Developmental Cell, Volume 28 Supplemental Information Polycomb Potentiates Meis2 Activation in Midbrain by Mediating Interaction of the Promoter with a Tissue-Specific Enhancer Takashi Kondo, Kyoichi Isono, Kaori Kondo, Takaho A. Endo, Shigeyoshi Itohara, Miguel Vidal, and Haruhiko Koseki Inventory for Supplemental Information There are 3 supplemental figures (Figure S1-3), 1 supplemental table (Table S1). -Figure S1 related to Figure 1. (A) ChIP-seq data at promoter and RBS of Meis2 gene, (B) section planes employed in Figure 1, (C) distribution of distances between promoter/RBS, (D) statistical analysis on the FISH experiments in Figures 1D, 1E and 1F. -Figure S2 related to Figure 2. (A) genomic map of tested fragments for transgenic analysis, (B) summary results for transgenic study, (C) 3C results between promoter and enhancer candidates, (D) ChIP-seq data by H3K4me1 and H3K27ac antibodies at the region surrounding Meis2 locus, (E) statistical analysis on the FISH experiments in Figure 2C and 2D, and (F) Immuno-FISH for PRT/MBE/RING1B. -Figure S3 related to Figure 3. (A) summary for the tripartite interaction between promomter/MBE/RBS in Figures 3B and 3C, (B) summary for association between RING1B and each FISH probe at different stage embryos Figures 3D and 3E, and (C) summary of the RING1B binding sites surrounding genes and several examples of gene loci bound by RING1B. -Table S1 related to Figure 4. (A) summarizes promoter/MBE/RBS association in Figure 4B, and (B) summarizes promoter/MBE/RBS association in Figure 4C.

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

Developmental Cell, Volume 28
Supplemental Information
Polycomb Potentiates Meis2 Activation
in Midbrain by Mediating Interaction
of the Promoter with a Tissue-Specific Enhancer
Takashi Kondo, Kyoichi Isono, Kaori Kondo, Takaho A. Endo, Shigeyoshi Itohara, Miguel
Vidal, and Haruhiko Koseki
Inventory for Supplemental Information
There are 3 supplemental figures (Figure S1-3), 1 supplemental table (Table S1).
-Figure S1 related to Figure 1. (A) ChIP-seq data at promoter and RBS of Meis2 gene, (B)
section planes employed in Figure 1, (C) distribution of distances between promoter/RBS, (D)
statistical analysis on the FISH experiments in Figures 1D, 1E and 1F.
-Figure S2 related to Figure 2. (A) genomic map of tested fragments for transgenic analysis, (B)
summary results for transgenic study, (C) 3C results between promoter and enhancer candidates,
(D) ChIP-seq data by H3K4me1 and H3K27ac antibodies at the region surrounding Meis2 locus,
(E) statistical analysis on the FISH experiments in Figure 2C and 2D, and (F) Immuno-FISH for
PRT/MBE/RING1B.
-Figure S3 related to Figure 3. (A) summary for the tripartite interaction between
promomter/MBE/RBS in Figures 3B and 3C, (B) summary for association between RING1B
and each FISH probe at different stage embryos Figures 3D and 3E, and (C) summary of the
RING1B binding sites surrounding genes and several examples of gene loci bound by RING1B.
-Table S1 related to Figure 4. (A) summarizes promoter/MBE/RBS association in Figure 4B, and
(B) summarizes promoter/MBE/RBS association in Figure 4C.

-Supplemental Figure Legends
-Supplemental Experimental Procedures
-Supplemental References

Kondo T. Figure S1
section planes
LM
MB
LGEMGE
CM
A
C
1050Log10(P-value)
PRT/RBS
MB
LGE
MGE
CM
LM
average distance (μm)
0.290
0.344
0.143
0.126
0.137
SD
0.136
0.169
0.143
0.121
0.0956
n
297
209
195
246
226
wt
wt
MB LGE MGE CM LM
MB
LGE
MGE
CM
LM
average distance (μm)
0.293
0.313
0.287
0.253
0.274
SD
0.124
0.126
0.107
0.0998
0.136
n
264
209
223
214
234
R1
Ring1 mut
MB LGE MGE CM LM
D
0 10
250
Distance (µm)
Num
ber o
f pai
red
foci
Cluster 2(50%)
Cluster 1(44%)
Cluster 3(6%)
0.16µm
71.2% 28.8%68.7% 31.3% 72.8% 27.2%MGE LM CM
0 0.75 0 0.75 0 0.75 0 0.750
100
Distance (μm)
Num
bers
LGE12.0% 88.0%
MB14.8% 85.2%
0 0.75
FB
1 2 3 4 5
B
RBS promoter
115920000
Meis2
ES
chr2:115660000 115710000 115870000
FB
MB
LM
2810405F15Rik
4.20 x 10-19.76 x 10-151.19 x 10-147.59 x 10-146.55 x 10-145.87 x 10-1
0
500
P value
TissueExpression
FB+
MB+
LM-
ES-
Rea
ds /
M re
ads
TSS
ES
TSSRING1B
RING1B
RING1B
RING1B
H3K27me3

Human chromosome: chr15 37,110,000-37,710,000 (hg19)
a b c d e f g
37,618k 37,318k37,418k37,518k 37,218k 37,118k
Meis2 (37,183,222-37.392,341)
mouse
chicken
fugu
100 %
50 %
75 %
100 %
50 %
75 %
100 %
50 %
75 %
1 kb
EcoRI EcoRIEcoRIEcoRI EcoRIEcoRIEcoRIEcoRI
promoter sequence c sequence vsequence d
v
mouse corresponding sequences
1
0.8
0.6
0.4
0.2
p-c p-d p-vassociating region
rela
tive
valu
e fo
r 3C
forebrain
midbrain
limb bud
A
RP11-597G23 (37,381,873-37,589,340)RP11-991D13 (37,381,873-37,589,340)
sequence a sequence b
sequence c sequence d sequence e sequence f sequence g
WISH RP11-597G23 RP11-991D13
B
C
E
1050Log10(P-value)
MB
LGE
MGE
CM
LM
average distance (μm)
0.118
0.301
0.258
0.269
0.259
SD
0.0898
0.118
0.0962
0.0963
0.0965
n
307
243
236
219
212
wt
MB
LGE
MGE
CM
LM
average distance (μm)
0.272
0.255
0.257
0.246
0.255
SD
0.0933
0.118
0.102
0.0984
0.105
n
392
266
222
217
209
R1
wt Ring1 mutPRT/MBE
MB LGE MGE CM LMMB LGE MGE CM LM
D
F
chr2; 115,500,000 116,000,000
H3K4me1
H3K27ac
C15orf41 Meis2
mid
brai
n
MBE
MB
2 μm
LMCMMGELGE
PRT MBERING1B
H3K4me1
H3K27ac
fore
brai
n
Kondo T. Figure S2

22%
55%
20%2%
RING1B binding sites
UpstreamPromoter (-4kb,TSS)Promoter (TSS,+4kb)Gene bodyDownstream
Meis1
chr11:18760000 18939000
Foxd1
chr13:99104000 99147000
Onecut3
chr10:79937000 80001000
Tbr1
chr2:61622000 61673000
10kb
CpG island
GSM656523
Pax2
chr19:44811000 44931000
Kondo T. Figure S3
B
6.5 dpc
16-20s
22-26s
colocalization
stage
RING1BPRT
82.0 (128)
82.8 (99)
22.2 (135)
RING1BMBE
28.7 (108)
79.8 (99)
22.2 (135)
RING1BRBS
87.5 (128)
79.0 (119)
78.6 (117)
association
stage
6.5 dpc epi
16-20 somite
22-26 somite
P/M/R
25.4
61.5
24.8
P/R (w/o M)
55.6
12.8
6.0
P/M (w/o R)
5.9
19.8
56.0
no proximity
13.1
5.9
13.2
totalnumber
205
187
234
A
C

P/M
P/R
P/M
P/R
average distance (μm)
0.148
0.160
0.240
0.272
SD
0.0919
0.136
0.109
0.159
n
173
173
190
190
cont.
mut.
A
Baverage distance (μm)
0.118
0.290
0.272
0.293
0.146
0.268
SD
0.0898
0.136
0.0933
0.124
0.0898
0.133
n
307
297
392
264
219
219
cont
rol
P/M
P/R
P/M
P/R
P/M
P/R
8.5
dpc
9.5
dpc
Kondo T. Table S1

SUPPLEMENTAL FIGURE LEGENDS
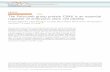
Figure S1, related to Figure 1. Section planes of 11 dpc embryos used for FISH analyses
and statistical analysis for promoter/RBS FISH in wild-type and Ring1 mutant embryos
at 11.5 dpc.
(A) RING1B distribution around Meis2 RBS region (mm9/Chr. 2:115660-115710 kb) and
promoter region (mm9/Chr. 2:115870-115920 kb) in ES cells (ES: GEM656523), forebrain,
midbrain and limb mesenchyme (FB, MB, and LM: GSE48464) revealed by ChIP-seq
analysis. Genomic structure/location of Meis2 and 2810405F15Rik genes is depicted below
the ChIP-seq data. (Right panel) Sequences mapped around Meis2 TSS (from -4kb to +4kb,
shaded rectangle) were collected for each tissue (forebrain (FB), midbrain (MB), limb bud
(LB) and ES cell) and statistically evaluated. P-values indicated in above graphs were
obtained using Wilcoxon test for normalized mapped read counts with 100 bp bins in these
regions and colored in blue (not significant) or red (significant, P < 10-2). (B) Representative
section planes used for FISH analyses of forebrain (FB), midbrain (MB) and limb
mesenchyme (LM) in Figures 1, 2 and 4 are indicated on a Meis2 in situ hybridization image
of an 11.5 dpc embryo. Immuno-histological image of a representative section plane through
the forebrain is shown in the right panel. Positions of lateral ganglionic eminence (LGE),
medial ganglionic eminence (MGE) and cephalic mesoderm (CM) are indicated by open
boxes. Note that MEIS2 is expressed only in LGE. (C) Determination of a threshold
promoter/RBS distance to distinguish paired and unpaired foci. We classified the distribution
of paired foci into three clusters. Details are described in Supplemental Experimental
Procedures. In the lower panels, the composition of cluster 1 and that of cluster 2 plus 3 in
midbrain (MB), LGE, MGE, limb mesenchyme (LM) and cephalic mesoderm (CM) are
shown. (D) Chi-square test of the distribution of promoter/RBS distances. In the top tables,

average distances, standard deviations (SD) and numbers of pairs tested in midbrain (MB),
LGE, MGE, limb mesenchyme (LM) and cephalic mesoderm (CM) in wild type (left) and
Ring1 mutant (right) are summarized. At the bottom, the results of chi-square test among
tissues are summarized by matrices. An example matrix is shown at the left. The chi-square
test results between sample 1 and 4 are indicated by a grey box in this matrix (pointed by
arrows). In the wild type, the average promoter/RBS distances in midbrain and LGE are
significantly different from those of MGE, limb mesenchyme and cephalic mesoderm, but are
not different from each other in the Ring1 mutants.
Figure S2, related to Figure 2. Transgenic analyses to identify Meis2 enhancers and
statistical analysis for promoter/MBE FISH in wild-type and Ring1 mutant embryos at
11.5 dpc.
(A) Genomic sequence similarity of Meis2 and its surrounding regions from mouse, chicken
and fugu compared to the human counterpart (hg19/Chr. 15: 37,110,000-37,710,000) as
revealed by the VISTA tool (http://genome.lbl.gov/vista/index.shtml). Genomic regions
corresponding to human BAC clones used in this study (RP11-597G23 and RP11-991D13)
are shown by bold lines below the homology plot panel. Thirteen transgenic mice harboring
the RP11-597G23 reporter were obtained, but none of them showed midbrain fluorescence.
On the other hand, the RP11-991D13 reporter gave 4 midbrain positive embryos out of 26
transgenic animals. Boxes indicate evolutionary highly conserved DNA sequences outside of
the coding regions. Mouse genomic sequences corresponding to the regions indicated by
boxes as “a” to “g” were tested for their transcriptional regulatory activity by conventional
transgenic assays. Positions corresponding to mouse genomic sequences used for 3C
(chromosomal conformation capture) assays are also shown. Evolutionarily highly conserved

regions within respective EcoRI fragments are indicated by open boxes. The mouse genomic
sequence corresponding to the region indicated by a dotted box as “v” is another midbrain
enhancer candidate from an enhancer browser site (http://enhancer.lbl.gov/) corresponding to
hs181 and was used as a negative control for the 3C assay. (B) Identification of the sequence
“d” as a midbrain-specific enhancer. Whole mount in situ image for Meis2 is also shown
(WISH). (C) Association of the sequence “d” with the promoter specifically in midbrain. (D)
Accumulation of histone H3K4me1 and H3K27ac in 11.5 dpc midbrain at the MBE (dotted
red box) revealed by ChIP-seq analyses. (E) Average distances, SD, numbers of pairs and
matrices of Chi-square test of the distribution of promoter/MBE distances tested in midbrain,
LGE, MGE, limb mesenchyme and cephalic mesoderm in wild type (left) and Ring1 mutant
(right) are summarized. (F) Immuno-FISH analyses of the topology of promoter, MBE and
RING1B bodies in midbrain (MB), LGE, MGE, cephalic mesoderm (CM) and limb
mesenchyme (LM).
Figure S3, related to Figure 3. Summary of colocalization of promoter/MBE/RBS FISH
and RING1B protein and analysis of the position of RING1B in the mouse genome.
(A) Supplement for Figures 3B and 3C. A summary of promoter/MBE/RBS interactions in
6.5 dpc epiblasts and 16-20 and 22-26 somites stage midbrain. (B) Supplement for Figures
3D and E. A summary of the association of promoter, MBE or RBS with RING1B bodies in
6.5 dpc epiblasts and 16-20 and 22-26 somites stage midbrain. (C) Distribution, relative to
the gene body, of the most prominent RING1B-binding sites in the mouse genome based on
ChIP-seq data from ES cells (GEM656523). Several genes harboring additional and more
prominent RING1B binding sites outside of their promoters are shown below the graph.

Tables S1, related to Figure 4. Summary table corresponding to Figure 4B and 4C,
respectively.
(A) Summary table shows averages, standard deviations and numbers of paired foci within
Figure 4B. (B) Summary table shows averages, standard deviations and numbers of paired
foci within Figure 4C.

SUPPLEMENTAL EXPERIMENTAL PROCEDURES
Animals
Transgenic constructs were made with PCR-isolated mouse genomic fragments linked with
the Hsp68-LacZ reporter plasmid (Sasaki and Hogan, 1996). PCR fragments were obtained
by Phusion DNA Polymerase (Thermo Fischer Scientific) and their positions within the
mouse genome (mm9) are listed below. Whole mount in situ hybridization was done using an
established protocol. The Meis2 probe corresponding to the 3’ UTR of Meis2 mRNA was
isolated and cloned by PCR.
Positions of fragments in transgenic analysis (positions are based on mm9:chr.2)
fragment position from to
a 116,026,875 116,028,222
b 115,952,276 115,955106
c 115,843,835 115,846,217
d 115,825,100 115,828,518
e 115,805,413 115,806,189
f 115,722,985 115,724,514
g 115,700,636 115,702,700
ChIP primers
Primers designed for ChIP-qpCR are summarized below.
ChIP primers used in this study (genomic position; mm9:chr.2)

name
(forward)
sequence position
from
to
name
(reverse)
sequence position
from
to
promoter 1f ttgactgaggtgtgcatggc 115,895,419 115,895,438 promoter 1r caacagcgaggtgtccacac 115,895,252 115,895,271
RBS 1f agggtgtgattatatgtgctc 115,681,932 115,681,952 RBS 1r tcaacaacgattccactcgg 115,681,822 115,681,841
MBE 1f cttagcgtgggatttatggc 115,827,231 115,827,250 MBE 1r agtggagagtttatgctccc 115,827,111 115,827,130
Co5 1f cagtactctgtgaggtagcc 115,968,325 115,968,344 Co5 1r gaggaaagacactgggatcg 115,968,153 115,968,172
Co3 1f tctgagctgtgagatgtctg 115,658,666 115,658,685 Co3 1r gtgagccacatatcaacagg 115,658,539 115,658,558
CoI 1f gtggtctatgcacaatcctg 115,809,788 115,809,07 CoI 1r tcagcaggctttgggtaatg 115,809,688 115,809,707
3D-FISH and Immuno-FISH analyses
FISH technology has been described previously (Chambeyron et al., 2005; Tanemura et al.,
2005; Isono et al., 2013). Mouse embryonic samples were fixed with 4% paraformaldehyde
and embedded in paraffin. Detailed information about the fosmids used as FISH probes is
summarized below. Probes were labeled with Alexa Fluor® 488-5-dUTP (Life
Technologies-Molecular Probe C11397), Alexa Fluor® 647-OBFA-dCTP (Life
Technologies-Molecular Probe C21559) and Cy3-dCTP (GE Healthcare PA53021) by
nick-translation (Roche 976776). The histological protocols have been described previously
(Tanemura et al., 2005). The thickness of the sections was 5 µm. After deparaffinization, all
sections were soaked and permeabilized in the HistoVT One solution (Nakalai Tesque,
06380-05) in a glass vessel, which was then incubated in a 100°C boiling water bath for 20
minutes. Immediately after the treatment, sections were washed with water to remove the
detergent prior to performing the FISH or Immuno-FISH processes. In the case of
Immuno-FISH, immuno-staining was carried out prior to the FISH process. The primary
antibody was a mouse monoclonal RING1B (Atsuta et al., 2001) or mouse monoclonal

MEIS2 (Abcam ab55647). Secondary antibodies were anti mouse IgGs coupled with Alexa
Fluor from Life Technologies (355; 10035, 488; A-11001, 555; A21422, 647; A-21235).
After the secondary antibody reaction, sections were postfixed with 4% PFA for 15 minutes,
and then were processed to the cooled 70% ethanol incubation step of the FISH reaction
(Isono et al., 2013).
Fosmids used for FISH analysis
probe name fosmid location (mm9:chr.2)
promoter WI1-703C19 115,882,765-115,924,771
MBE WI1-1321D12 115,795,659-115,836,845
RBS WI1-275J3 115,659,499-115,695,402
The confocal images were captured with an inverted microscope IX71 with oil immersion
objectives (Olympus UPlanSApo 100x NA 1.40 and PlanApo N 60x NA 1.42) as described
previously (Isono et al., 2013). Images with 65 nm pixels in X-Y and 300 nm steps in Z were
deconvoluted one iteration with AutoDeblur®/AutoVisualize® software (AutoQuant Imaging,
Inc.). Deconvoluted images along the Z-axis were analyzed with Volocity software
(Improvision) (Isono et al., 2013).
We determined a threshold promoter/RBS distance to distinguish paired and unpaired foci as
following procedure. Distribution of distances between promoter/RBS FISH signals in
midbrain, LGE, MGE, limb mesenchyme and cephalic mesoderm are summed (Figure S1C;
top panel). The summed distribution was fitted to mixed Gaussian distributions using the EM
algorithm. The numbers of distributions were estimated with Bayesian information criteria
(BIC) so that we applied three normal distributions (cluster 1, 2, and 3). We classified three

clusters of FISH probes using these distributions and determined 0.16µm, which represents
the boundary of cluster 1 and 2, as the threshold to distinguish paired and unpaired signals.
BAC recombineering and production of BAC transgenic mice
Each BAC was modified by insertion of eGFP in-frame from the Meis2 initiation codon by
BAC recombineering (GENE BRIDGES). The deleted sequence of the delta MBE BAC
corresponds to 37,329,745-37,330,178 of human chromosome 15 (hg19) whose mouse
corresponding sequence is located at 115826623-115827057 of chromosome 2 (mm9) and
has 97.7 % sequence identity. Embryos were isolated at 11.5 dpc. BAC insertions were tested
with 4 pairs of PCR primers listed below.
BAC typing primer (human sequence positions are based on hg19:chr.15)
forward sequence position from to reverse sequence position from to
EGFP-140 f gttcatctgcaccaccggc Gfp Gfp EGFP-424 r ttgtgccccaggatgttgc Gfp Gfp
G-Bf agcttgccgtaggtggcatc Gfp Gfp G-B r cttcgttgtctagactgggc 37,391,824 37,391,843
hMeis2B1f aggttgctgaggttcagagc 37,321,049 37,321,068 hMeis2B1r ctagaggagcttctaggatc 37,320,701 37,320,720
hMeis2B2f ttgtcaccaccttagcgtcc 37,224,146 37,224,165 hMeis2B2r gccctgtgacattgttgcac 37,223,770 37,223,789
3C (chromosomal conformation capture)
The 3C technique was performed as described previously (Splinter et al., 2004) with slight
modifications. 106 to 5 x 106 cells were used for 3C analysis, and chromatin was digested
with 400U of the EcoRI restriction enzyme. Primers used for 3C are summarized below.
Quantification of ligation products was based on standard curves made using the cloned
fusion fragments for each expected product. Relative values for 3C were further normalized

by 3C values obtained from the results using the ERCC3 allele in each experiment.
Primer used for 3C analysis
primer name sequence promoter gtttgtcccacactccagcccttg
sequence C acggattgaagctggtcgctcttc sequence V gaagagaaaggcgactcagtctc Ercc3-1-r gcttgagcatgacgtcactg Ercc3-2-r gctgagatcaaggatgaggc
SUPPLEMENTAL REFERENCE Atsuta, T., Fujimura, S., Moriya, H., Vidal, M., Akasaka, T., and Koseki, H. (2001). Production of monoclonal antibodies against mammalian Ring1B proteins. Hybridoma 20, 43-46. Chambeyron, S., Da Silva, N.R., Lawson, K.A., and Bickmore, W.A. (2005). Nuclear re-organisation of the Hoxb complex during mouse embryonic development. Development 132, 2215-2223. del Mar Lorente, M., Marcos-Gutierrez, C., Perez, C., Schoorlemmer, J., Ramirez, A., Magin, T., and Vidal, M. (2000). Loss-and gain-of- function mutations show a Polycomb group function for Ring1A in mice. Development 127, 5093-5100. De Napoles, M., Merrnoud, J.E., Wakao, R., Tang, Y.A., Endoh, M., Appanah, R., Nesterova, T.B., Silva, J., Otter, A.P., Vidal, M., Koseki, H., and Brockdorff, N. (2004). Polycomb group proteins Ring1A/B link ubiquitylation of histone H2A to heritable gene silencing and X inactivation. Dev. Cell 7, 663-676. Isono, K., Endo, T.A., Ku, M., Yamada, D., Suzuki, R., Sharif, J., Ishikura, T., Toyoda, T., Bernstein, B.E., and Koseki, H. (2013). SAM domain polymerization links subnuclear clustering of PRC1 to gene silencing. Dev. Cell 26, 565-577. Kondo, T., and Duboule, D. (1999). Breaking colinearity in the mouse HoxD complex. Cell 97, 407-417. Sasaki, H. and Hogan, B.L.M. (1996). Enhancer analysis of HNF-3b gene: regulatory elements for node/notochord and floor plate are independent and consist of multiple sub-elements. Genes to Cells 1, 59-72. Splinter, E., Grosveld, F., and de Laat, W. (2004). 3C technology: analyzing the spatial

organization of genomic loci in vivo. Methods Enzymol. 375, 493-507. Tanemura, K., Ogura, A., Cheong, C., Gotoh, H., Matsumoto, K., Sato, E., Hayashi, Y., Lee, H.W., and Kondo, T., (2005). Dynamic rearrangement of telomeres during spermatogenesis in mice. Dev. Biol. 281, 196-207.
Related Documents