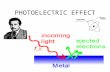
Photoelectric Effect

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript
Photoelectric Effect
formation and
liberation of
electrically charged
particles in matter
when it is irradiated
by light or other
electromagnetic
radiation.
photo for light and electric for the current
The photoelectric effect was first observed in 1887 by Heinrich Hertz (1857-1894) during experiments with a spark-gap generator — the earliest form of radio receiver.
Later studies by J. J. Thomson (1856-1940) showed that this increased sensitivity was the result of light pushing on electrons (which he discovered in 1897).
f
In the research, Thomson enclosed a metal plate
(a cathode) in a vacuum tube, and exposed it to
high frequency radiation. It was thought that the
oscillating electromagnetic fields caused the
atoms' field to resonate and, after reaching a
certain amplitude, caused a subatomic
"corpuscle" to be emitted, and current to be
detected.
-The amount of this current varied with the
intensity and color of the radiation. Larger
radiation intensity or frequency would produce
more current.
Ejected Electrons
Under the right circumstances light can be used to push
electrons, freeing them from the surface of a solid.
This process is called the photoelectric
effect (or photoelectric emission or photoemission),
a material that can exhibit this phenomena is said to
be photoemissive, and the ejected electrons are
called photoelectrons
Lenard’s Observations -In 1902, Philipp von Lenard observed the variation in electron energy
with light frequency. He used a powerful electric arc lamp which enabled him to investigate large changes in intensity, and had sufficient power to enable him to investigate the variation of potential with light frequency.
-His experiment directly measured potentials, not electron kinetic energy: he found the electron energy by relating it to the maximum stopping potential (voltage) in a phototube. He found that the calculated maximum electron kinetic energy is determined by the frequency of the light.
-increase in frequency results in an increase in the maximum kinetic energy calculated for an electron upon liberation - ultraviolet radiation would require a higher applied stopping potential to stop current in a phototube than blue light. However Lenard's results were qualitative rather than quantitative because of the difficulty in performing the experiments
-The current emitted by the surface was determined by the light's intensity, or brightness: doubling the intensity of the light doubled the number of electrons emitted from the surface.
Threshold Frequency
Minimum amount of freq. that is contained
by light.
Red light – Low Energy
Blue right – High Energy
Automatic Doors
Many elevators and garage-door systems use a beam of light and a photoelectric device known as a photocell as a safety feature. As long as the beam of light strikes the photocell, the photoelectric effect generates enough ejected electrons to produce a detectable electric current. (Walker, 988) When the light beam is blocked (by a person) the electric current is interrupted and the doors are signaled to open.
• Solar Energy Panels
Photocells are also the basic unit in the solar energy panels that convert some of the energy in sunlight into electrical energy. These panels are able to operate billboards and safety lights in remote areas far from power lines (Walker, 988). Large solar panels (240 ft.) will power the International Space Station.
Night Vision Device
The Photoelectric effect has numerous applications, for example night vision devices take advantage of the effect. Photons entering the device strike a plate which causes electrons to be emitted, these pass through a disk consisting of millions of channels, the current through these are amplified and directed towards a fluorescent screen which glows when electrons hit it. In these devices an optical image incident on a semitransparent photocathode is used to transform the light image into an “electron image.”
Related Documents