and multivariate
analysis in assessing reference conditions and benthic ecological status, according to the
European Water Framework Directive. Marine Pollution Bulletin. 55: 16-29
NBN. 2012. NBN Gateway – Sabellaria alveolata. Available at, http://staging.testnbn.net/Taxa/NBNSYS0000188452 [Accessed 10/03/12]
O’Gorman, E.J.O., Emmerson, M.C. 2009. Perturbations to trophic interactions and the
stability of complex food webs. PNAS. 106(32): 13393-13398
O’Gorman, E.J.O., Jacob, U., Jonsson, T., Emmerson, M.C. 2010. Interaction strength, food
web topology and the relative importance of species food webs. Journal of Animal Ecology.
79: 682-692
Oksanen, J., Minchin, P.R. 1997. Instability of ordination results under changes in input
order: explanation and remedies. Journal of Vegetation Science. 8: 447-454
56

Palmer, M.W. Not Dated. Ordination Methods – an overview. OSU Ecology, available at,
http://ordination.okstate.edu/overview.htm [Accessed 20/03/13]
Palmer, M.W. 1993. Putting Things in Even Better Order: The Advantages of Canonical
Correspondence Analysis. Ecology. 74(8): 2215–2230
Park. C. 2008. Oxford Dictionary of Environment and Conservation. Oxford University Press.
Oxford. pp. 47
Pawlik, J.R. 1988. Larval settlement and metamorphosis of two gregarious sabellariid
polychaetes: Sabellaria alveolata compared with Phragmatopoma californica. 68: 101-124
Pusceddu, A., Bianchelli, S., Gambi, C., Danovaro, R. 2011. Assessment of benthic trophic
status of marine coastal ecosystems: Significance of meiofaunal rare taxa. Estuarine,
Coastal and Shelf Science. 93: 420–430
QGIS. 2010. Quantum GIS (QGIS) v. 1.7.4. Wroclaw.
Raffaelli, D.G., White, P.C.L., Renwick, A. 2004. Health of Ecosystems: the Ythan estuary
case study. pp 379–394 in Jorgensen, S.E., Costanza, R., Xu, F.L. (eds). Handbook of
indicators for assessment of ecosystem health. CRC Press. Florida.
Ramsay, P.M., Kent. M., Reid, C., Duckworth, J.C. 2006. Taxonomic, morphological and
structural surrogates for the rapid assessment of vegetation. Journal of Vegetation Science.
17: 747–754
Ridley, C., Harrison, N.M., Phillips, R.A., Pugh, P.J.A. 2010. Identifying the origins of fishing
gear ingested by seabirds: a novel multivariate approach. Aquatic Conservation: Marine And
Freshwater Ecosystems. 20: 621-631
Ruppert, E.E., Fox, R.S., Barnes, R.D. 2004. 7th Ed. Invertebrate Zoology – A Functional
Evolutionary Approach. Brooks / Cole Cengage Learning. Belmont pp 452–455
Sebens, K.P., Witting, J., Helmuth, B. 1997. Effects of water flow and branch spacing on
particle capture of the reef coral Madracis mirablis (Duchassaing and Michelotti). Journal of
Experimental Marine Biology and Ecology. 211: 1–28
Seed, R. & Suchanek, T.H. 1992. Population and community ecology of Mytilus. pp. 87 - 170
In, Gosling, E. The mussel Mytilus: ecology, physiology, genetics and culture. Developments
in Aquaculture and Fisheries Science: Volume 25. (eds.). Elsevier. Philadelphia.
57

Silvertown, J., Poulton, P. Johnston, E., Edwards, G., Heard, M., Biss, P.M. 2006. The Park
Grass Experiment 1856 – 2006: its contribution to ecology. Journal of Ecology. 94: 801–814
Smith, V.H., Joye, S.B., Howarth, R.W. 2006. Eutrophication of freshwater and marine
ecosystems. Limnology of Oceanography. 51(1): 351-355
Snoeijs, P.J.M., Prentice, I.C. 1989. Effects of cooling water discharge on the structure and
dynamics of epilithic algal communities in northern Baltic. Hydrobiologia. 184: 99-123
ter Braak, C.J.F. 1986. Canonical Correspondence Analysis: A New Eigenvector Technique
for Multivariate Direct Gradient Analysis. Ecology. 67(5): 1167–1179
ter Braak, C.J.F. 1987. The analysis of vegetation-environment relationships by canonical
correspondence analysis. Vegetatio. 69: 69-77
ter Braak, C.J.F. 1994. Canonical community ordination. Part 1: Basic theory and linear
methods. Ecoscience. 1: 127–140
ter Braak, C.J.F., Verdonschot, P.F.M. 1995. Canonical correspondence analysis and
related multivariate methods in aquatic ecology. Aquatic Sciences. 57(3): 255–289
Townsend, C.R., Begon, M., Harper, J.L. 2008. 3rd Ed. Essentials in Ecology. Blackwell
Publishing. Oxford. pp. 247
UK Marine. Not dated. UK Marine SACs Project - Mytilus edulis. Available at,
http://www.ukmarinesac.org.uk/communities/biogenic-reefs/br9.htm [Accessed 10/04/13]
UKMPA Centre. 2012. Introduction to managing European marine sites, in UK Marine
Special Areas of Conservation, available at, www.ukmarinesac.org.uk/ms1.htm [Accessed
21/03/12]
Wentworth, C.K. 1922. A Scale of Grade and Class Terms for Castic Sediments. Journal of
Geology. 30(5): 377–392
Wethey, D.S., Woodin, S.A., Hilbish, T.J., Jones, S.J., Lima, F.P., Brannock, P.M. 2011.
Response of intertidal populations to climate: Effects of extreme events versus long term
change. Journal of Experimental Marine Biology and Ecology. 400: 132–144
58

Worm, B. Lotze, H.K., Sommer, U. 2000. Coastal food web structure, carbon storage, and
nitrogen retention regulated by consumer pressure nutrient loading. Limnology
Oceanography. 45(2): 339-349
Zhang, X., Ye, N., Liang, C., Mou, S., Fan., X., Xu, J., Xu, D., Zhuang, Z. De novo
sequencing and analysii of the Ulva linza transcriptome to discover putative mechanisms
associated with its successful colonization of coastal ecosystems. BMC Genomics. 13: 2-13
Appendices
59

Appendix 1. Species list of taxa present in study
Fucus vesiculosus
F. serratus
F. spiralis
Ascophyllum nodosum
Gibbula umbilicalis
Pelvetia canaliculata
Gelidium latifolium
Porphyra umbilicalis
Ulva lactuca
Laminara digitata
Leathesia diffromis
Osmundia pinnatifida
Palmaria pulmata
Osmundia obtuse
Chondrus crispus
Mastocarpus
Corallina officinalis
Other coralline algae
Lomentaria articulate
Cladophera rupestris
Enteromorpha intestinalis
D. contorta
Codium tomentosum
Sargassum muticum
Laminaria saccharina
Osmundia obtuse
Chaetomorpha linum
Dictyota sp.
60

Sabellaria alveolata (Live)
S. alveolata (Dead)
Mytilus edulis
Littorina littoralis
Littorina littorea
Littorina saxtillis
G. cineraria
G. umbilicalis
Cardium edule
Chthalamus montagui
Semibalanus balanoides
Elminius modestus
Nucella lapillus
Arenicola marinara
Pomotocerus lamarki
Spirorbis spirorbis
Porifera sp.
Decapoda
Pagurids
Aquina equine
Crangon vulgaris
Gigas sp.
Patella vulgaris
Chiton sp.
61
Related Documents