NEWS IN BRIEF 1298 | CANCER DISCOVERYDECEMBER 2016 www.aacrjournals.org • Gilead Sciences’ entospletinib, an inhibitor of the tyrosine kinase Syk, which blocks B-cell receptor signaling Three to six more drugs may be added over the course of the trial, which will run for 3 to 5 years. “This is about working toward a common goal,” said Levine. “We really want to make a difference for our patients.” –Karen Weintraub ■ Nobel Prize Honors Autophagy Discovery Japanese cell biologist Yoshinori Ohsumi, PhD, has been awarded this year’s Nobel Prize in Physiology or Medicine for his discovery of autophagy. His groundbreaking studies in yeast illuminated how cells collect and break down intracellular proteins and organelles, a process that is critical to the survival of both normal cells and some cancerous ones. “His discoveries opened the path to understanding the fundamental importance of autophagy in many physiological processes, such as in the adaptation to starvation or response to infection,” noted the Nobel Committee in announcing the award in October. The concept of autophagy was first observed in the 1960s, but little was known about its underlying mecha- nisms until Ohsumi conducted a series of experiments with baker’s yeast in the early 1990s. Those studies eventually led him to identify the genes—and the proteins they encode—that control autophagy, and to show that a corre- sponding mechanism exists in humans. Autophagy is a self-defense mecha- nism that prevents the accumulation of garbage or potentially toxic material in cells, such as damaged proteins and organelles. In normal cells, it serves as a buffer during metabolic stress by recycling intracellular components. It also helps to eliminate invading bac- teria and viruses following infection, promote embryo development and cell differentiation, and counteract the negative consequences of aging. Ohsumi verified that the process exists in yeast cells by studying how autophagy delivers cargo for degra- dation in the vacuole. (In human cells, similarly, autophagosomes fuse with lysosomes, which contain enzymes that degrade proteins and organelles.) He then cultivated cells that lacked vacuolar degradation enzymes, starved them, and was able to observe the vacuoles fill with small vesicles holding proteins to be degraded—known as autophagosomes. In another set of experiments, Ohsumi exposed engineered yeast cells to a chem- ical that randomly introduced mutations in many genes, then induced autophagy. As a result, he identified many different proteins and protein complexes that regulate distinct stages of autophago- some initiation and formation. “Ohsumi used yeast as a model system to identify the mechanisms it uses to survive nitrogen starvation and the genes essential for the autophagy pathway,” says Eileen White, PhD, professor of molecular biology and biochemistry at Rutgers University and deputy director and associate director for Basic Science at Rutgers Cancer Institute of New Jersey, in New Brunswick. “By doing this, he opened up a whole new field of investigation.” Recent research has revealed that some cancer cells in hypoxic regions also use autophagy to survive metabolic stress, suggesting that the process may be a via- ble drug target, says White. Companies are now working on autophagy inhibi- tors, possibly to augment the activity of targeted drugs. Multiple clinical trials are testing hydroxychloroquine (HCQ), an antimalarial drug that interferes with lysosome function, in combination cancer therapy. For example, a phase II trial is assessing HCQ combined with the BRAF inhibitor dabrafenib (Tafinlar; Novartis) and trametinib, a MEK inhibi- tor (Mekinist; Novartis), in patients with advanced BRAF-mutant melanoma. “The discovery of autophagy genes and how they enable lysosomal deg- radation and recycling of organelles and proteins has provided a critical platform for understanding how autophagy plays a role in resistance to cancer therapy,” says Ravi Amaravadi, MD, a medical oncologist at the Uni- versity of Pennsylvania in Philadelphia, who led a series of early-phase clinical trials testing HCQ in combination with targeted therapies. “Ohsumi’s work provided the framework to pro- pose biomarkers and targets for drug discovery that could improve the efficacy of cancer therapies in a number of cancers.” –Janet Colwell ■ announcing the trial. With LLS acting as a neutral party, more pharmaceuti- cal companies may join the effort to bring drugs to market more quickly. “The secret sauce of this trial is the idea that we’re going to work faster by collaborating in open and transparent ways,” said Ross Levine, MD, of Mem- orial Sloan Kettering Cancer Center (MSKCC) in New York, NY. In addition to MSKCC, the Ohio State University Comprehensive Cancer Center (OSUCCC) in Columbus, Oregon Health & Science University Knight Cancer Institute in Portland, and Dana-Farber Cancer Institute and Massachusetts General Hospital, both in Boston, will serve as the initial trial sites. Researchers plan to enroll 500 patients over age 60 newly diagnosed with AML in the trial. Participants will have their cancer’s genome sequenced and be put into a trial arm based on nine different genomic signatures, said John Byrd, MD, of OSUCCC. Unlike other trials, none of the participants will receive standard chemotherapy alone. If there is not a suitable drug match for a patient’s genetic markers, they will be offered chemotherapy in combination with an existing targeted therapy, Byrd explained. Patients who do not res- pond to their investigational drug can also receive conventional therapy with the targeted therapy, he added. Most of the patients who will be eligible for the trial are unable to tol- erate intensive chemotherapy, so their outcomes are even worse than average, said Brian Druker, MD, director of the Knight Cancer Institute. The targeted therapies are expected to be better tolerated than toxic chemotherapy, and will be given after diagnosis, when they will likely be more effective, instead of waiting for disease relapse, he added. DeGennaro said that the trial will begin by testing these four drugs: • Alexion’s samalizumab (ALXN6000), a humanized monoclonal antibody that targets CD200 • Boehringer Ingelheim’s BI 836858, which induces antibody-dependent cellular cytotoxicity against CD33- expressing tumor cells • Celgene and Agios’s enasidenib (AG- 221/ CC-90007), a selective IDH2 inhibitor on July 26, 2020. © 2016 American Association for Cancer Research. cancerdiscovery.aacrjournals.org Downloaded from Published OnlineFirst October 17, 2016; DOI: 10.1158/2159-8290.CD-NB2016-127

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

NEWS IN BRIEF
1298 | CANCER DISCOVERY�DECEMBER 2016 www.aacrjournals.org
• Gilead Sciences’ entospletinib, an
inhibitor of the tyrosine kinase Syk,
which blocks B-cell receptor signaling
Three to six more drugs may be
added over the course of the trial, which
will run for 3 to 5 years.
“This is about working toward a
common goal,” said Levine. “We really
want to make a difference for our
patients.” –Karen Weintraub ■
Nobel Prize Honors Autophagy Discovery
Japanese cell biologist Yoshinori
Ohsumi, PhD, has been awarded this
year’s Nobel Prize in Physiology or
Medicine for his discovery of autophagy.
His groundbreaking studies in yeast
illuminated how cells collect and
break down intracellular proteins and
organelles, a process that is critical to
the survival of both normal cells and
some cancerous ones.
“His discoveries opened the path
to understanding the fundamental
importance of autophagy in many
physiological processes, such as in the
adaptation to starvation or response to
infection,” noted the Nobel Committee
in announcing the award in October.
The concept of autophagy was fi rst
observed in the 1960s, but little was
known about its underlying mecha-
nisms until Ohsumi conducted a series
of experiments with baker’s yeast in the
early 1990s. Those studies eventually
led him to identify the genes—and the
proteins they encode—that control
autophagy, and to show that a corre-
sponding mechanism exists in humans.
Autophagy is a self-defense mecha-
nism that prevents the accumulation
of garbage or potentially toxic material
in cells, such as damaged proteins and
organelles. In normal cells, it serves
as a buffer during metabolic stress by
recycling intracellular components. It
also helps to eliminate invading bac-
teria and viruses following infection,
promote embryo development and
cell differentiation, and counteract the
negative consequences of aging.
Ohsumi verifi ed that the process
exists in yeast cells by studying how
autophagy delivers cargo for degra-
dation in the vacuole. (In human cells,
similarly, autophagosomes fuse with
lysosomes, which contain enzymes that
degrade proteins and organelles.) He
then cultivated cells that lacked vacuolar
degradation enzymes, starved them, and
was able to observe the vacuoles fi ll with
small vesicles holding proteins to be
degraded—known as autophagosomes.
In another set of experiments, Ohsumi
exposed engineered yeast cells to a chem-
ical that randomly introduced mutations
in many genes, then induced autophagy.
As a result, he identifi ed many different
proteins and protein complexes that
regulate distinct stages of autophago-
some initiation and formation.
“Ohsumi used yeast as a model
system to identify the mechanisms it
uses to survive nitrogen starvation and
the genes essential for the autophagy
pathway,” says Eileen White, PhD,
professor of molecular biology and
biochemistry at Rutgers University
and deputy director and associate
director for Basic Science at Rutgers
Cancer Institute of New Jersey, in New
Brunswick. “By doing this, he opened
up a whole new fi eld of investigation.”
Recent research has revealed that some
cancer cells in hypoxic regions also use
autophagy to survive metabolic stress,
suggesting that the process may be a via-
ble drug target, says White. Companies
are now working on autophagy inhibi-
tors, possibly to augment the activity of
targeted drugs. Multiple clinical trials
are testing hydroxychloroquine (HCQ),
an antimalarial drug that interferes
with lysosome function, in combination
cancer therapy. For example, a phase II
trial is assessing HCQ combined with
the BRAF inhibitor dabrafenib (Tafi nlar;
Novartis) and trametinib, a MEK inhibi-
tor (Mekinist; Novartis), in patients with
advanced BRAF-mutant melanoma.
“The discovery of autophagy genes
and how they enable lysosomal deg-
radation and recycling of organelles
and proteins has provided a critical
platform for understanding how
autophagy plays a role in resistance to
cancer therapy,” says Ravi Amaravadi,
MD, a medical oncologist at the Uni-
versity of Pennsylvania in Philadelphia,
who led a series of early-phase clinical
trials testing HCQ in combination
with targeted therapies. “Ohsumi’s
work provided the framework to pro-
pose biomarkers and targets for drug
discovery that could improve the effi cacy
of cancer therapies in a number of
cancers.” –Janet Colwell ■
announcing the trial. With LLS acting
as a neutral party, more pharmaceuti-
cal companies may join the effort to
bring drugs to market more quickly.
“The secret sauce of this trial is the
idea that we’re going to work faster by
collaborating in open and transparent
ways,” said Ross Levine, MD, of Mem-
orial Sloan Kettering Cancer Center
(MSKCC) in New York, NY. In addition
to MSKCC, the Ohio State University
Comprehensive Cancer Center (OSUCCC)
in Columbus, Oregon Health & Science
University Knight Cancer Institute
in Portland, and Dana-Farber Cancer
Institute and Massachusetts General
Hospital, both in Boston, will serve as
the initial trial sites.
Researchers plan to enroll 500
patients over age 60 newly diagnosed
with AML in the trial. Participants will
have their cancer’s genome sequenced
and be put into a trial arm based on
nine different genomic signatures, said
John Byrd, MD, of OSUCCC.
Unlike other trials, none of the
participants will receive standard
chemotherapy alone. If there is not
a suitable drug match for a patient’s
genetic markers, they will be offered
chemotherapy in combination with
an existing targeted therapy, Byrd
explained. Patients who do not res-
pond to their investigational drug can
also receive conventional therapy with
the targeted therapy, he added.
Most of the patients who will be
eligible for the trial are unable to tol-
erate intensive chemotherapy, so their
outcomes are even worse than average,
said Brian Druker, MD, director of the
Knight Cancer Institute. The targeted
therapies are expected to be better
tolerated than toxic chemotherapy, and
will be given after diagnosis, when they
will likely be more effective, instead of
waiting for disease relapse, he added.
DeGennaro said that the trial will
begin by testing these four drugs:
• Alexion’s samalizumab (ALXN6000),
a humanized monoclonal antibody
that targets CD200
• Boehringer Ingelheim’s BI 836858,
which induces antibody-dependent
cellular cytotoxicity against CD33-
expressing tumor cells
• Celgene and Agios’s enasidenib (AG-
221/ CC-90007), a selective IDH2
inhibitor
on July 26, 2020. © 2016 American Association for Cancer Research. cancerdiscovery.aacrjournals.org Downloaded from
Published OnlineFirst October 17, 2016; DOI: 10.1158/2159-8290.CD-NB2016-127

NEWS IN BRIEF
DECEMBER 2016�CANCER DISCOVERY | 1299
Anderson Cancer Center in Houston.
“We are now trying to identify markers
of response because we don’t yet know
why these drugs work better in some
patients than others.”
Researchers are also exploring
whether ribociclib may be effective
in second- or third-line settings or in
combination with inhibitors of other
signaling pathways involved in breast
cancer, adds Hortobagyi.
Ribociclib, which has been submit-
ted for FDA approval, is one of three
CDK4/6 inhibitors either approved
or being tested in combination with
antiestrogen therapy for advanced
HR-positive, HER2-negative breast
cancer. Palbociclib (Ibrance; Pfizer)
received FDA approval last year and
abemaciclib (Eli Lilly) is in early clini-
cal trials.
Identifi cation of biomarkers is criti-
cal, considering that CDK4/6 inhibi-
tors are known to cause significant
side effects, notes V.K. Gadi, MD, PhD,
of the University of Washington and a
clinical researcher at Fred Hutchinson
Cancer Research Center in Seattle.
In the MONALEESA-2 trial, neutro-
penia and leukopenia were far more
common in the intervention versus
the control group (59.3% vs. 0.9% and
21.0% vs. 0.6%, respectively). However,
severe (grade 4) neutropenia was
relatively uncommon (9.6% vs. 0%)
and could be managed with dose
interruptions or reductions.
Although ribociclib appears to have
a similar toxicity profi le to palbociclib,
abemaciclib may be less toxic than
either drug, notes Gadi. For example,
a recent phase I study found that
single-agent abemaciclib triggered
neutropenia in just 9% of 173 patients
with breast cancer, non–small cell lung
cancer, and other solid tumors (Cancer
Discov 2016;6:740–53).
“The high rate of neutropenia seen
with ribociclib and palbociclib makes
treating these patients very challeng-
ing,” he says. “What we really need is
a good marker of response so we can
decide who actually needs this therapy
and who could be spared the toxicity.”
–Janet Colwell ■
Rib ociclib Lengthens Breast Cancer Survival
The combination of antiestrogen
therapy and ribociclib (Novartis), an
investigational CDK4/6 inhibitor, pro-
longed progression-free survival (PFS)
in women with metastatic HR-positive
HER2-negative breast cancer, according
to fi ndings presented at the European
Society for Medical Oncology’s 2016
Congress in Copenhagen, Denmark.
Data from the large phase III trial,
which were published concurrently in
The New England Journal of Medicine, may
lead to FDA approval of the second
CDK4/6 inhibitor for this disease
(N Engl J Med 2016;375:1738–48).
A planned interim analysis of the
MONALEESA-2 trial, which included
almost 700 postmenopausal women,
found that fi rst-line therapy with riboci-
clib in addition to the aromatase inhibi-
tor letrozole signifi cantly improved
outcomes. After 18 months of treat-
ment, median PFS was not reached in
the ribociclib arm compared with 14.7
months in the control group—a 44%
improvement.
“The majority of patients treated with
ribociclib completed therapy and received
signifi cant benefi t,” notes the study’s
lead investigator Gabriel Hortobagyi,
MD, of the University of Texas MD
For more news on cancer research, visit Cancer Discovery online at http://cancerdiscovery.aacrjournals.org/content/early/by/section.
NOTED
Vice President Joe Biden delivered a report on the Cancer Moonshot, summa-rizing the work of its Task Force and lay-ing out his strategic plan for transforming cancer research and care. In addition, the report notes areas of “scientific opportu-nity” as determined by a Blue Ribbon Panel and announced in September. It also outlines several new collaborative efforts between the public and private sectors. For example, the NCI, Amazon Web Services, and Microsoft are building a sustainable model for maintaining can-cer genomic data in the cloud.
Researchers at Roswell Park Cancer Institute in Buffalo, NY, got the FDA’s go-ahead to evaluate CIMAvax-EGF, a lung cancer vaccine developed in Cuba, in a U.S. clinical trial. Roswell Park also received authorization from the U.S. Department of the Treasury to establish a joint business venture with the Center of Molecular Immunology in Havana, aimed at discover-ing and developing new biotech products.
The U.S. Multi-Society Task Force on Colorectal Cancer released a consensus statement on using the fecal immuno-chemical test (FIT) for colorectal cancer screening (Ann Intern Med 2016 Oct 25 [Epub ahead of print]). FIT is “an excellent alternative” to colonoscopy for preventing this disease, the authors said, adding that “surprisingly little published evidence sup-ports the predominance of colonoscopy.”
Genentech announced that the FDA approved atezolizumab (Tecentriq) for the treatment of metastatic non–small cell lung cancer (NSCLC) that has progressed during or after treatment with platinum-containing chemotherapy. Atezolizumab is the first PD-L1 inhibitor given the green light by the agency for NSCLC; in May, it received accelerated approval for the treatment of locally advanced or meta-static urothelial carcinoma.
Celgene acquired Swiss biotech EngMab for $600 million, expanding its pipeline of immunotherapies targeting BCMA, an antigen almost exclusively expressed on plasma cells and multiple myeloma cells. EngMab is developing a bispecific anti-body that binds to BCMA on multiple myeloma cells and to CD3 on T cells, trig-gering T cell–mediated cytotoxicity.
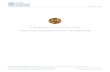
Autophagy represents a cell’s response to chemotherapy and radiation that may be one component of resistance to therapy. H460 non–small cell lung cancer cells were exposed to the antitumor drug etoposide. The image shows the H460 cells (in green) containing acidic autophagic vacuoles (orange) that are extranuclear. The small bright green bodies are likely to be micronuclei.
Dav
id A
. Gew
irtz
an
d T
are
q S
ale
h
on July 26, 2020. © 2016 American Association for Cancer Research. cancerdiscovery.aacrjournals.org Downloaded from
Published OnlineFirst October 17, 2016; DOI: 10.1158/2159-8290.CD-NB2016-127

2016;6:1298-1299. Published OnlineFirst October 17, 2016.Cancer Discov Nobel Prize Honors Autophagy Discovery
Updated version
10.1158/2159-8290.CD-NB2016-127doi:
Access the most recent version of this article at:
E-mail alerts related to this article or journal.Sign up to receive free email-alerts
Subscriptions
Reprints and
To order reprints of this article or to subscribe to the journal, contact the AACR Publications Department at
Permissions
Rightslink site. Click on "Request Permissions" which will take you to the Copyright Clearance Center's (CCC)
.http://cancerdiscovery.aacrjournals.org/content/6/12/1298To request permission to re-use all or part of this article, use this link
on July 26, 2020. © 2016 American Association for Cancer Research. cancerdiscovery.aacrjournals.org Downloaded from
Published OnlineFirst October 17, 2016; DOI: 10.1158/2159-8290.CD-NB2016-127
Related Documents


![A Nobel Prize for Asymmetric Information:cob.jmu.edu/rosserjb/Nobel Prize. Akerlof-Spence-Stiglitz... · Web view[Review of Political Economy, forthcoming] A Nobel Prize for Asymmetric](https://static.cupdf.com/doc/110x72/5aaecdfd7f8b9adb688cd0a5/a-nobel-prize-for-asymmetric-informationcobjmuedurosserjbnobel-prize-akerlof-spence-stiglitzweb.jpg)