Management of Low Grade Gliomas (LGG) Current & Future Trends Dr. Gaurav Kumar Narayana Superspeciality Hospital

Low grade glioma evidence based management
Aug 07, 2015
Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

Management of Low Grade Gliomas (LGG)Current & Future Trends
Dr. Gaurav KumarNarayana Superspeciality Hospital

Gliomas are divided into low grade and high grade based on: nuclear abnormalities, mitoses, endothelial
proliferation, and necrosis.
Currently Most widely used: WHO grading system
WHO Grade I lesions have low proliferative potential.WHO Grade II neoplasms are infiltrative, recur, and
tend to progress to higher grades of malignancy despite low
levelproliferative activity
WHO Grade III & IV: High grade Gliomas with marked nuclear atypia & high mitotic counts.

Grade I: Pilocytic Astrocytoma, typically have very slow growth rate, rare mitosis &rarely transforms and possibile to be cured by surgery alone.
Grade II: Diffusely infiltrative low-grade astrocytomas include the fibrillary, protoplasmic, and gemistocytic types.
This group poses the challenge: They can recur/ProgressThey can transform to high gradeThey occur in relatively younger population
So, there lies the true need of
Multimodal treatment

Most oligoastrocytomas and 50% to 75% of oligodendrogliomas recur as AAs or GBM.
Tumor Aggressiveness can be assessed with Ki-67 labeling.
Multiple studies has revealed correlation between high Ki-67/MIB 1 labeling index with increasing grade of malignancy.

Genetic Makeup :
1p and 19q codeletion : Associated with better prognosis and are common in oligodendrogliomas. TP53 mutations: Common in diffuse astrocytomas and aremutually exclusive from 1p/19q co-deletions.
IDH1 mutations found both in tumors with TP53 mutations and in tumors with1p/19q co-deletions.

Case Summary:

Clinical:42 yrs old right handed male P/W: 04 months history of on-off headache 01 month history of focal seizuresNo comorbidities
MRI Findings:Hypo-intense on T1, Hyper-intense on T2/FLAIR , Non-enhancing, Well-circumscribed, solid SOL,Size- 5.5x6 cm,Not crossing midline.
Surgery:Maximum Safe Resection

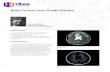
Images:
A: T1 Non Contrast B: T2 C:T1 Contrast

HP:Mixed Oligoastrocytoma; WHO Grade II
IHC:1p&19q Codeleted
Postop MRI: Small residual disease with moderate perilesional edema & P/O Changes. Clinically patient is asymptomatic, no neurological deficit.
Now what?

Questions needed to be addressed:
1] Immediate Postop RT vs RT at Disease Progression?
2] Dose of RT?
3] RT Alone or RT + Chemotherapy?
4] If Chemotherapy added to RT- Which Chemotherapy?
5] RT alone vs Chemotherapy alone at Disease progression?
6] Disease Progression after PORT?

1] Immediate vs delayed PORT
Evidence
Phase III adult low grade glioma trials (EORTC 22844 and
22845): Risk Factors identified & Validated
Age>40 yearsSize>6cm
Crossing MidlinePure Astrocytoma histology
Neurological deficit before Surgery
Low Risk Patient: </= 2 factors (Median Survival- 7.7 years)
High Risk: 3 or more factors (Median Survival- 3.2 years)

EORTC 22845 (Karim et al, 2002 & Van den Bent et al, 2005)
Randomised phase III trialRT Dose (54Gy/30#)Immediate RT vs RT at Progression
Results: Improved median progression free survival(5.3 yrs vs 3.4 yrs)Better seizure control ratesNo difference in Median survival (7.4yrs vs 7.2 yrs) No difference in rate of malignant transformation.
Pitfall: No in-depth quality of life adjusted analysis.

RTOG 9802 (phase II portion of protocol)
Risk Factors predictive of a poorer PFS Astrocytoma histology Residual tumor of >/=1 cm on Postop MR Pre-operative tumor diameter of >/=4 cm
Patients with:All three unfavourable factors- PFS at 5years 13%None of the three factors- PFS at 5years 70%

So, on the basis of above data
Observation seems to be a reasonable strategy for the most favorable subset i.e.
<1 cm residual tumorPreoperative tumor diameter <4 cmOligodendroglioma histologyYounger patients Following a gross total resection (GTR).
Mature result of this trial is pending !!!!

2] Dose of RT?
Evidence
EORTC 22844 (Karim et al. 1996) – phase III:Postoperative RT 45 Gy vs. 59.4 Gy
5-year OS 58% with 45 Gy 59% with 59.4 Gy.
INT/NCCTG (Shaw et al. 2002) – phase III:Postoperative RT 50.4 Gy vs. 64.8 Gy
5-year OS 73% with 50.4 Gy 68% with 64.8 Gy.

Based on these Phase III trials and Extrapolation of data of in-field recurrences in high grade gliomas
It will be prudent to limit the Postoperative RT Dose to 54 Gy.

3] RT Alone or RT + Chemotherapy?
Evidence
INT/RTOG 9802 trial (ASCO abstract 2008): phase III
Low-risk (<40 year + GTR) observed until symptoms
High-risk (>40 year or STR or biopsy) patients randomized to RT alone vs. RT --> PCV ×6 cycles q8 weeks
5 year OS was 72 vs. 63% (p = 0.33) 5-year PFS was 63 vs. 46%(p = 0.06) in favour of chemotherapy

Largest reported retrospective analysis of 149 patients
Temozolomide at Progression (1p/19q LOH was present in 42%)
53% - Objective response (15% - Partial response and 38% minor response) 37% - Stable disease10% - progressive disease.
Kaloshi G, Benuaich-Amiel A, Diakite F, et al: Temozolomide for low grade gliomas: predictive impact of 1p/19q loss on response and outcome. Neurology 2007; 68:1831-1836

Phase II Trial of Temozolomide in Patients With Progressive Low-Grade Glioma(Jennifer A. Quinn et al)Objective response rate - 61% (24% CR and 37% PR)Stable disease - 35%
IDH1 or IDH2 mutations predict longer survival and response to temozolomide in low-grade gliomas.(C. Houillier et al) Neurology October 26, 2010 vol. 75 no. 17 1560-1566
1p-19q codeletion, MGMT promoter methylation, and IDH mutation (p = 0.01) were correlated with a higher rate of response to temozolomide

EORTC 22033-26033/CE5 phase III randomized trial for low grade glioma: Phase III EORTC 22033-26033/NCIC CE5 intergroup trial compares 50.4 Gy radiotherapy with up-front temozolomide
in previously untreated low-grade glioma(Open to accrual)
Conclusion:
Low-grade gliomas respond to temozolomide
Loss of chromosome 1p/19q predicts both a durable chemosensitivity and a favorable outcome

4] If Chemotherapy added to RT- Which Chemotherapy?
Concerns about toxicity profile of PCV
Nitrosoureas (In PCV) – Notorious for secondary malignancy
Procarbazine - Infertility
Availability of lesser toxic and effective substitute as Temozolomide
Oral administration- Convenient dosing of Temozolomide
Makes Temozolomide more preferable an option with respect to PCV chemotherapy

5] RT alone vs Chemotherapy alone at Disease progression?
Head to Head trial lacking
HOANG-XUANK., CAPELLEL. et al. : Temozolomide as initial treatment for adults with low-grade oligo-dendrogliomas or oligoastrocytomas and correla-tion with chromosome 1p deletions. J. Clin.Oncol.,
2004, 22(15) : 3133-8.
BRADA M., VIVIERS L. et al. Phase II study of primary temozolomide chemotherapy in patients with WHO grade II
gliomas. Ann. Oncol., 2003, 14(12) : 1715-21

Ann Oncol. 2003 Dec;14(12):1722-6.Temozolomide chemotherapy for progressive low-grade glioma: clinical benefits and radiological response.Pace A et. al.
High response rate of 47% (95% CI 31% to 61%) confirms that TMZ chemotherapy is a valid option in the treatment of progressive LGG specially in children below 5 years age.
For Adults: Replacing RT with chemotherapy at progression need more robust & head to head comparable data.

6] Disease Progression after PORT?
Options Include
Resurgery (If resectable)
Chemotherapy (Unresectable disease)
Reirradiation with SRS/FSRT (Small recurrences)
Newer agents under trial (Blocking mTOR with an investigational agent ridaforolimus ).

Take Home Massage:
1] Immediate PORT High Risk Low Grade Glioma (Astrocytoma histology, Residual >1 cm, Preop tumour > 4/6 cm, Age >40 years).
2] Dose of PORT – 54 Gy/30# (No role of dose escalation)
3] Adjuvant PCV/ Temozolomide –Still not standard of care but can be considered in patients with High Risk Disease with 1p & 19q codeleted & IDH1/2 mutated (Subject to individual risk benefit assessment).
4] Radiotherapy still standard of care (above chemotherapy) in adult patients with progressive disease (Patient kept under observation after surgery)
5] Chemotherapy with Temozolomide is equally effective and less toxic as compared to PCV and can be incorporated as adjuvant, salvage therapy.

Thank You
Related Documents