10/ MPN / MARCH | APRIL 2012 However, the most obvious outcome of the event is synergy, the buzz word that describes when the combined effect of a merger is greater than the sum of its parts. And aſter analysing what Phillips and Medisize have to offer in terms of technology, innovation and regional prowess, synergy really is the only word that springs to mind. Already the combination of key strengths are shining through — Medisize is renowned for its industry-leading automated assembly, particularly in the lucrative mid to high volume assembly runs while Phillips’s expertise in multi- shot moulding is second to none. To give you the full picture, Bill Welch, the newly formed company’s Chief Technical Officer, and Ma Jennings, President and CEO, write exlusively for MPN. Bill Welch, Chief Technical Officer on Multi-Material Moulding and Automation Multi-Material Moulding Multi-material (multi- shot) moulding has been extensively used in the commercial markets for over 25 years, but has had slower adoption in medical. When properly integrated into a design, it can be the elegant solution to reduce overall cost and improve quality, being mindful to balance the upfront tooling costs with the manufacturing savings and quality improvement. While multi-material moulding was initially focused on two materials, we today have examples of four materials being used in a single injection mould. In cases such as this, multiple benefits are being realised in a single part, including integrated sealing, reduced part count (eliminates assembly), integrated graphics (eliminates secondary printing operations), soſt touch (improves tactile feel and grip) and dampening — sound and vibration isolation. Another specialty within multi-material moulding is the creation of a Moulded Interconnect Device (MID). An MID is created by moulding two materials — one that can be plated and one that resists plating — to form a 3D circuit board. is can be very effective in eliminating the need for multiple flex or hardboard circuits, and provides the added benefit of creating complex shapes. Phillips-Medisize has also developed a system for hardcoat (a common requirement for PC lenses) that enables digital printing of the hardcoat, effectively reducing cost and lead time, and masking requirements when a multi-material component requires a hardcoat. << e OmniPod is a modern diabetic meter and drug delivery device. It contains a multi- material, multi-shot MID electronic component manufactured by Phillips-Medisize. >> Automated Assembly e Phillips-Medisize approach is to offer customers both technology and market-focused facilities in multiple, convenient locations. Consistent with this approach is the development of Centers of Excellence for varying levels of automated assembly. Several Phillips-Medisize facilities have unique expertise in advanced assembly, for example, the new Richmond, Wisconsin facility is focused on lower volume, complex single-use products, and heavily leverages low volume “smart” assembly. e Menomonie facility, also in Wisconsin, is focused on mid- to mid-high volumes and has demonstrated expertise in complex flexible automation cells with many operations, and higher volume automation modules with compact machine footprints. World English Dictionary synergy — n , pl -gies 1. Also called: synergism, the potential ability of individual organisations or groups to be more successful or productive as a result of a merger 2. e interaction of two or more agents or forces so that their combined effect is greater than the sum of the effect of their individual parts. [C19: from New Latin synergia, from Greek sunergos; see synergism] COVER STORY e plant in Kontiolahti, Finland, is focused on high volume, fully automated assembly and is best-in-class in managing and validating complex systems that have many moulded and purchased components. All Phillips-Medisize facilities leverage robotics and press-side automation to reduce cost and lead time by working within the machine cycle time. Customised production — partnerships built on innovation e primary benefit of the Phillips-Medisize approach is to develop a customised solution based on programme volume, compexity and risk. By offering our customers options from manual assembly in low-cost countries to highly automated and engineered solutions, Phillips- Medisize is able to select a manufacturing strategy that is in the customer’s best interest. Further, by gaining early involvement via its design centres, Phillips-Medisize can scale operations, starting with low-volume clinical or pilot builds and ending at the desired scale, managing capital cost and manufacturing risk along the way. A meeting of minds Ma Jennings, President and CEO on Partnerships rough Innovation Today there’s continued pressure for our drug delivery and medical device manufacturers to innovate with new products appropriate for the global markets to drive top line growth, continually improve quality, stay compliant with the changing regulatory landscape and simplify the supply chain to reduce costs. at is a mouthful but true. As a result, our customers are looking for partners that understand the market challenges and that can be a valued resource to help them achieve their goals. Partners that can help them design, develop and industrialise their ideas to help with accelerating products’ time to market is critical. Our customers are also looking for partners that can reduce supply chain length and complexity by offering locations close to end markets. Partners that proactively find ways to take out costs in the supply chain and improve quality is always a requirement. at’s what Phillips–Medisize does well every day. We take a customer-centric approach and align our strategy from design and development through manufacturing with what they’re trying to accomplish around the globe. But true partnerships need to be a two-way street, with the OEM willing to share their strategy and objectives, be open about resource constraints and limitations and have a willingness to share the responsibility. Medical devices are becoming smaller and smarter and OEMs are placing more focus on cosmetic and human factor engineering into the product design in addition to the traditional form, function and fit of medical design elements. As a result, drug delivery devices and diagnostic devices that must maintain very high tolerances and quality standards while incorporating cosmetic finishes is something we see more and more, particularly with high-volume patient use items. Another important development is people are increasingly integrating electronics into their devices. It’s something that we’ve been positioned well to do with our advanced capabilities in multi- shot moulding, automated assembly and quality control and advanced inline coatings and finishing technologies to help achieve the desired design intent and user experience. Today we offer advanced multi-material (multi-shot) capabilities that include up to four different materials in a single, complex mould. We also have the capability to have one or two of the materials be a silicone. is provides an elegant solution to sealing issues, while reducing part count and creating a shorter supply chain. Also, our metal injection moulding (MIM) is a great process for components that require tight tolerances, with surface finish that is much improved compared with the industry’s early years. On hearing the news of the merger between Phillips and Medisize, a number of keywords sprung to mind, such as Euro-US transatlantic cooperation, what a great idea and why didn’t they do it earlier? What I didn’t expect was that the new logo would be everywhere at MD&M West, including on the room key of the Marriot hotel at the Anaheim Convention Center. << e images show the automation capabilities of the company. Notice the blur in the first image, reflecting the speed at which devices are being assembled. >>

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

10/ MPN / MARCH | APRIL 2012
However, themost obviousoutcome of theevent is synergy,the buzz wordthat describeswhen thecombined effectof a merger isgreater than thesum of its parts.And afteranalysing whatPhillips and
Medisize have to offer in terms of technology, innovationand regional prowess, synergy really is the only word thatsprings to mind.
Already the combination of key strengthsare shining through — Medisize is renownedfor its industry-leading automated assembly,particularly in the lucrative mid to high volumeassembly runs while Phillips’s expertise in multi-shot moulding is second to none. To give youthe full picture, Bill Welch, the newly formedcompany’s Chief Technical Officer, and MattJennings, President and CEO, write exlusively forMPN.
Bill Welch, Chief Technical Officer on Multi-MaterialMoulding and Automation
Multi-Material MouldingMulti-material (multi-
shot) moulding has beenextensively used in thecommercial markets for over25 years, but has had sloweradoption in medical. Whenproperly integrated into adesign, it can be the elegantsolution to reduce overallcost and improve quality,being mindful to balance the upfront tooling costs with themanufacturing savings and quality improvement. Whilemulti-material moulding was initially focused on twomaterials, we today have examples of four materials beingused in a single injection mould. In cases such as this,multiple benefits are being realised in a single part,including integrated sealing, reduced part count (eliminatesassembly), integrated graphics (eliminates secondaryprinting operations), soft touch (improves tactile feel and
grip) and dampening — sound and vibration isolation.Another specialty within multi-material moulding is the
creation of a Moulded Interconnect Device (MID). An MIDis created by moulding two materials — one that can beplated and one that resists plating — to form a 3D circuitboard. This can be very effective in eliminating the needfor multiple flex or hardboard circuits, and provides theadded benefit of creating complex shapes.
Phillips-Medisize has also developed a system forhardcoat (a common requirement for PC lenses) thatenables digital printing of the hardcoat, effectively reducingcost and lead time, and masking requirements when amulti-material component requires a hardcoat.
<< The OmniPod is a modern diabetic meter and drug delivery device. It contains a multi-
material, multi-shot MID electronic componentmanufactured by Phillips-Medisize. >>
Automated AssemblyThe Phillips-Medisize approach is to offer customers
both technology and market-focused facilities in multiple,convenient locations. Consistent with this approach is thedevelopment of Centers of Excellence for varying levels ofautomated assembly.
Several Phillips-Medisize facilities have unique expertisein advanced assembly, for example, the new Richmond,Wisconsin facility is focused on lower volume, complexsingle-use products, and heavily leverages low volume“smart” assembly.
The Menomonie facility, also in Wisconsin, is focused onmid- to mid-high volumes and has demonstrated expertisein complex flexible automation cells with many operations,and higher volume automation modules with compactmachine footprints.
World English Dictionarysynergy
— n , pl -gies1. Also called: synergism, the potential ability of individual organisations or groups to be more successful or productive as a result of a merger2. The interaction of two or more agents or forces so that their combined effect is greater than the sum of the effect of their individual parts.
[C19: from New Latin synergia, from Greek sunergos; see synergism]
COVER STORY
The plant in Kontiolahti, Finland, is focused on highvolume, fully automated assembly and is best-in-class inmanaging and validating complex systems that havemany moulded and purchased components.
All Phillips-Medisize facilities leverage robotics andpress-side automation to reduce cost and lead time byworking within the machine cycle time.
Customised production — partnerships built oninnovation
The primary benefit of the Phillips-Medisize approach isto develop a customised solution based on programmevolume, compexity and risk. By offering our customersoptions from manual assembly in low-cost countries tohighly automated and engineered solutions, Phillips-Medisize is able to select a manufacturing strategy that is inthe customer’s best interest. Further, by gaining earlyinvolvement via its design centres, Phillips-Medisize can scaleoperations, starting with low-volume clinical or pilot buildsand ending at the desired scale, managing capital cost andmanufacturing risk along the way.
A meeting of mindsMatt Jennings, President and CEOon Partnerships ThroughInnovation
Today there’s continued pressurefor our drug delivery and medicaldevice manufacturers to innovate withnew products appropriate for theglobal markets to drive top linegrowth, continually improve quality,stay compliant with the changing
regulatory landscape and simplify the supply chain to reducecosts. That is a mouthful but true.
As a result, our customers are looking for partners thatunderstand the market challenges and that can be a valuedresource to help them achieve their goals. Partners that canhelp them design, develop and industrialise their ideas to helpwith accelerating products’ time to market is critical.
Our customers are also looking for partners that canreduce supply chain length and complexity by offeringlocations close to end markets. Partners that proactively findways to take out costs in the supply chain and improve qualityis always a requirement. That’s what Phillips–Medisize doeswell every day. We take a customer-centric approach andalign our strategy from design and development throughmanufacturing with what they’re trying to accomplish aroundthe globe. But true partnerships need to be a two-way street,with the OEM willing to share their strategy and objectives, beopen about resource constraints and limitations and have awillingness to share the responsibility.
Medical devices are becoming smaller and smarter andOEMs are placing more focus on cosmetic and human factorengineering into the product design in addition to thetraditional form, function and fit of medical design elements.As a result, drug delivery devices and diagnostic devices thatmust maintain very high tolerances and quality standards whileincorporating cosmetic finishes is something we see more andmore, particularly with high-volume patient use items. Anotherimportant development is people are increasingly integratingelectronics into their devices. It’s something that we’ve beenpositioned well to do with our advanced capabilities in multi-shot moulding, automated assembly and quality control andadvanced inline coatings and finishing technologies to helpachieve the desired design intent and user experience.
Today we offer advanced multi-material (multi-shot)capabilities that include up to four different materials in asingle, complex mould. We also have the capability to haveone or two of the materials be a silicone. This provides anelegant solution to sealing issues, while reducing part countand creating a shorter supply chain. Also, our metal injectionmoulding (MIM) is a great process for components thatrequire tight tolerances, with surface finish that is muchimproved compared with the industry’s early years.
On hearing the news of themerger between Phillips and
Medisize, a number of keywordssprung to mind, such as Euro-US
transatlantic cooperation, what agreat idea and why didn’t they
do it earlier? What I didn’t expectwas that the new logo would be
everywhere at MD&M West,including on the room key of the
Marriot hotel at the AnaheimConvention Center.
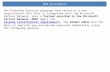
<< The images show the automation capabilities of thecompany. Notice the blur in the first image, reflecting the
speed at which devices are being assembled. >>
MPN 5 Progress_BP&R New Grid 02/04/2012 11:24 Page 10
Related Documents