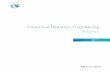
CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 1 PART A Energetics in Chemical Reaction USEFUL CONCEPTS AND FORMULAS 1. )G o (reaction) = 3 n )G o f (products) ! 3 n )G o f (reactants) )G o < 0, the forward reaction occurs spontaneous. )G o = 0, the reaction system is at equilibrium. )G o > 0, the forward reaction is nonspontaneous. 2. )G = )H ! T )S )G depends on T, but )H and )S dont. For a phase transition, )G = 0 3. )S (reaction) = 3 n )S (products) ! 3 n )S (reactants) )S > 0, more random (disorder) in products than in reactants S gas > S liquid > S solid 4. Effect of Temperature on the Spontaneity of Reaction )H )S Temperature )G = )H ! T )S Spontaneous direction s r all always s forward s s low s forward high r reverse r r low r reverse high s forward r s all always r reverse

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 1
PART A Energetics in Chemical Reaction
USEFUL CONCEPTS AND FORMULAS
1.)Go (reaction) = 3 n )Go
f (products) ! 3 n )Gof (reactants)
)Go < 0, the forward reaction occurs spontaneous. )Go = 0, the reaction system is at equilibrium. )Go > 0, the forward reaction is nonspontaneous.
2.)G = )H ! T )S )G depends on T, but )H and )S don�t.
For a phase transition, )G = 0
3.)S (reaction) = 3 n )S (products) ! 3 n )S (reactants)
)S > 0, more random (disorder) in products than in reactantsSgas > Sliquid > Ssolid
4. Effect of Temperature on the Spontaneity of Reaction
)H )S Temperature )G = )H ! T )S Spontaneous direction
s r all always s forward
s s low s forwardhigh r reverse
r r low r reversehigh s forward
r s all always r reverse

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 2
Compare to a straight-line linear equation:y = m x + b
where y = Rn Keqx = 1 / Tm = slope = ! )Ho / Rb = y-intercept = )So / R
5. Under non-standard state conditions,
)G = )Go + R T Rn Q
6. At equilibrium, Q = Keq and )G = 0OR
)Go = ! R T Rn Keq
Keq = Kc for aqueous reactants and products in terms of mol L!1 (M) Keq = Kp for gaseous reactants and products in terms of atm
)Go < 0, Rn Keq > 0, Keq > 1 the products are favoured over reactants(i.e. the forward reaction proceeds spontaneously)
)Go = 0, Rn Keq = 0, Keq = 1 the reaction is at equilibrium )Go > 0, Rn Keq < 0, Keq < 1 the reactants are favoured over products
(i.e. the reverse reaction is spontaneous)
Q < Keq, the reaction proceeds in the forward direction spontaneously Q = Keq, the reaction is at equilibrium Q > Keq, the reaction favours the reverse direction.
7. where equilibrium constant K1 at temperature T1
equilibrium constnat K2 at temperature T2
8.

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 3
PART AQUESTION 1
Using tabulated Thermodynamic data, To find: )Go, )Ho and )So at 298 K To verify: )Go = )Ho ! T )So
To determine: conditions of spontaneous reaction
(i) 4 HCl(g) + O2(g) ÷ 2 H2O(g) + 2 Cl2(g)
(a) )Go (reaction) = 3 n )Gof (products) ! 3 n )Go
f (reactants)= {2× )Go
f [H2O(g)] + 2× )Gof [Cl2(g)]}
! {4× )Gof [HCl(g)] + )Go
f [O2(g)]}= {2× (228.59) + 2× (0)} ! {4× (95.30) + (0)} kJ= !75.98 kJ
(b) )Ho (reaction) = 3 n )Hof (products) ! 3 n )Ho
f (reactants)= {2× )Ho
f [H2O(g)] + 2× )Hof [Cl2(g)]}
! {4× )Hof [HCl(g)] + )Ho
f [O2(g)]}= {2× (!241.82) + 2× (0)} ! {4× (!92.31) + (0)} kJ= !114.40 kJ
)So (reaction) = 3 n )So (products) ! 3 n )So (reactants)= {2× )So [H2O(g)] + 2× )So [Cl2(g)]}
! {4× )So [HCl(g)] + )So [O2(g)]}= {2× (222.96) + 2× (188.72)} ! {4× (186.80) + (205.03)} J K!1
= !128.87 J K!1
At 298 K, )Go = )Ho ! T )So
= (!114.40 kJ) ! (298 K) (!128.87 × 10!3 kJ K!1)= !76.0 kJ
In comparison to Part (a), )Go is not absolutely equal since we assumed )Ho
and )So to be temperature independent.
(c) Since )Go < 0 (i.e. !75.98 kJ), the reaction is spontaneous under standard conditions at298K (i.e. 25oC). According to )Go = )Ho ! T )So, the reaction is:
(1) favoured by negative )Ho (i.e. enthalpy driven, !114.40 kJ)(2) disfavoured by negative )So (!128.87 J K!1)
(d) )Go is negative and the reaction is spontaneous up to the temperature at which )Ho = T )So.ˆ T = )Ho ÷ )So = (!114.40 kJ) ÷ (!128.87 × 10!3 kJ K!1) = 887.7 K
Thus, the reaction is spontaneous at T < 888 K.

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 4
(ii) H2(g) + Cl2(g) ÷ 2 HCl(g)
(a) )Go (reaction) = 3 n )Gof (products) ! 3 n )Go
f (reactants)= {2× )Go
f [HCl(g)]} ! {)Gof [H2(g)] + )Go
f [Cl2(g)]}= {2× (!95.3)} ! {(0) + (0)} kJ= !190.6 kJ
(b) )Ho (reaction) = 3 n )Hof (products) ! 3 n )Ho
f (reactants)= {2× )Ho
f [HCl(g)]} ! {)Hof [H2(g)] + )Ho
f [Cl2(g)]}= {2× (!92.31)} ! {(0) + (0)} kJ= !184.62 kJ
)So (reaction) = 3 n )So (products) ! 3 n )So (reactants)= {2× )So [HCl(g)]} ! {)So [H2(g)] + )So [Cl2(g)]}= {2× (186.8)} ! {(130.6) + (222.9)} J K!1
= +20.1 J K!1
At 298 K, )Go = )Ho ! T )So
= (!184.62 kJ) ! (298 K) (20.1 × 10!3 kJ K!1)= !190.6 kJ
(c) Since )Go < 0 (i.e. !190.6 kJ), the reaction is spontaneous under standard conditions at298 K (i.e. 25oC). According to )Go = )Ho ! T )So, the reaction is:
(1) favoured by negative )Ho (i.e. enthalpy driven, !184.62 kJ)(2) favoured by positive )So (i.e. entropy driven, +20.1 J K!1)
(d) Since )Go is always negative, the reaction occurs spontaneously at all temperatures.
(iii) C(graphite) ÷ C(diamond)
(a) )Go (reaction) = 3 n )Gof (products) ! 3 n )Go
f (reactants)= {)Go
f [C(diamond)]} ! {)Gof [C(graphite)]}
= {(2.900)} ! {(0)} kJ= +2.900 kJ
(b) )Ho (reaction) = 3 n )Hof (products) ! 3 n )Ho
f (reactants)= {)Ho
f [C(diamond)]} ! {)Hof [C(graphite)]}
= {(1.895)} ! {(0)} kJ= +1.895 kJ
)So (reaction) = 3 n )So (products) ! 3 n )So (reactants)

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 5
= {)So [C(diamond)]} ! {)So [C(graphite)]}= {(2.377)} ! {(5.74)} J K!1
= !3.363 J K!1
At 298 K, )Go = )Ho ! T )So
= (1.895 kJ) ! (298 K) (!3.363 × 10!3 kJ K!1)= +2.897 kJ
(c) Since )Go > 0 (i.e. +2.900 kJ) at 298 K, the reaction is not spontaneous as written underthe standard conditions.
)Ho is positive (+1.895 kJ): unfavoured )Go being negative)So is negative (!3.363 J K!1): unfavoured )Go being negative
(d) )Go is always positive at any temperatures. Thus, the reaction proceeds nonspontaneouslyat any temperatures.
PART AQUESTION 2
For a spontaneous reaction to occur, )Go must be negative.)Go = )Ho ! T )So
)Ho = !ve: favours s)Go (spontaneous))So = +ve: favours s)Go (spontaneous)
(a) a reaction with )Ho = +53.4 kJ and )So = +112.4 J K!1
Since both )Ho and )So are positive (i.e. unfavoured enthalpy and favoured entropy),the reaction is spontaneous above the temperature at which )Ho = T )So.
ˆ T > )Ho ÷ )So = (+53.4 kJ) ÷ (+112.4 × 10!3 kJ K!1) = 475 KThus, the reaction is spontaneous at T > 475 K.
(b) a reaction with )Ho = !29.4 kJ and )So = !91.2 J K!1
Since both )Ho and )So are negative (i.e. favoured enthalpy and unfavoured entropy),the reaction is spontaneous up to the temperature at which )Ho = T )So.
ˆ T < )Ho ÷ )So = (!29.4 kJ) ÷ (!91.2 × 10!3 kJ K!1) = 322 KThus, the reaction is spontaneous at T < 322 K.

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 6
Î For a spontaneous reaction to occur, )Go must be negative.Ï Only reactions having like signs for both )Ho and )So exhibit temperature-
dependent spontaneity (‡ one term is unfavourable).)Go = )Ho ! T )So
when )Ho = !ve and )So = !veat low T: )Go = sve (spontaneous forward reaction)at high T: )Go = rve (nonspontaneous forward reaction)
when )Ho = +ve and )So = +veat low T: )Go = rve (nonspontaneous forward reaction) at high T: )Go = sve (spontaneous forward reaction)
PART AQUESTION 3
Wanted: a spontaneous reaction (i.e. )Go < 0) when a forward reaction occurs at low temperatures a reverse reaction occurs at high temperatures (i.e. a forward reaction occurs nonspontaneous at high temperatures)
To find: signs of )Ho and )So for such a reaction
(1) At low temperatures, the forward reaction proceeds spontaneously.ˆ )Go = !ve
It means )Ho term is dominant with the negative sign (i.e. favourable enthalpy change).
(2) At high temperatures, the forward reaction becomes nonspontaneous.It indicates sT )So term is dominant with the positive sign.(i.e. implying )S < 0, unfavourable entropy change).
ˆ Both )Ho and )So for such a reaction are negative.
PART AQUESTION 4
)Go depends on temperatures, but )Ho and )So don�t.
Strategy: Î Apply the Thermodynamics table to calculate )Ho(reaction) and )So(reaction) at 25oC. Ï Use the equation: )Go = )Ho ! T )So to calculate )Go at different temperatures. Ð Then use the equation: )Go = ! R T Rn Keq to calculate Kp at various temperatures.

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 7
(a) CS2(R) at 5oC (278 K) CS2(R) º CS2(g) Keq = Kp = p(CS2,g)
)Ho (reaction) = 3 n )Hof (products) ! 3 n )Ho
f (reactants)= {)Ho
f [CS2(g)]} ! {)Hof [CS2(R)]}
= {(117.36)} ! {(89.70)} kJ mol!1
= +27.66 kJ mol!1
)So (reaction) = 3 n )So (products) ! 3 n )So (reactants)= {)So [CS2(g)]} ! {)So [CS2(R)]}= {(237.73)} ! {(151.34)} J K!1 mol!1
= +86.39 J K!1 mol!1
ˆ )Go (278 K) = )Ho ! T )So = (27.66 kJ) ! (278 K) (86.39 × 10!3 kJ K!1) = +3.64 kJ
At 278 K , )Go = ! R T Rn Keq
3.64 kJ mol!1 = ! (8.314 × 10!3 kJ mol!1 K!1) (278 K) Rn Kp
ˆ Kp = 0.207 atm = p(CS2,g)
(b) CCl4(R) at 29oC (302 K) CCl4(R) º CCl4(g) Keq = Kp = p(CCl4,g)
)Ho (reaction) = 3 n )Hof (products) ! 3 n )Ho
f (reactants)= {)Ho
f [CCl4(g)]} ! {)Hof [CCl4(R)]}
= {(!102.9)} ! {(!135.44)} kJ mol!1
= +32.54 kJ mol!1
)So (reaction) = 3 n )So (products) ! 3 n )So (reactants)= {)So [CCl4(g)]} ! {)So [CCl4(R)]}= {(309.74)} ! {(216.40)} J K!1 mol!1
= +93.34 J K!1 mol!1
ˆ )Go (302 K) = )Ho ! T )So = (32.54 kJ) ! (302 K) (93.34 × 10!3 kJ K!1) = +4.35 kJ
At 302, )Go = ! R T Rn Keq
4.35 kJ mol!1 = ! (8.314 × 10!3 kJ mol!1 K!1) (302 K) Rn Kp
ˆ Kp = 0.177 atm = p(CCl4,g)

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 8
(c) CH3CHO(R) at 45oC (318 K)
CH3CHO(R) º CH3CHO(g) Keq = Kp = p(CH3CHO,g)
)Ho (reaction) = 3 n )Hof (products) ! 3 n )Ho
f (reactants)= {)Ho
f [CH3CHO(g)]} ! {)Hof [CH3CHO(R)]}
= {(!166.19)} ! {(!192.30)} kJ mol!1
= +26.11 kJ mol!1
)So (reaction) = 3 n )So (products) ! 3 n )So (reactants)= {)So [CH3CHO(g)]} ! {)So [CH3CHO(R)]}= {(250.2)} ! {(160.2)} J K!1 mol!1
= +90.0 J K!1 mol!1
ˆ )Go (318 K) = )Ho ! T )So = (26.11 kJ) ! (318 K) (90.0 × 10!3 kJ K!1) = !2.51 kJ
At 318 K, )Go = ! R T Rn Keq
!2.51 kJ mol!1 = ! (8.314 × 10!3 kJ mol!1 K!1) (318 K) Rn Kp
ˆ Kp = 2.58 atm = p(CH3CHO,g)
PART AQUESTION 5
Given: Reaction studied: CH3CH2OH(s) ÷ CH3CH2OH(R))Ho
rxn = 4.60 kJ mol!1
)Sorxn = +29.7 J K!1 mol!1 = +29.7 × 10!3 J K!1 mol!1
To find: freezing point for CH3CH2OH
For a phase transition, )G = 0 (i.e. the (s) and (R) phases coexist in equilibrium)
From )G = )H ! T )S,
Assume )H and )S are independent of temperatures, )Hfus . )Horxn and )Sfus . )So
rxn

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 9
)Go depends on temperatures, but )Ho and )So don�t. Strategy: Î Use the Thermodynamic table to calculate )Ho(reaction) and )So(reaction) at 25oC. Ï Apply the equation: )Go = )Ho ! T )So to calculate )Go at 100oC. Ð Then use the equation: )Go = ! R T Rn Keq to calculate Kc at 100oC.
Note that: Kc = [H+] [OH!]
For comparison: At 25oC, Kc = Kw = 1.0 × 10!14 M2
At 100oC, Kc = 9.34 × 10!13 M2
PART AQUESTION 6
Given: H2O(R) ÷ H+(aq) + OH!(aq)From the Thermodynamic table at 25oC,)Ho
f [H+(aq)] = 0 )So [H+(aq)] = 0
)Hof [OH!(aq)] = !229.94 J mol!1 )So [OH!,aq] = !10.5 J K!1 mol!1
)Hof [H2O(R)] = !285.84 J mol!1 )So
[H2O(R)] = 69.94 J K!1 mol!1
To find: Kc for the reaction at 100oC
Î )Ho (reaction) = 3 n )Hof (products) ! 3 n )Ho
f (reactants)= {)Ho
f [H+(aq)] + )Hof [OH!(aq)]} ! {)Ho
f [H2O(R)]}= {(0) + (!229.94)} ! {(!285.84)} kJ mol!1
= +55.90 kJ mol!1
)So (reaction) = 3 n )So (products) ! 3 n )So (reactants)= {)So [H+(aq)] + )So [OH!(aq)]} ! {)So [H2O(R)]}= {(0) + (!10.5)} ! {(69.94)} J K!1 mol!1
= !80.44 J K!1 mol!1
Ï At 100oC (i.e. 373 K),
)Go = )Ho ! T )So
= (+55.90 kJ mol!1) ! (373 K) (!80.44 × 10!3 kJ K!1 mol!1) = +85.90 kJ mol!1
Ð At 100oC (i.e. 373 K),
)Go = ! R T Rn Keq
+85.90 kJ mol!1 = ! (8.314 × 10!3 kJ mol!1 K!1) (373 K) Rn Kc
ˆ Kc = 9.3367 × 10!13 M2
= 9.34 × 10!13 M2

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 10
PART AQUESTION 7
Given: At 298 K, the reaction CO2(g) º CO2(aq) has:
)Ho = !23.0 kJ mol!1
)Gof [CO2(g)] = !394.0 kJ mol!1
)Gof [CO2(aq)] = !386.0 kJ mol!1
(a) Henry's Law Constant, KH, for CO2(g) at 298 K
)Go (reaction) = 3 n )Gof (products) ! 3 n )Go
f (reactants)= {)Go
f [CO2(aq)]} ! {)Gof [CO2(g)]}
= {(!386.0)} ! {(!394.0)} kJ mol!1
= +8.0 kJ mol!1
CO2(g) º CO2(aq)
At 298 K, )Go = ! R T Rn Keq
+8.0 kJ mol!1 = ! (8.314 × 10!3 kJ mol!1 K!1) (298 K) Rn KH
ˆ KH = 3.96 × 10!2 M atm!1
(b) )So for the reaction at 298 K
)Go = )Ho ! T )So
+8.0 kJ mol!1 = (!23.0 kJ mol!1) ! (298 K) )So
ˆ )So = !0.104 kJ K!1 mol!1
= !104 J K!1 mol!1
The reaction is:(1) favoured by negative )Ho (!23.0 kJ mol!1)(2) unfavoured by negative )So (!0.104 kJ K!1 mol!1)
The reaction occurs spontaneously up to the temperature at which )Ho = T )So.ˆ T < )Ho ÷ )So = (!23.0 kJ mol!1) ÷ (!0.104 kJ K!1 mol!1) = 221 K (!52oC)
Thus, the reaction is only spontaneous at T < 221 K, but not at T = 298 K (where )Go = +8.0 kJ mol!1).

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 11
)Go depends on temperatures, but )Ho and )So don�t.
ˆ )Go at 298 K will be somewhat different from )Go at 2000 K.
Strategy: Î Use the Thermodynamic table to calculate )Ho(reaction) and )So(reaction) at 25oC. Ï Apply the equation: )Go = )Ho ! T )So to calculate )Go at 2000K. Ð Then use the equation: )Go = ! R T Rn Keq to calculate Kp at 2000K.
PART AQUESTION 8
Given: For the reaction: H2(g) + ½ O2(g) ÷ H2O(R)
At 25oC )Hof (kJ mol!1) )Go
f (kJ mol!1) )So (J K!1 mol!1)H2(g) 0 0 130.6O2(g) 0 0 205H2O(R) !285.8 !237.2 69.9
(a) )Go and Kp at 298 K
)Go (reaction) = 3 n )Gof (products) ! 3 n )Go
f (reactants)= {)Go
f [H2O(R)]} ! {)Gof [H2(g)] + ½ )Go
f [O2(g)]}= {(!237.2)} ! {(0) + ½ (0)} kJ mol!1
= !237.2 kJ mol!1
At 298 K,)Go = ! R T Rn Keq
!237.2 kJ mol!1 = ! (8.314 × 10!3 kJ mol!1 K!1) (298 K) Rn Kp
ˆ Kp = 3.79 × 1041 atm!3/2
Since )Go is negative (i.e. !237.2 kJ mol!1) and Kp is >> 1 at 298 K, the reactionoccurs spontaneously as written.
H2(g) + ½ O2(g) ! H2O(R)
(b) )Go and Kp at 2000 K

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 12
Î )Ho (reaction) = {)Hof [H2O(R)]} ! {)Ho
f [H2(g)] + ½ )Hof [O2(g)]}
= {(!285.8)} ! {(0) + ½ (0)} kJ mol!1
= !285.8 kJ mol!1
)So (reaction) = {)So [H2O(R)]} ! {)So
[H2(g)] + ½ )So [O2(g)]}
= {(69.9)} ! {(130.6) + ½ (205)} J K!1 mol!1
= !163.2 J K!1 mol!1
Ï At 2000 K, )Go = )Ho ! T )So
= (!285.8 kJ mol!1) ! (2000 K) (!163.2 × 10!3 kJ K!1 mol!1)= +40.6 kJ mol!1
Ð At 2000 K, )Go = ! R T Rn Keq
40.6 kJ mol!1 = ! (8.314 × 10!3 kJ mol!1 K!1) (2000 K) Rn Kp
ˆ Kp = 8.70 × 10!2 atm!3/2
The reaction H2(g) + ½ O2(g) ÷ H2O(R) is temperature-dependent because it is:(1) favoured (driven) by negative )Ho (!285.8 kJ mol!1)(2) unfavoured by negative )So (!163.2 J K!1 mol!1)
The reaction proceeds spontaneous up to the temperature at which )Ho = T )So
ˆ T = )Ho ÷ )So = (!285.8 kJ mol!1) ÷ (!163.2 × 10!3 kJ K!1 mol!1) = 1751 KAt 298 K (i.e. T < 1751 K), )Go = s237.2 kJ mol!1 (spontaneous reaction)At 2000 K (i.e. T > 1751 K), )Go = r40.6 kJ mol!1 (nonspontaneous reaction)
(c) )G at 2000 K when H2(g) and O2(g) are at 10.0 atm pressure each
Under non-standard state condition, p(H2) = p(O2) = 10.0 atm
)G = )Go + R T Rn Q
ˆ )G = [40.6 + (!57.43)] kJ mol!1 = !16.83 kJ mol!1 = !16.8 kJ mol!1
Since )G is negative (i.e. !16.8 kJ mol!1), the reaction proceeds spontaneously from LEFTto RIGHT as written (i.e. the forward reaction is favourable).

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 13
PART AQUESTION 9
Given: CuSO4AH2O(s, white) + 4 H2O(g) ÷ CuSO4A5H2O (s, blue)
At 298 K )Hof (kJ mol!1) So (J K!1 mol!1)
CuSO4AH2O(s, white) !1084 150CuSO4A5H2O(s, blue) !2278 305H2O(g) !242 189
(a) )G for the reaction at 15oC when p(H2O,g) = 0.025 atm
)Go depends on temperatures. ˆ )Go (at 15oC) … )Go (at 25oC).
)Ho (reaction) = 3 n )Hof (products) ! 3 n )Ho
f (reactants)= {)Ho
f [CuSO4A5H2O(s, blue)]} ! {)Hof [CuSO4AH2O(s, white)] + 4× )Ho
f [H2O(g)]}= {(!2278)} ! {(!1084) + 4× (!242)} kJ mol!1
= !226 kJ mol!1
)So (reaction) = 3 n )So (products) ! 3 n )So (reactants)= {)So
[CuSO4A5H2O(s, blue)]} ! {)So [CuSO4AH2O(s, white)] + 4× )So
[H2O(g)]= {(305)} ! {(150) + 4× (189)} J K!1 mol!1
= !601 J K!1 mol!1
At 15oC (i.e. 288 K), )Go = )Ho ! T )So
= (!226 kJ mol!1) ! (288 K) (!601 × 10!3 kJ K!1 mol!1) = !52.912 kJ mol!1
Under non-standard state conditions, p(H2O,g) = 0.025 atm
)G = )Go + R T Rn Q
ˆ )G = [(!52.912) + (+35.331)] kJ mol!1 = !17.581 kJ mol!1 = !17.6 kJ mol!1
(b) Since )G is negative (!17.6 kJ mol!1), the reaction is spontaneous under non-standard conditionsat 15oC when p(H2O,g) = 0.025 atm (i.e. the blue form is more stable).

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 14
PART AQUESTION
10
Given: AgCl(s) º Ag+(aq) + Cl!(aq)
From the Thermodynamic table,)Go
f [AgCl(s)] = !109.805 kJ mol!1
)Gof [Ag+(aq)] = 77.107 kJ mol!1
)Gof [Cl!(aq)] = !131.244 kJ mol!1
To find: Ksp and the solubility of AgCl(s) at 25oC
AgCl(s) º Ag+(aq) + Cl!(aq) (1:1 electrolyte)
)Go (reaction) = 3 n )Gof (products) ! 3 n )Go
f (reactants)= {)Go
f [Ag+(aq)] + )Gof [Cl!(aq)]} ! {)Go
f [AgCl(s)]}= {(77.107) + (!131.244)} ! {(!109.805)} kJ mol!1
= +55.668 kJ mol!1 (nonspontaneous at 25oC)
ˆ Keq = [Ag+(aq)] [Cl!(aq)] = Ksp [AgCl(s)]
At 25oC (i.e. 298 K),
)Go = ! R T Rn Keq
+55.668 kJ mol!1 = ! (8.314 × 10!3 kJ mol!1 K!1) (298 K) Rn Ksp [AgCl(s)]ˆ Ksp [AgCl(s)] = 1.746 × 10!10 M2
= 1.75 × 10!10 M2
Ksp [AgCl(s)] = [Ag+(aq)] [Cl!(aq)]1.746 × 10!10 M2 = (s) (s)
ˆ s = (1.746 × 10!10 M2)½
= 1.321 × 10!5 M = 1.32 × 10!5 M
ˆ The solubility of AgCl(s) at 25oC is: 1.32 × 10!5 M

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 15
PART AQUESTION
11
Given: For the reaction: 2 H2O(R) º H3O+(aq) + OH!(aq)
Kw = 1.0 × 10!14 M2 at 25oC)H = 55.8 kJ mol!1 (endothermic)
(a) Kw at 100oC
When T1 = 25oC (i.e. 298 K), K1 = Kw = 1.0 × 10!14 M2
When T2 = 100oC (i.e. 373 K), K2 = ?
ˆ K2 = Kw at 100oC = 9.262 × 10!13 M2 = 9.3 × 10!13 M2
(b) pH of boiling water
Since Kw = [H3O+] × [OH!]and [H3O+] = [OH!]
Kw (at 100oC) = [H3O+] × [OH!] = [H3O+]2
9.262 × 10!13 M2 = [H3O+]2
ˆ [H3O+] = 9.624 × 10!7 M
pH = ! log [H3O+]= ! log (9.624 × 10!7 M)= 6.0166= 6.02

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 16
PART AQUESTION
12
Given: CO(g) + Cl2(g) ÷ COCl2(g)at 506oC, Keq = 1.3at 530oC, Keq = 0.78
(a) )Ho and )So for the reaction
T1 = 506oC = (506 + 273) K = 779 KK1 = 1.3
T2 = 530oC = (530 + 273) K = 803 KK2 = 0.78
ˆ )Ho = !110.7 kJ mol!1 = !1.1 × 102 kJ mol!1 (endothermic reaction)
To calculate )So, you must first calculate )Go using the equation: )Go = ! R T Rn KeqYou can calculate )Go either at 506oC or at 530oC.
At 506oC (i.e. 779 K), )Go = ! R T Rn Keq
= ! (8.314 × 10!3 kJ mol!1 K!1) (779 K) Rn (1.3)= !1.6992 kJ mol!1
)Go = )Ho ! T )So
!1.6992 kJ mol!1 = (!110.7 kJ mol!1) ! (779 K) )So
ˆ )So = !0.140 kJ K!1 mol!1
= !1.4 × 102 J K!1 mol!1
(b) Under standard-state conditions, over what temperature range is the reaction spontaneous?
)Ho = !110.7 kJ mol!1 (favoured the reaction for )Go being negative))So = !0.14 kJ K!1 mol!1 (disfavoured the reaction for )Go being negative)
The reaction is spontaneous at temperature below which )Ho > T )So
ˆ T < )Ho ÷ )So = (!110.7 kJ mol!1) ÷ (!0.14 kJ K!1 mol!1) = 790.7 K
Thus, the reaction is spontaneous when T < 791 K.

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 17
In comparison, )Go (not )G) at 25oC (i.e. 298 K) is calculated as follows. Î Kp must be expressed in atm. Ï Since 1 atm = 760 torr = 760 mmHg
23.76 torr = (23.76 torr ÷ 760 torr atm!1) = 3.1263 × 10!2 atm = Kp = p(H2O,g) Ð )Go (at 298 K) = ! R T Rn Kp
= ! (8.314 × 10!3 kJ mol!1 K!1) (298 K) Rn (3.1263 × 10!2)= +8.59 kJ mol!1 (nonspontaneous)
PART AMC #1
Given: H2O(R) ÷ H2O(g)p(H2O) = 23.76 torr (at 25oC)
To find: )G at 25oC
This reaction is a phase transition process from H2O(R) to H2O(g) (i.e. vaporization).It indicates the reaction is at equilibrium, since the gas pressure equals the vapour pressure atthe temperature interest.
ˆ )G = 0
PART AMC #2
Given: For the reaction: HCO2H(R) ÷ HCO2H(g) at 298 K)Ho = 46.60 kJ)Go = 10.3 kJ)So = 122 J K!1 = 122 × 10!3 kJ K!1
To find: the normal boiling point of HCO2H(R)
At normal boiling point, the transition phase process from (R) to (g) is at equilibrium.Thus, )G = 0.
From )G = )H ! T )S,
Assume )Ho and )So are independent of temperatures (ˆ )Hvap . )Ho and )Svap . )So)
ˆ Tb.p. (HCO2H) = 382 K ! 273 K = 109oC

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 18
PART AMC #3
Given: Reaction studied: Hg(R) ÷ Hg(g)Kp = p(Hg,g) = 36.38 Pa (Note that Kp must be in terms of atm.)T = 100oC = (100 + 273) K = 373 K
To find: )Go of vaporization of Hg(R) at 100oC
Since 1 atm = 101.3 kPa = 101.3 × 103 Pa, 36.38 Pa = (36.38 Pa ÷ 101.3 × 103 Pa atm!1) = 3.5913 × 10!4 atm
Keq = Kp = p(Hg,g) = 3.5913 × 10!4 atm
At 100oC (i.e. 373 K), )Go = ! R T Rn Kp= ! (8.314 × 10!3 kJ mol!1 K!1) (373 K) Rn (3.591 × 10!4)= +24.59 kJ mol!1
= +24.6 kJ mol!1 (nonspontaneous at 100oC)
PART AMC #4
1. Entropy (disorder) decreases: Sgas > Sliquid > Ssolid
2. )n (g) = 3 n (gaseous products) ! 3 n (gaseous reactants)
)S = +ve, when )n(g) = +ve3 n (gaseous products) > 3 n (gaseous reactants)(i.e. randomness increases and entropy increases)
)S = !ve, when )n(g) = !ve3 n (gaseous products) < 3 n (gaseous reactants)(i.e. disorder decreases and entropy decreases)
(a) CO2(g) ÷ CO2(s) INCORRECT
(‡ Sgas ÷ Ssolid; more order; ˆ )Sorxn = !ve )
(b) NH4+(aq) + NO3
!(aq) ÷ NH4NO3(s) INCORRECT
(‡ Sliquid ÷ Ssolid; more order; ˆ )Sorxn = !ve)
(c) Zn2+(aq) + H2(g) ÷ Zn(s) + 2 H+(aq) INCORRECT
(‡ 1 mole gas ÷ no gases; )n(g) = !1; more order; ˆ )Sorxn = !ve)
(d) C6H6(R) + 9/2 O2(g) ÷ 6 CO2(g) + 3 H2O(R) TRUE(‡ 4.5 moles gas ÷ 6 moles gas; )n(g) = +1.5; more disorder; ˆ )So
rxn = +ve)(e) 5 CO2(g) + 6 H2O(g) ÷ C5H12(g) + 8 O2(g) INCORRECT
(‡ 11 moles gas ÷ 9 moles gas; )n(g) = !2; less disorder; ˆ )Sorxn = !ve)

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 19
PART AMC #5
Given: The reaction 2 C(s) + O2(g) ÷ 2 CO(g))H = !ve (i.e. exothermic)
To find: conditions of temperature on reaction spontaneity
For a spontaneous reaction ()G < 0),favourable conditions: Î !ve )H (i.e. exothermic reaction)
Ï +ve )S (i.e. more disorder in products than in reactants)
Î )H = !veÏ )n(g) = 2 moles CO(g) ! 1 mole O2(g) = +1 (ˆ )S = +ve)
)G = )H ! T )S
)G = (s) ! T (r)
‡ Both )H (!ve) and )S (+ve) a re favourable for negative )G.ˆ )G is always negative at all temperatures, and the reaction is spontaneous at all
temperatures and favours the forward reaction.
PART AMC #6
Given: Ag2CrO4(s) º 2 Ag+(aq) + CrO42!(aq)
)Go = +66.2 kJ mol!1 (at 25oC)T = 25oC = (25 + 273) K = 298 K[CrO4
2!] = 2.0 × 10!3 M at equilibrium
To find: [Ag+] at equilibrium
At 25oC (i.e. 298 K), )Go = ! R T Rn Keq
+66.2 kJ mol!1 = ! (8.314 × 10!3 kJ mol!1 K!1) (298 K) Rn Keq
Keq = 2.488 × 10!12 M3
where Keq = [Ag+]2 [CrO42!] = Ksp [Ag2CrO4(s)]
Ksp [Ag2CrO4(s)] = [Ag+]2 [CrO42!]
2.488 × 10!12 M3 = [Ag+]2 (2.0 × 10!3 M)ˆ [Ag+] = 3.527 × 10!5 M = 3.5 × 10!5 M

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 20
PART AMC #7
Given: Reaction studied:2 H2(g) + CO(g) º CH3OH(g)
T1 = 120oC = (120 + 273) K = 393 K K1 = Kp = 0.495 atm!2
T2 = 240oC = (240 + 273) K = 513 K K2 = ?
)Go (at 240oC) = +42.2 kJ mol!1 (nonspontaneous)
To find: )Ho
Î First calculate K2 at 240oC using the equation: )Go = ! R T Rn Keq.
At 240oC (i.e. 513 K), )Go = ! R T Rn Keq
+42.2 kJ mol!1 = ! (8.314 × 10!3 kJ mol!1 K!1) (513 K) Rn K2
ˆ K2 = 5.046 × 10!5 atm!2
Ï Then calculate )Ho using the equation:
ˆ )Ho = !128.38 kJ mol!1 = !128 kJ mol!1 (exothermic reaction)
We assume )Ho is independent of temperature changeand there is relatively no difference in )Ho at 120oC and 240oC.

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 21
Compare to a straight-line linear equation:y = m x + b
where y = Rn (vapour pressure)x = 1 / Tm = slope = ! )Ho / Rb = y-intercept = )So / R
PART AMC #8
Given: Reaction studied: C2H5OH(R) º C2H5OH(g)a plot of Rn (vapour pressure) versus 1/Tslope from a straight line = !5.09 × 103 KR = 8.314 × 10!3 kJ K!1 mol!1
To find: parameter obtained from the slope (i.e. )Ho)
Assuming )Ho is independent of temperatures, )Ho . )Hvap
1 / T
l n
( v a
p o
u r
p r
e s
s u r
e ) ∆ S o / R
s l o p e = - ∆ H o / R
ˆ )Ho = +42.318 kJ mol!1 = +42.3 kJ mol!1 (endothermic reaction)
Since the reaction is a vaporization process from (R) ÷ (g),)Ho . )Hvap = 42.3 kJ mol!1.

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 22
PART B Electrochemistry
USEFUL CONCEPTS AND FORMULAS
1. Oxidizing agent undergoes reduction (GER: Gain Electron(s) = Reduction).! Best oxidizing agent has the largest value of õo
red (i.e. greatest tendency to be reduced).
Reducing agent undergoes oxidation (LEO: Loss Electron(s) = Oxidation).! Best reducing agent has the largest value of õo
ox (i.e. greatest tendency to be oxidized).
2. Reduction occurs at the cathode.(Xn+ + n e! ÷ X where Xn+ is the oxidizing agent)
Oxidation occurs at the anode.(X ÷ Xn+ + n e! where X is the reducing agent)
3. For any cell, õo
cell = õoox + õo
red
(If õo > 0, then the reaction occurs spontaneously.)
4. and )Go = ! n ö õo )Go = ! R T Rn Keq
)Go Keq õocell Reaction under standard-state condition
!ve > 1 +ve spontaneous
0 1 0 at equilibrium
+ve < 1 !ve nonspontaneous
5. Nernst equation (under non-standard state conditions):
At 25oC, (R = 8.314 × 10!3 kJ K!1 mol!1; T = 298 K; ö = 96.5 kJ V!1 mol!1)

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 23
6. At equilibrium, õ = 0
At 25oC, (R = 8.314 × 10!3 kJ K!1 mol!1; T = 298 K; ö = 96.5 kJ V!1 mol!1)
7.charge (Q) = current (i) × time (t) [ Units: C = A × s ]
ö (Faraday Constant) = 96500 C / mol e!
8. Standard Reduction Potentials (SRP, õored) used in this Problem Set:
õored
F2(g) + 2 e! ÷ 2 F!(aq) +2.866 VCl2(g) + 2 e! ÷ 2 Cl!(aq) +1.358 VO2(g) + 4 H+(aq) + 4 e! ÷ 2 H2O(R) +1.229 VBr2(R) + 2 e! ÷ 2 Br!(aq) +1.066 VHg2+(aq) + 2 e! ÷ Hg(R) +0.851 VAg+(aq) + e! ÷ Ag(s) +0.799 VFe3+(aq) + e! ÷ Fe2+(aq) +0.771 VI2(s) + 2 e! ÷ 2 I!(aq) +0.536 VO2(g) + 2 H2O(R) + 4 e! ÷ 4 OH!(aq) +0.401 VCu2+(aq) + 2 e! ÷ Cu(s) +0.337 VAgCl(s) + e! ÷ Ag(s) + Cl!(aq) +0.222 V2 H+(aq) + 2 e! ÷ H2(g) 0Pb2+(aq) + 2 e! ÷ Pb(s) !0.126 VNi2+(aq) + 2 e! ÷ Ni(s) !0.257 VZn2+(aq) + 2 e! ÷ Zn(s) !0.762 V2 H2O(R) + 2 e! ÷ H2(g) + 2 OH!(aq) !0.828 VAl3+(aq) + 3 e! ÷ Al(s) !1.622 VNa+(aq) + e! ÷ Na(s) !2.71 VK+(aq) + e! ÷ K(s) !2.93 V

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 24
Oxidation (loss of e!) occurs at the anode. Reduction (gain of e!) occurs at the cathode.
PART BQUESTION 1
Given: For the cell:Pt(s) , Fe2+(aq), Fe3+(aq) 1 Hg2+(aq) , Hg(R)
Î The anode is on the extreme left of the line (i.e. Pt(s)). Ï The cathode is on the extreme right (i.e. Hg(R)). Ð , is used to denote a change of physical state. Ñ 1 is used to denote the liquid junction.
(a) the anode half-reaction
Oxidation: Fe2+(aq) ÷ Fe3+(aq) + e! õoox = !0.771 V
(b) the cathode half-reaction
Reduction: Hg2+(aq) + 2 e! ÷ Hg(R) õored = +0.851 V
(c) the standard cell potential for the cell
2× Oxidation: 2 Fe2+(aq) ÷ 2 Fe3+(aq) + 2 e! õoox = !0.771 V
Reduction: Hg2+(aq) + 2 e! ÷ Hg(R) õored = +0.851 V
Redox Reaction: 2 Fe2+(aq) + Hg2+(aq) ÷ 2 Fe3+(aq) + Hg(R) õocell = ?
õocell = õo
ox + õored = (!0.771 V) + (+0.851 V) = +0.080 V
Î The anode is Pt(s) and the cathode is Hg(R). Ï Fe2+(aq) acts as reducing agent and oxidizes to Fe3+(aq) under oxidation.
Hg2+(aq) acts as oxidizing agent and reduces to Hg(R) under reduction. Ð Electrons flow from Pt(s) anode to the Hg(R) cathode.
There are two e! being transferred in the balanced redox reaction. Ñ The anions migrate toward the Pt(s) anode through a salt bridge.
The cations diffuse toward the Hg(R) cathode through a salt bridge. Ò Since õo
cell is positive, the reaction is spontaneous under standard conditions. Ó The õo
ox and õored is not affected by the stoichiometric coefficient of a half-cell reaction.

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 25
PART BQUESTION 2
Given: 1 Cu2+(aq, 0.0356 M) , Cu(s)
To find: the potential for the half-cell (õ)
The cell diagram represents the cathode reduction half-reaction:Cu2+(aq) + 2 e! ÷ Cu(s) õo
red = +0.337 V
There are 2 e! being transferred in the reduction half-reaction. (ˆ n = 2)
ˆ õ = (+0.337 V) ! (0.04280 V) = +0.2942 V = +0.294 V
PART BQUESTION 3
Given: Zn(s) , Zn2+(aq, 0.322 M) 1 Cu2+(aq, 0.0665 M) , Cu(s) To find: the potential of the cell (õ)
From the cell diagram,Anode (oxidation): Zn(s) ÷ Zn2+(aq) + 2 e! õo
ox = +0.762 VCathode (reduction): Cu2+(aq) + 2 e! ÷ Cu(s) õo
red = +0.337 VRedox Reaction: Zn(s) + Cu2+(aq) ÷ Zn2+(aq) + Cu(s) õo
cell = ?
õocell = õo
ox + õored = (+0.762 V) + (+0.337 V) = +1.099 V
There are 2 e! being transferred in the balanced redox reaction. (ˆ n = 2)
ˆ õ = (+1.099 V) ! (0.02024 V) = +1.0788 V = +1.079 V

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 26
PART BQUESTION 4
Given: Ni(s) , NiCl2(aq, 0.0200 M) 1 CaCl2(aq) , Cl2(g, 1.97 atm) , Pt(s)[Ni2+] in the anode compartment = 0.0200 Mp(Cl2,g) in the cathode compartment = 1.97 atmõ = 1.80 V (at 298 K)
To find: [Cl!(aq)] in the cathode (reduction) compartment
According to the cell diagram,Anode (oxidation): Ni(s) ÷ Ni2+(aq) + 2 e! õo
ox = +0.257 VCathode (reduction): Cl2(g) + 2 e! ÷ 2 Cl!(aq) õo
red = +1.358 V
Redox Reaction: Ni(s) + Cl2(g) ÷ Ni2+(aq) + 2 Cl!(aq) õocell = ?
õocell = õo
ox + õored = (+0.257 V) + (+1.358 V) = +1.615 V
2 e! are being transferred in the balanced redox reaction (ˆ n = 2)
ˆ [Cl!] = 7.352 × 10!3 M= 7.35 × 10!3 M

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 27
(1) The anode is Ni(s) and the cathode is Pt(s). (2) Ni(s) acts as reducing agent and oxidizes to Ni2+(aq) under oxidation.
Cl2(g) acts as oxidizing agent and reduces to Cl!(aq) under reduction. (3) Electrons flow from Ni(s) anode to the Pt(s) cathode.
There are two e! being transferred in the balanced redox reaction. (4) The anions (Cl!) migrate toward the Ni(s) anode via a salt bridge.
The cations (Ni2+) diffuse toward the Pt(s) cathode via a salt bridge. (5) Under standard conditions,
õocell is positive (+1.615 V), and the reaction is spontaneous.
Under non-standard state conditions,õ is also positive (+1.80 V), and the reaction occurs spontaneous as written.
(6) As the reaction proceeds, the Ni(s) anode loses mass and the concentrations of Ni2+(aq) andCl!(aq) increase in the anode and cathode compartments, respectively.

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 28
PART BQUESTION 5
Given: For the reaction:AgCl(s) + Fe2+(aq) ÷ Ag(s) + Cl!(aq) + Fe3+(aq)
(a) a �shorthand� cell diagram for the reaction at an inert Pt anode
Î The anode (where oxidation occurs) is on the extreme left of the line (i.e. Pt(s)). Ï The cathode (where reduction occurs) is on the extreme right (i.e. Ag(s)). Ð , is used to separate the solid electrode from the liquid solution in contact. Ñ 1 is used to denote a salt bridge or porous barrier between the two solutions.
The cell diagram is: Pt(s) , Fe2+(aq), Fe3+(aq) 1 Cl!(aq) , AgCl(s) , Ag(s)
(b) õo for the reaction
Anode (oxidation): Fe2+(aq) ÷ Fe3+(aq) + e! õoox = !0.771 V
Cathode (reduction): AgCl(s) + e! ÷ Ag(s) + Cl!(aq) õored = +0.222 V
Redox Reaction: Fe2+(aq) + AgCl(s) ÷ Fe3+(aq) + Ag(s) + Cl!(aq)
õocell = õo
ox + õored = (!0.771 V) + (+0.222 V) = !0.549 V
The negative õocell indicates that this redox reaction is not spontaneous at standard conditions.
AgCl(s) will not dissociate into Ag(s) and Cl!(aq), and Fe2+(aq) will not oxidize to Fe3+(aq).
(c) )Go for the reaction
)Go = ! n ö õo (n = 1, only 1 e! transferred)= ! (1) (96.5 kJ V!1 mol!1) (!0.549 V)= + 52.98 kJ mol!1
= +53.0 kJ mol!1
(d) the equilibrium constant for the reaction
At 25oC (i.e. 298 K), )Go = ! R T Rn Keq
+52.98 kJ mol!1 = ! (8.314 × 10!3 kJ mol!1 K!1) (298 K) Rn Kc
ˆ Kc = 5.169 × 10!10 M = 5.17 × 10!10 M

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 29
PART BQUESTION 6
Given: Cd(s) , Cd2+(aq, 0.0250 M) 1 Pb2+(aq, 0.150 M) , Pb(s)õ = 0.293 V (at 298 K)
To find: )G and )Go for the cell reaction at 298 K(with NO access to SRP Table)
From the cell diagram,Anode (oxidation): Cd(s)(aq) ÷ Cd2+(aq) + 2 e! õo
ox = ?Cathode (reduction): Pb2+(s) + 2 e! ÷ Pb(s) õo
red = ?Redox Reaction: Cd(s) + Pb2+(aq) ÷ Cd2+(aq) + Pb(s) õo
cell = ?
Calculate )G using the formula: )G = ! n ö õ with õ = 0.293 V at 298 K. (n = 2) )G = ! n ö õ = ! (2) (96.5 kJ V!1 mol!1) (0.293 V)
= !56.549 kJ mol!1
= !56.5 kJ mol!1 (spontaneous at 298 K)
After )G is calculated, there are two approaches to solve for )Go.
METHOD 1: Calculate )Go at 298 K under non-standard state conditions.
)G = )Go + R T Rn Q
ˆ )Go = = !52.11 kJ mol!1 = !52.1 kJ mol!1 (spontaneous at 298 K)
METHOD 2: First calculate õo using Nernst equation, then solve for )Go using calculated õo value.
0.293 V = õo ! (!0.0230 V)ˆ õo = 0.293 V ! 0.0230 V = +0.270 V
ˆ )Go = ! n ö õo = ! (2) (96.5 kJ V!1 mol!1) (+0.270 V) = !52.1 kJ mol!1

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 30
PART BQUESTION 7
Given: Zn(s) , Zn2+(aq, ? M) 1 Zn2+(aq, 0.050 M) , Zn(s)õ = 0.015 V
To find: [Zn2+] in the anode compartment
This is a concentration cell, ˆ õocell = 0
According to the cell diagram,Zn(s, anode) + Zn2+(aq, 0.050 M) ÷ Zn2+(aq, ? M) + Zn(s, cathode)
ˆ [Zn2+]ox = 0.0155 M = 1.6 × 10!2 M
PART BQUESTION 8
Given: 2 H+(aq) + 2 e! ÷ H2(g)p(H2,g) = 2.5 atmpH = 6.00 ˆ [H+] = 1.0 × 10!6 M
To find: õ of the hydrogen electrode
For the reduction of hydrogen electrode,2 H+(aq) + 2 e! ÷ H2(g) õo
red = 0
2 e! are being transferred in this reduction half-reaction. (ˆ n = 2)
ˆ õ = 0 ! (+0.366 V) = !0.366 V = !0.37 V (nonspontaneous)

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 31
Oxidation state of oxygen changes from zero in O2(g) to !2 in H2O(R) by gaining 2 e!. There are 7 oxygen atoms of initially elemental oxygen (i.e. 7/2 O2(g)), thus it requires
7× 2 e! = 14 e! to be transferred in the reaction.
PART BQUESTION 9
Given: A possible reaction for a fuel cell is:C2H6(g) + 7/2 O2(g) ÷ 2 CO2(g) + 3 H2O(R)
To find: )Go and õocell
)Go must be calculated from a tabulated Thermodynamic data using )Gof values.
)Go (reaction) = 3 n )Gof (products) ! 3 n )Go
f (reactants)= {2× )Go
f [CO2(g)] + 3× )Gof [H2O(R)]}
! {)Gof [C2H6(g)] + 7/2× )Go
f [O2(g)]}= {2× (!394.359) + 3× (!237.178)} ! {(!32.92) + 7/2× (0)} kJ mol!1
= !1467.332 kJ mol!1 (spontaneous t 25oC)
(n = 14) )Go = ! n ö õo
!1467.332 kJ mol!1 = ! (14) (96.5 kJ V!1 mol!1) õo
ˆ õo = +1.086 V = +1.09 V (spontaneous at 25oC)
PART BQUESTION
10
To find: reagents that could reduce Cu2+(aq) to Cu(s),but not Al3+(aq) to Al(s)(from SRP Table)
Any reducing agent (via oxidation by losing e!) with SRP between +0.337 V and !1.662 V(i.e. between Cu(s) and Al(s)) can reduce Cu2+(aq) ÷ Cu(s), but cannot reduce Al3+(aq) ÷ Al(s).For examples, Pb(s), Ni(s), Cd(s), Fe(s), Cr(s), Zn(s), Mn(s) etc. can be the reducing agents forsuch a reaction.
Ni(s) + Cu2+(aq) ÷ Ni2+(aq) + Cu(s) õocell = +0.594 V (spontaneous)
Zn(s) + Cu2+(aq) ÷ Zn2+(aq) + Cu(s) õocell = +1.099 V (spontaneous)
Ni(s) + Al3+(aq) ÷ Ni2+(aq) + Al(s) õocell = !1.405 V (nonspontaneous)
Zn(s) + Al3+(aq) ÷ Zn2+(aq) + Al(s) õocell = !0.900 V (nonspontaneous)

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 32
PART BQUESTION
11
Given: Ag(s) electrode (anode or cathode ?)Cu(s) electrode (anode or cathode ?)[AgNO3] = 0.50 M = [Ag+]õ = 0.62 VAs the cell works, [Cu2+] is increasing.
To find: initial [Cu2+]
Based on the statement �As the cell works, concentration of Cu2+(aq) is increasing.�,Cu metal must be dissolving as it is oxidized (i.e. Cu2+ ions go into the solution), and so Cu(s)
must be the anode.. Therefore, the cathode must be Ag(s).
Anode (oxidation): Cu(s) ÷ Cu2+(aq) + 2 e! õoox = !0.337 V
Cathode (reduction): Ag+(aq) + e! ÷ Ag(s) õored = +0.799 V
2× Cathode (reduction): 2 Ag+(aq) + 2 e! ÷ 2 Ag(s) õored = +0.799 V
The Overall Redox Reaction is: Cu(s) + 2 Ag+(aq) ÷ Cu2+(aq) + 2 Ag(s)
õocell = õo
ox + õored = (!0.337 V) + (+0.799 V) = +0.462 V
There are two electrons being transferred in the balanced redox reaction. (ˆ n = 2)
ˆ [Cu2+] = 1.125 × 10!6 M = 1.1 × 10!6 M
For the reduction of Ag+(aq) to Ag(s),Ag+(aq) + e! ÷ Ag(s) õo
red = +0.799 V2 Ag+(aq) + 2 e! ÷ 2 Ag(s) õo
red = +0.799 VThe SRP are the same in both cases. The charge on the silver electrode is related to the number of
electrons per surface area of silver. Thus, the ration of e! to Ag atom in both cases is the same.
Therefore, electrode potentials are intensive properties and do not depend on the size of the electrode or themanner in which the half-reaction is balanced.

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 33
PART BQUESTION
12
Given: AgBr(s) + e! ÷ Ag(s) + Br!(aq) õored = +0.071 V
To find: Ksp of AgBr(s)
The solubility equilibrium of AgBr(s) is: AgBr(s) º Ag+(aq) + Br!(aq) The Ksp expression for AgBr(s) is: Ksp [AgBr(s)] = [Ag+(aq)] [Br!(aq)]
Î From the SRP table,Ag+(aq) + e! ÷ Ag(s) õo
red = +0.799 V
Ï Given: AgBr(s) + e! ÷ Ag(s) + Br!(aq) õored = +0.071 V
In order to obtain the Ksp [AgBr(s)] expression, both Ag+(aq) and Br!(aq) must be on the�product� side. Therefore, rearrange the above two equations as follows:
!1× Î Ag(s) ÷ Ag+(aq) + e! õoox = !0.799 V (i.e. oxidation)
Ï AgBr(s) + e! ÷ Ag(s) + Br!(aq) õored = +0.071 V (i.e. reduction)
Overall Reaction: AgBr(s) ÷ Ag+(aq) + Br!(aq) õocell = ?
õocell = õo
ox + õored = (!0.799 V) + (+0.071 V) = !0.728 V
Since õocell is negative, the reaction does not take place spontaneously as written. It
implies Ksp[AgBr(s)] is less than 1.
At equilibrium, õ = 0
( where n = 1, only 1 e! transferred in the redox reaction)
( where Keq = Ksp [AgBr(s)] = [Ag+(aq)] [Br!(aq)] )
ˆ Ksp [AgBr(s)] = 4.807 × 10!13 M2 = 4.81 × 10!13 M2

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 34
PART BQUESTION
13
Given: aqueous copper sulfate, CuSO4(aq)current (i) = 2.50 Atime (t) = 50.0 minutes = (50.0 × 60 s) = 3000 sboth Cu(s) and O2(g) are produced in the electrolysis
Anode (oxidation): 2 H2O(R) ÷ O2(g) + 4 H+(aq) + 4 e! õoox = !1.229 V
Cathode (reduction): Cu2+(aq) + 2 e! ÷ Cu(s) õored = +0.337 V
ˆ O2(g) is produced in the anode and Cu(s) is formed in the cathode.
charge (Q) = current (i) × time (t)
ˆ Q = (2.50 A) × (3000 s) = 7.50 × 103 C
n(e!) = Q ÷ ö = (7.50 × 103 C) ÷ (96500 C / mol e!) = 7.772 × 10!2 mol e!
(a) mass of Cu metal producedIt requires 2 moles of e! to produce 1 mole of Cu(s)
n(Cu) produced = n(e!) × (1 mole Cu(s) ÷ 2 moles e!) = (7.772 × 10!2 mol) × (½) = 3.886 × 10!2 mol
M(Cu) = 63.546 g mol!1
ˆ mass(Cu) deposited = n(Cu) × M(Cu)= (3.886 × 10!2 mol) × (63.546 g mol!1)= 2.469 g= 2.47 g
(b) volume of O2(g) produced at STPIt requires 4 moles of e! to produce 1 mole of O2(g)
n(O2) = n(e!) × (1 mole O2(g) ÷ 4 moles e!)= (7.772 × 10!2 mol) × (¼)= 1.943 × 10!2 mol
At STP, molar volume of O2(g) = 22.4 L mol!1
ˆ V(O2) produced = n(O2) × 22.4 L mol!1
= (1.943 × 10!2 mol) × 22.4 L mol!1
= 0.4352 L = 0.435 L

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 35
PART BQUESTION
14
CATHODE is an electrode where reduction takes place (i.e. gaining e!). ANODE is an electrode where oxidation takes place (i.e. losing e!).
(a) NaBr Given: aqueous solution of NaBr(aq)(i.e. Na+(aq), Br!(aq) and H2O(R))
To write: cathode and anode reactions
CATHODE
In aqueous solution, the cation (Na+) is not the only species that can be reduced. H2O(R) canbe reduced as well. The two possible reduction reactions are:
Î Na+(aq) + e! ÷ Na(s) õored = !2.71 V
Ï 2 H2O(R) + 2 e! ÷ H2(g) + 2 OH!(aq) õored = !0.828 V
‡ H2O(R) has a higher reduction potential (i.e. more +ve) than Na+(aq).ˆ H2O(R) is more readily reduced than Na+(aq)
andH2(g) is liberated at the cathode.
ANODE
In aqueous solution, the anion (BrG) is not the only species that can be oxidized. H2O(R) can beoxidized as well. The two possible oxidation reactions are:
Î 2 Br!(aq) ÷ Br2(R) + 2 e! õoox = !1.066 V
Ï 2 H2O(R) ÷ O2(g) + 4 H+(aq) + 4 e! õoox = !1.229 V
‡ Br!(aq) has a higher oxidation potential (i.e. more +ve) than H2O(R).ˆ Br!(aq) is more readily oxidized than H2O(R)
andBr2(R) is produced at the anode.
CATHODE: 2 H2O(R) + 2 e! ÷ H2(g) + 2 OH!(aq) (reduction)ANODE: 2 Br!(aq) ÷ Br2(R) + 2 e! (oxidation)

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 36
PART BQUESTION
14
(b) AgF Given: aqueous solution of AgF(aq)(i.e. Ag+(aq), F!(aq) and H2O(R))
To write: cathode and anode reactions
CATHODE
In aqueous solution, the cation (Ag+) is not the only species that can be reduced. H2O(R) canbe reduced as well. The two possible reduction reactions are:
Î Ag+(aq) + e! ÷ Ag(s) õored = +0.799 V
Ï 2 H2O(R) + 2 e! ÷ H2(g) + 2 OH!(aq) õored = !0.828 V
‡ Ag+(aq) has a higher reduction potential (i.e. more +ve) than H2O(R).ˆ Ag+(aq) is more readily reduced than H2O(R)
andAg(s) is formed at the cathode.
ANODE
In aqueous solution, the anion (FG) is not the only species that can be oxidized. H2O(R) can beoxidized as well. The two possible oxidation reactions are:
Î 2 F!(aq) ÷ F2(g) + 2 e! õoox = !2.866 V
Ï 2 H2O(R) ÷ O2(g) + 4 H+(aq) + 4 e! õoox = !1.229 V
‡ H2O(R) has a higher oxidation potential (i.e. more +ve) than F!(aq).ˆ H2O(R) is more readily oxidized than F!(aq)
andO2(g) is released at the anode.
CATHODE: Ag+(aq) + e! ÷ Ag(s) (reduction)ANODE: 2 H2O(R) ÷ O2(g) + 4 H+(aq) + 4 e! (oxidation)

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 37

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 38
PART BQUESTION
14
(c) NaF Given: aqueous solution of NaF(aq)(i.e. Na+(aq), F!(aq) and H2O(R))
To write: cathode and anode reactions
CATHODE
In aqueous solution, the cation (Na+) is not the only species that can be reduced. H2O(R) canbe reduced as well. The two possible reduction reactions are:
Î Na+(aq) + e! ÷ Na(s) õored = !2.71 V
Ï 2 H2O(R) + 2 e! ÷ H2(g) + 2 OH!(aq) õored = !0.828 V
‡ H2O(R) has a higher reduction potential (i.e. more +ve) than Na+(aq)ˆ H2O(R) is more readily reduced than Na+(aq)
andH2(g) is liberated at the cathode.
ANODE
In aqueous solution, the anion (FG) is not the only species that can be oxidized. H2O(R) can beoxidized as well. The two possible oxidation reactions are:
Î 2 F!(aq) ÷ F2(g) + 2 e! õoox = !2.866 V
Ï 2 H2O(R) ÷ O2(g) + 4 H+(aq) + 4 e! õoox = !1.229 V
‡ H2O(R) has a higher oxidation potential (i.e. more +ve) than F!(aq).ˆ H2O(R) is more readily oxidized than F!(aq)
andO2(g) is produced at the anode.
CATHODE: 2 H2O(R) + 2 e! ÷ H2(g) + 2 OH!(aq) (reduction)ANODE: 2 H2O(R) ÷ O2(g) + 4 H+(aq) + 4 e! (oxidation)

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 39
PART BQUESTION
15
Given: aqueous solution of NaOH(i.e. Na+(aq), OH!(aq) and H2O(R))
To find: products in each electrode To write: the overall reaction
CATHODE is an electrode where reduction takes place (i.e. gaining e!).ANODE is an electrode where oxidation takes place (i.e. losing e!).
CATHODE
In aqueous solution, the cation (Na+) is not the only species that can be reduced. H2O(R) canbe reduced as well. The two possible reduction reactions are:
Î Na+(aq) + e! ÷ Na(s) õored = !2.71 V
Ï 2 H2O(R) + 2 e! ÷ H2(g) + 2 OH!(aq) õored = !0.828 V
‡ H2O(R) has a higher reduction potential (i.e. more +ve) than Na+(aq).ˆ H2O(R) is more readily reduced than Na+(aq)
andH2(g) is liberated at the cathode.
ANODE
In aqueous solution, the anion (OHG) is not the only species that can be oxidized. H2O(R) canbe oxidized as well. The two possible oxidation reactions are:
Î 4 OH!(aq) ÷ O2(g) + 2 H2O(R) + 4 e! õoox = !0.401 V
Ï 2 H2O(R) ÷ O2(g) + 4 H+(aq) + 4 e! õoox = !1.229 V
‡ OH!(aq) has a higher oxidation potential (i.e. more +ve) than H2O(R).ˆ OH!(aq) is more readily oxidized than H2O(R)
andO2(g) is formed at the anode.
2× CATHODE: 4 H2O(R) + 4 e! ÷ 2 H2(g) + 4 OH!(aq) (reduction)ANODE: 4 OH!(aq) ÷ O2(g) + 2 H2O(R) + 4 e! (oxidation)
OVERALL REDOX: 2 H2O(R) ÷ 2 H2(g) + O2(g)

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 40
(1) Reducing agent is a species that can lose e! (i.e. oxidation) andtherefore become more positive (i.e. higher oxidation state).
(2) Reducing agent (X) undergoes oxidation and oxidizes to Xn+.X ÷ Xn+ + n e!
PART BMC #1
To find: the oxidation numbers of chlorine in Mg(ClO)2 and ClO3!
Mg(ClO)2 (1) Mg has an oxidation state +2 as Mg2+.
O has an oxidation state !2 as O2!.
(2) Mg(ClO)2 is a neutral compound and has a net charge of zero.(i.e. the sum of the oxidation numbers of all atoms in the compound is zero
and anion ClO! has a net charge of !1.)
+2 (Mg) + [ 2× (Cl) + 2× (!2) O] ] = 0ˆ Cl = +1
ClO3! (1) O has an oxidation state !2 as O2!.
(2) Anion ClO3! has a net charge of !1.
(Cl) + 3× (!2) O = !1ˆ Cl = +5
PART BMC #2
Given: Î A+ + B ÷ A + B+
Ï A+ + C ÷ no reactionÐ 2 B+ + D ÷ 2 B + D2+
To find: the decreasing order of reactivity as reducing agent
From Î: A+ + B ÷ A + B+ B is more easily oxidized than A.(i.e. B can reduce A+ to A.) (i.e. B > A)
From Ï A+ + C ÷ no reaction C cannot reduce A+ to A. (i.e. A > C)From Ð 2 B+ + D ÷ 2 B + D2+ D is more easily oxidized than B. (i.e. D > B)
Thus, the decreasing order of reactivity as reducing agent is:(strongest reducing agent) D > B > A > C (weakest reducing agent)

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 41
(1) Oxidizing agent is a species that can gain e! (i.e. reduction) andtherefore become more negative (i.e. lower oxidation state).
(2) Oxidizing agent (Xn+) undergoes reduction and reduces to X.Xn+ + n e! ÷ X
PART BMC #3
Given: reactants: potassium dichromate (K2Cr2O7)sodium chloride (NaCl)
products: Cr3+(aq) and Cl2(g)
To find: the oxidizing agent
Î The unbalanced redox reaction is:Cr2O7
2!(aq) + Cl!(aq) ÷ Cr3+(aq) + Cl2(g)
(Note that K+(aq) and Na+(aq) are spectator ions in aqueous solution.)
Ï The oxidation state of chlorine increases from !1 in Cl!(aq) to zero in Cl2(g). Therefore, Cl islosing one e! in this half-reaction (i.e. oxidation), and so it is oxidized.
Cl!(aq) ÷ Cl2(g)2 Cl!(aq) ÷ Cl2(g)2 Cl!(aq) ÷ Cl2(g) + 2 e!
Ð The oxidation state of chromium decreases from +6 in Cr2O72!(aq) to +3 in Cr3+(aq). Therefore,
Cr is gaining 3 e! in this half-reaction (i.e. reduction), and so it is reduced.Cr2O7
2!(aq) ÷ Cr3+(aq)Cr2O7
2!(aq) ÷ 2 Cr3+(aq)Cr2O7
2!(aq) ÷ 2 Cr3+(aq) + 7 H2O(R)Cr2O7
2!(aq) + 14 H+(aq) ÷ 2 Cr3+(aq) + 7 H2O(R)Cr2O7
2!(aq) + 14 H+(aq) + 6 e! ÷ 2 Cr3+(aq) + 7 H2O(R)
ˆ Cr2O72!(aq) is the oxidizing agent which undergoes reduction.
(Cl!(aq) is the reducing agent which undergoes oxidation.)
For the overall balanced redox reaction: 3× Ï 6 Cl!(aq) ÷ 3 Cl2(g) + 6 e!
Ð Cr2O72!(aq) + 14 H+(aq) + 6 e! ÷ 2 Cr3+(aq) + 7 H2O(R)
REDOX: Cr2O72!(aq) + 6 Cl!(aq) + 14 H+(aq) ÷ 2 Cr3+(aq) + 3 Cl2(g) + 7 H2O(R)
(Note that there are 6 e! being transferred in the redox reaction.)

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 42
Oxidation = loss of e!
Reduction = gain of e!
H2S(aq): acts as reducing agent which undergoes oxidation. MnO4
!(aq): acts as oxidizing agent which undergoes reduction.
PART BMC #4
Given: For the balanced redox reaction:5 H2S(aq) + 2 MnO4
!(aq) + 6 H+(aq) ÷ 2 Mn2+(aq) + 5 S(s) + 8 H2O(R)
To find: the number of electrons transferred
Î OxidationThe oxidation state of sulfur increases from !2 in H2S(aq) to zero in S(s). Therefore, S
atom is losing 2 e! in this half-reaction (i.e. oxidation), and so it is oxidized.H2S(aq) ÷ S(s)H2S(aq) ÷ S(s) + 2 H+(aq)H2S(aq) ÷ S(s) + 2 H+(aq) + 2 e!
Ï ReductionThe oxidation state of manganese decreases from +7 in MnO4
!(aq) to +2 in Mn2+(aq).Therefore, Mn atom is gaining 5 e! in this half-reaction (i.e. reduction), and so it is reduced.
MnO4!(aq) ÷ Mn2+(aq)
MnO4!(aq) ÷ Mn2+(aq) + 4 H2O(R)
MnO4!(aq) + 8 H+(aq) ÷ Mn2+(aq) + 4 H2O(R)
MnO4!(aq) + 8 H+(aq) + 5 e! ÷ Mn2+(aq) + 4 H2O(R)
For the overall balanced redox reaction: 5× Î 5 H2S(aq) ÷ 5 S(s) + 10 H+(aq) + 10 e!
2× Ï 2 MnO4!(aq) + 16 H+(aq) + 10 e! ÷ 2 Mn2+(aq) + 8 H2O(R)
REDOX: 5 H2S(aq) + 2 MnO4!(aq) + 6 H+(aq) ÷ 2 Mn2+(aq) + 5 S(s) + 8 H2O(R)
ˆ There are 10 e! being transferred in the redox reaction.
ALTERNATE EXPLANATION:Each sulfur atom loses 2 e!, thus it requires 10 e! transferred for 5 sulfur atoms in the redox
reaction. On the other hand, each manganese atom gains 5 e!, thus it also requires 10 e! transferredfor 2 Mn atoms in the redox reaction.

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 43
PART BMC #5
Given: C6H12O6(s) + 6 O2(g) ÷ 6 CO2(g) + 6 H2O(R)
To find: the number of electrons transferred
Î Oxidation state of oxygen decreases from zero in O2(g) to !2 in H2O(R) by gaining 2 e!.There are 12 oxygen atoms of initially elemental oxygen (i.e. 6 O2(g)), thus it requires:
12× 2 e! = 24 e!
Ï Oxidation state of carbon increases from zero in C6H12O6(s) to +4 in CO2(g) by losing 4 e!.There are 6 carbon atoms in C6H12O6(s), thus it requires:
6× 4 e! = 24 e!
ˆ There are 24 e! being transferred in the reaction.
ALTERNATE METHOD by balancing the redox reaction
Î OxidationC6H12O6(s) ÷ CO2(g)C6H12O6(s) ÷ 6 CO2(g)C6H12O6(s) + 6 H2O(R) ÷ 6 CO2(g)C6H12O6(s) + 6 H2O(R) ÷ 6 CO2(g) + 24 H+(aq)C6H12O6(s) + 6 H2O(R) ÷ 6 CO2(g) + 24 H+(aq) + 24 e!
Ï ReductionO2(g) ÷ H2O(R)O2(g) ÷ 2 H2O(R)O2(g) + 4 H+(aq) ÷ 2 H2O(R)O2(g) + 4 H+(aq) + 4 e! ÷ 2 H2O(R)
For the overall balanced redox reaction:Î C6H12O6(s) + 6 H2O(R) ÷ 6 CO2(g) + 24 H+(aq) + 24 e!
6× Ï 6 O2(g) + 24 H+(aq) + 24 e! ÷ 12 H2O(R)REDOX: C6H12O6(s) + 6 O2(g) ÷ 6 CO2(g) + 6 H2O(R)
ˆ There are 24 e! being transferred in the redox reaction.

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 44
PART BMC #6
Given: Sc(s) , Sc+(aq) 1 Cl!(aq) , AgCl(s) , Ag(s)õo
cell = 0.56 VAgCl(s) + e! ÷ Ag(s) + Cl!(aq) õo
red = +0.22 V
To find: the standard reduction potential (SRP) of Sc+(aq)Sc+(aq) + e! ÷ Sc(s) õo
red = ?
From the cell diagram, the redox reaction is:Sc(s) + AgCl(s) ÷ Sc+(aq) + Ag+(aq) + Cl!(aq) õo
cell = 0.56 Vˆ Anode (oxidation): Sc(s) ÷ Sc+(aq) + e! õo
ox = ?ˆ Cathode (reduction): AgCl(s) + e! ÷ Ag(s) + Cl!(aq) õo
red = +0.22 V
õocell = õo
ox + õored
0.56 V = õoox + (+0.22 V)
õoox = +0.34 V
ˆ The SRP of Sc+ is: Sc+(aq) + e! ÷ Sc(s) õored = !0.34 V
PART BMC #7
Given: 2 Ag+(aq) + H2(g) ÷ 2 Ag(s) + 2 H+(aq)[Ag+(aq)] = 0.10 Mp(H2,g) = 1 atmpH = 8.00 ˆ [H+] = 1.0 × 10!8 M
To find: õ at 25oC
Redox Reaction: 2 Ag+(aq) + H2(g) ÷ 2 Ag(s) + 2 H+(aq) õocell = ?
ˆ Anode (oxidation): H2(g) ÷ 2 H+(aq) + 2 e! õoox = 0
ˆ Cathode (reduction): 2 Ag+(aq) + 2 e! ÷ 2 Ag(s) õored = +0.799 V
õocell = õo
ox + õored = (0) + (+0.799 V) = +0.799 V
ˆ õ = 0.799 V ! (!0.4137 V) = +1.2127 V = +1.21 V

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 45
PART BMC #8
Given: 1.0 M aqueous solution of KI(aq)(i.e. K+(aq), I!(aq) and H2O(R))
To find: product(s) formed at the anode
ANODE is the electrode where oxidation takes place (i.e. losing e!).
ANODE
In aqueous solution, the anion (IG) is not the only species that can be oxidized. H2O(R) can beoxidized as well. The two possible oxidation reactions are:
Î 2 I!(aq) ÷ I2(s) + 2 e! õoox = !0.536 V
Ï 2 H2O(R) ÷ O2(g) + 4 H+(aq) + 4 e! õoox = !1.229 V
‡ I!(aq) has a higher oxidation potential (i.e. more +ve) than H2O(R).ˆ I!(aq) is more readily oxidized than H2O(R)
andI2(s) is produced at the anode.
CATHODE
In aqueous solution, the cation (K+) is not the only species that can be reduced. H2O(R) can bereduced as well. The two possible reduction reactions are:
Î K+(aq) + e! ÷ K(s) õored = !2.93 V
Ï 2 H2O(R) + 2 e! ÷ H2(g) + 2 OH!(aq) õored = !0.828 V
‡ H2O(R) has a higher reduction potential (i.e. more +ve) than K+(aq).ˆ H2O(R) is more readily reduced than K+(aq)
andH2(g) is liberated at the cathode.
ANODE: 2 I!(aq) ÷ I2(s) + 2 e! (oxidation)CATHODE: 2 H2O(R) + 2 e! ÷ H2(g) + 2 OH!(aq) (reduction)

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 46
PART BMC #9
Given: 500 mL of 0.100 M NiSO4(aq)current (i) = 250 mA = 0.250 A
To find: time required to electrodeposit 1.0 g of Ni(s)
The cathode reaction (i.e. reduction) will produce Ni(s).Ni2+(aq) + 2 e! ÷ Ni(s)
M(Ni) = 58.70 g mol!1
n(Ni) produced = mass(Ni) ÷ M(Ni) = 1.0 g ÷ 58.70 g mol!1
= 1.703 × 10!2 mol
For 500 mL of 0.100 M NiSO4(aq),n(Ni2+) = c(Ni2+) × V(Ni2+)
= (0.100 mol L!1) × (500 × 10!3 L)= 5.00 × 10!2 mol
1 mole Ni2+(aq) produces 1 mole Ni(s)ˆ maximum n(Ni) produced = n(Ni2+) supplied
= 5.00 × 10!2 mol
Thus, 1.0 g Ni(s) can be produced from 500 mL of 0.100 M NiSO4 solution. (i.e. NiSO4(aq) is in excess, maximum of 2.935 g Ni(s) can be produced)
It requires 2 moles of e! to plate out 1 mole of Ni(s)n(e!) = n(Ni) × (2 moles e! ÷ 1 mole Ni(s))
= (1.703 × 10!2 mol) × (2)= 3.406 × 10!2 mol
electrical charge of the system (Q) = n(e!) × ö= (3.406 × 10!2 mol e!) × (96500 C / mol e!)= 3.287 × 103 C
charge (Q) = current (i) × time (t)
3.287 × 103 C = (0.250 A) × time (t)
ˆ t = 1.315 × 104 sec = 219.2 min = 220 min

CHEM*131 (W 02) REVIEW QUESTIONS FOR FINAL EXAM PAGE - 47
PART BMC#10
Given: 250 mL of a 0.500 M Fe(NO3)3 solutioncurrent (i) = 25.0 Atime (t) = 30 min = (30 × 60 s) = 1800 s
To find: mass of Fe(s) deposited at the cathode
Cathode allows reduction to take place. Thus the half-reaction for the deposit of Fe(s) is:Fe3+(aq) + 3 e! ÷ Fe(s)
First determine which item is the limiting factor (Î Fe(NO3)3 solution or Ï time).
Î n(Fe(NO3)3) supplied = c(Fe(NO3)3) × V(Fe(NO3)3)= (0.500 mol L!1) × (250 × 10!3 L)= 1.25 × 10!1 mol= n(Fe3+)
1 mole of Fe3+(aq) deposits 1 mole of Fe(s)ˆ n(Fe) = n(Fe3+) = 1.25 × 10!1 mol
Ïcharge (Q) = current (i) × time (t)
ˆ Q = (25.0 A) × (1800 s) = 4.50 × 104 C
n(e!) = Q ÷ ö= (4.50 × 104 C) ÷ (96500 C / mol e!)= 4.663 × 10!1 mol e!
It requires 3 moles of e! to plate out 1 mole of Fe(s)n(Fe) deposited = n(e!) × (1 mole Fe(s) ÷ 3 moles e!)
= (4.663 × 10!1 mol) × (a)= 1.554 × 10!1 mol
ˆ Fe(NO3)3 solution is the limiting reagent, not the time and current.(It takes only -24 min with a constant current of 25.0 A to completely deposit Fe(s) from 250 mLof a 0.500 M Fe(NO3)3 solution. Verify the calculated time yourself!)
M(Fe) = 55.847 g mol!1
ˆ mass(Fe) deposited = n(Fe) × M(Fe)= (1.25 × 10!1 mol) × (55.847 g mol!1)= 6.9809 g= 6.98 g
Related Documents




![Beyond Chemical - Exotic Energetics/ Propulsion · PDF fileBeyond Chemical - Exotic Energetics/ Propulsion ... time scales] and is exceedingly ... a collective proton resonance in](https://static.cupdf.com/doc/110x72/5aa89cbb7f8b9a6c188bc37b/beyond-chemical-exotic-energetics-propulsion-chemical-exotic-energetics-propulsion.jpg)