Project Project Laser Facial Nerve Laser Facial Nerve Welding in a Rabbit Welding in a Rabbit Model Model Jason D. Bloom, M.D. Jason D. Bloom, M.D. University of Pennsylvania University of Pennsylvania Department of Otorhinolaryngology: Department of Otorhinolaryngology: Head & Neck Surgery Head & Neck Surgery June 25 June 25 th th , 2009 , 2009 Faculty Mentors: Noam A. Cohen, M.D. & Faculty Mentors: Noam A. Cohen, M.D. & Stephen A. Goldstein, M.D. Stephen A. Goldstein, M.D. Special Thanks: Benjamin S. Bleier, M.D. Special Thanks: Benjamin S. Bleier, M.D. Otorhinolaryngology: Head and Neck Surgery at PENN Excellence in Patient Care, Education and Research since 1870

Laser Facial Nerve Welding in a Rabbit Model - Temporal Bone ...
Jan 12, 2015
Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

Resident Final Research Project Resident Final Research Project
Laser Facial Nerve Welding Laser Facial Nerve Welding in a Rabbit Modelin a Rabbit Model
Jason D. Bloom, M.D.Jason D. Bloom, M.D.University of PennsylvaniaUniversity of Pennsylvania
Department of Otorhinolaryngology: Department of Otorhinolaryngology: Head & Neck SurgeryHead & Neck Surgery
June 25June 25thth, 2009, 2009
Faculty Mentors: Noam A. Cohen, M.D. & Faculty Mentors: Noam A. Cohen, M.D. & Stephen A. Goldstein, M.D.Stephen A. Goldstein, M.D.
Special Thanks: Benjamin S. Bleier, M.D.Special Thanks: Benjamin S. Bleier, M.D.Otorhinolaryngology: Head and Neck Surgery at PENN
Excellence in Patient Care, Education and Research since 1870

Research Approval & FundingResearch Approval & FundingThis research project has been approved by both the U of Penn IACUC (Protocol # 802421) & Philadelphia VA ACORP
Awarded 2009-2010AAO-HNSF / AAFPRS Leslie Bernstein Resident Research CORE Grant

BackgroundBackgroundCharles Gould (1957) - Introduces the term LASER (Light Amplification by Stimulated Emission of Radiation)
– In his doctoral thesis on energy levels of excited thallium
Sigel & Acevado (1962) - Reported thermal energy in tissue adhesion
– Passing high frequency electric current through coapted vein edges to anastomose end-to-side porto-caval shunts in dogs (1)
Yahr & Strully (1966) – First description of laser tissue welding in blood vessel anastomosis (2)
– Nd-YAG laser
Almquist et al. (1984) – Argon laser in peripheral nerve repair (3)
Fischer et al. (1985) – Published positive reports using the CO2 laser for rat nerve repair (4)

BackgroundBackgroundBiologic Solder Biologic Solder
Allows for target specific laser energy absorption resulting in tissue bonds with greater tensile strength than wounds sealed with laser energy alone (5)
The use of biologic solders has been shown to provide an added benefit in the promotion of native wound healing mechanisms.
– Contrast to the granulomatous inflammatory response seen with suture material
– The lased solder coagulum provides a non-immunogenic scaffold (6)
– Gradually absorbed during the normal wound healing process (7,8)
Biologic solders are now being combined with wavelength specific chromophores (9,10)
- Increased target specific energy absorption - Decreased thermal energy leakage - Objective basis of gauging adequacy of laser welding by providing a predictable color change - Examples: Carbon Black 808nm
Fluorescein Dye 532nm
Indocyanine Green Dye 805nm

Laser Tissue WeldingLaser Tissue Welding
Laser Biologic Solder Chromophore
ECM REMODELING
LASER WELDLASER WELD

BackgroundBackground
Problems associated w/ classic neurorrhaphy
– Trauma induced by handling & sutures– Neuroma formation– Long OR times & technically difficult surgically– “Leakage” of axons at site of anastomosis &
in-growth of connective tissue– Foreign body reaction from suture material

BackgroundBackgroundLaser Nerve Welding
Eppley et al. (1989) – Laser nerve repair of the rabbit facial nerve with CO2 laser (11)– neuroma formation– connective tissue invasion– axonal proliferation or extravasation outside of epineurium
– Problems:• Histopathologic weld analysis
• Demonstrated some deleterious effects of the heat from the laser, such as destruction of myelin and loss of axons immediately adjacent to the anastomotic site.
• Poor anastomosis tensile strength & dehiscence

BackgroundBackgroundLaser Nerve Welding
Trickett et al. (1997) – Albumin based solder with indocyanine green chromophore to laser weld rat sciatic nerves with an 800nm diode laser (12)- Laser activated solders = stronger bonds at the anastomosis
site & less thermal damage to underlying nerve tissue
- Histology = denatured solder and underlying epineurium with no axonal damage
Hwang et al. (2005 & 2006) – CO2 laser welding of rat facial nerve w/o solder (13,14)
- Laser repaired nerve regeneration = microsuture repaired nerve regeneration immmunohistochemical nerve tracer
- Less cellular & fibroblastic response- Less scar & neuroma formation

HypothesisHypothesisLaser welding of the rabbit facial nerve provides a method of creating an atraumatic, immediate and strong repair bond capable of allowing axonal regeneration across the injury site without damage to the nerve itself or the complications associated with suture neurorrhaphy.
Null Hypothesis: Laser nerve welding is not an effective way to perform neurorrhapy, as compared to suture nerve repair.

Time TableTime TableDay Procedure Data Point -
Functional testing
Data Point – EMG testing
Data Point - Histology (H&E and Masson’s Trichrome Stain)
0 Rabbit Acquisition N/A N/A N/A
4 Rabbit Acclimation N/A N/A N/A
5 Survival Surgery/ Functional & EMG tests
Laser Weld x 6, Suture x 6, Neg Control x 2
Laser Weld x 6, Suture x 6, Neg Control x 2
N/A
33 (wk 4) Functional & EMG tests/ Sac-Experimental Group #1
Laser Weld x 6, Suture x 6, Neg Control x 2
Laser Weld x 6, Suture x 6, Neg Control x 2
Laser Weld x 2, Suture x 2
61 (wk 8) Functional & EMG testing
Laser Weld x 4, Suture x 4, Neg Control x 2
Laser Weld x 4, Suture x 4, Neg Control x 2
N/A
89 (wk 12) Functional & EMG testing
Laser Weld x 4, Suture x 4, Neg Control x 2
Laser Weld x 4, Suture x 4, Neg Control x 2
N/A
117 (wk 16)
Functional & EMG tests/ Sac-Experimental Group #2
Laser Weld x 4, Suture x 4, Neg Control x 2
Laser Weld x 4, Suture x 4, Neg Control x 2
Laser Weld x 4, Suture x 4, Neg Control x 2

Rabbit Survival SurgeryRabbit Survival Surgery2 of the rabbit facial nerves (negative control group) = facial nerves transected with a 1cm piece of nerve excised w/o re-anastomosis
6 of the rabbits facial nerves = suture anastomosis w/ three 9-0 monofilament nylon sutures on an atraumatic taper needle
6 of the rabbit facial nerves = laser welded utilizing an 810nm diode laser and a “ribbon” of 42% albumin-based biologic solder coupled with an indocyanine green dye chromophore applied with a 27 G needle
14 New Zealand White Rabbits (3-4kg) – Housed at the Animal
Research Facility (ARF) of the Philadelphia Veterans Affairs Medical Center (PVAMC)

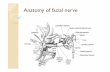
Rabbit Facial Nerve AnatomyRabbit Facial Nerve Anatomy

OR SetupOR Setup

Identifying the Facial NerveIdentifying the Facial Nerve
Hand-held nerve stimulator was usedto identify the facial nerve and verify facial movement.
* Also, used to verify that the facial nerve was cut no longer innervating the face

Suture NeurorrhaphySuture Neurorrhaphy
Suture anastomosis w/ three Suture anastomosis w/ three 9-0 monofilament 9-0 monofilament polypropylene (Prolene) polypropylene (Prolene) sutures on an atraumatic sutures on an atraumatic taper needletaper needle
* Carl Zeiss OPMI CS-1 * Carl Zeiss OPMI CS-1 operating microscope operating microscope

Laser/Solder PlatformLaser/Solder Platform
Biologic solder - 42% albumin solutionIndocyanine green
dyeHyaluronic acid
sodium
Diode laser module- Iridex Oculight SLx Power: 0.5WPulse Duration: 0.5sPulse Interval: 0.1sPower Density: 15.9W/cm2Fluency: 8.0 J/cm2Major Wavelength Output: 810 +/- 1nm
* 600mm core diameter quartz-silica fiberoptic cable

Laser Solder NeurorrhaphyLaser Solder Neurorrhaphy
Microvascular clip – Microvascular clip – used to hold used to hold nerve ends in placenerve ends in place
Adequacy of weld was visually Adequacy of weld was visually assessed by the green colored assessed by the green colored chromophore changing to a brown chromophore changing to a brown color s/p laser energycolor s/p laser energy

Surgical ComplicationsSurgical Complications
Surgical Complications:Sialocele (5/14 rabbits = 35%)
* All treated w/ repeated 20G needle aspirations all completely resolved in 2wks
* No pain, distress, feeding issues, effect on wt. gain

EMG TestingEMG Testing
Nicolet Viking IV EMGNicolet Viking IV EMG- subdermal electrodes- subdermal electrodes
Nerve Conduction StudyNerve Conduction Study- measuring CMAPs- measuring CMAPs- amplitude- amplitude- nerve latency- nerve latency

Functional / Clinical TestingFunctional / Clinical Testing
Positioning of the rabbits that allows standardized visualization and video recording of facial movements.
Gentle nasal & forehead taps performed in an attempt to elicit facial nerve motion.
Video tapes will then be analyzed to objectively grade facial nerve recovery by 2 different blinded observers.
Assess the rabbit's bilateral upper lip & whisker movement.
A scale of facial nerve movement from 0-3 will be used to record this functional testing with 0=no movement and 3=normal facial movement (16).
Normal Facial Nerve Function
Left Facial Nerve Paralysis

Histological TestingHistological Testing6 nerve repairs from each experimental group were harvested and fixed in 10% buffered formalin for subsequent H&E staining
Analysis by our histopathologist (Eugene Einhorn, MD) for immune response to the solder, native wound healing progression, and extent of collateral nerve thermal injury.
The welds were also be prepared and stained with Masson’s Trichrome by our histopathologist, in order to view the axon counts and arrangement of nerve axons across the repair sites.
Nerve Laser Weld (H&E 4x) – 4wks
* 16 week histological analysis = ongoing(1) Axon counts growing across the nerve repair site (2) Organization of the axons (3) Amount of neuroma formation (4) Amount of foreign body/granulomatous reaction

RESULTSRESULTS

Operative TimeOperative Time
*
* P < 0.01
N=6 N=6

Learning CurveLearning Curve

EMG Nerve Recovery EMG Nerve Recovery

Clinical Nerve RecoveryClinical Nerve Recovery

Problems with the StudyProblems with the Study• Multiple buccal branches of the rabbit facial nerve distal to the site of the nerve injury & repair
• Small number of rabbits in each group
• Inability to circumferentially laser weld the nerve
• Inconsistent EMG electrode placement in the facial musculature for each separate trial
• No adequate method to measure synkinesis in a rabbit model
•Testing EMG vs. ENoG

Discussion / Discussion / Future DirectionsFuture Directions
Further areas of research with this laser welding technology:– Peripheral nerve repair
– Microvascular arterial or venous anastomosis
– Pharyngocutaneous fistula repair
– Transoral robotic surgery models
Augmentation of the biologic solder with growth factors and antibiotics to further optimize wound healing
If these welds prove efficacious in larger trials, this technology would be ideally transitioned to clinical trials of facial nerve injury repair.

LNW ConclusionsLNW ConclusionsClinical / Functional:― Improved facial nerve functional recovery on video analysis with the laser weld
group
Electrophysiological:― Improved facial nerve electrophysiological/EMG nerve recovery at all time points
in the laser weld group, especially at early (1 month) & late (4 month) time points
Histological:― H&E staining demonstrating negligible collateral thermal nerve or tissue damage
― Biologic solder supporting the progression of the native wound healing response
― Ongoing histopath: Utilizing Masson’s Trichrome stain (1) Axon counts growing across the nerve repair site (2) Organization of the axons (3) Amount of neuroma formation (4) Amount of foreign body/granulomatous reaction
Mechanical:― No learning curve or operating microscope necessary with the laser weld group
― Significantly reduced neurorrhaphy time with the laser weld group

ReferencesReferences1. Sigel B, Acevado FJ, “Vein anastomosis by electrocoaptive union,” Surg Forum. 1962;13:233-5.
2. Yahr WZ, Strully KJ, “Blood vessel anastamosis by laser and other biomedical applications,” J Assoc Adv Med Instrum. 1966;(1):28-31.
3. Almquist EE, Nachemson A, Auth D, Almquist B, Hall S. “Evaluation of the use of the argon laser in repairing rat and primate nerves,” J Hand Surg Am. 1984;9:792-299.
4. Fischer DW, Beggs JL, Kenshalo DJ, Shetter AG. “Comparative study of microepineurial anastomoses with the use of CO2 laser and suture techniques in rat sciatic nerves: Part 1. Surgical technique, nerve action potentials, and morphological studies,” Neurosurgery. 1985;17:300-308.
5. Gil Z, Shaham A, Vasilyev T, Brosh T, Forer B, Katzir A, Fliss DM. “Novel laser tissue-soldering technique for dural reconstruction,” J Neurosurg. 2005 Jul;103(1):87-91.
6. Kirsch AJ, Miller MI, Hensle TW, Chang DT, Shabsigh R, Olsson CA, Connor JP, “Laser tissue soldering in urinary tract reconstruction: first human experience,” Urology. 1995 Aug;46(2):261-6.
7. Lauto A, Trickett R, Malik R, Dawes JM, Owen ER, “Laser-activated solid protein bands for peripheral nerve repair: an vivo study,” Lasers Surg Med. 1997;21(2):134-41.
8. Lauto A, Kerman I, Ohebshalon M, Felsen D, Poppas DP, “Two-layer film as a laser soldering biomaterial,” Lasers Surg Med. 1999;25(3):250-6.
9. Talmor M, Bleustein CB, Poppas DP, “Laser tissue welding: a biotechnological advance for the future,” Arch Facial Plast Surg. 2001 Jul-Sep;3(3):207-13.
10. Oz MC, Johnson JP, Parangi S, Chuck RS, Marboe CC, Bass LS, Nowygrod R, Treat MR, “Tissue soldering by use of indocyanine green dye-enhanced fibrinogen with the near infrared diode laser,” J Vasc Surg. 1990 May;11(5):718-25.
11. Eppley BL, Kalenderian E, Winkelmann T, Delfino JJ. “Facial nerve graft repair: suture versus laser-assisted anastomosis,” Int J Oral Maxillofacial Surg. 1989;18:50-54.

ReferencesReferences12. Trickett I, Dawes JM, Knowles DS, Lanzetta M, Owen ER. “In vitro Laser Nerve Repair: Protein Solder Strip Irradiation or Irradiation Alone?,” Int Surg. 1997;82:38-41.
13. Hwang K, Kim SG, Kim DJ, Lee CH. “Laser Welding of Rat’s Facial Nerve,” Journal of Craniofacial Surgery. 2005;16(6):1102-1106.
14. Hwang K, Kim SG, K DJ. “Facial-Hypoglossal Nerve Anastomosis Using Laser Nerve Welding,” Journal of Craniofacial Surgery. 2006;17(4):687-691.
15. Yian CH, Paniello RC, Spector JG. “Inhibition of Motor Nerve Regeneration in a Rabbit Facial Nerve Model.” Laryngoscope. 2001;111:786-791.
16. Bleier BS, Palmer JN, Sparano AM, et al. “Laser-assisted cerebrospinal fluid leak repair: an animal model to test feasibility.” Otolaryngol Head Neck Surg. 2007;137(5):810-4.
17. Bleier BS, Palmer JN, Gratton MA, Cohen NA. “In Vivo Laser Tissue Welding in the Rabbit Maxillary Sinus.” American Journal of Rhinology. 2008;22:625-628.
18. Bleier BS, Gratton MA, Leibowitz JM, Palmer JN, Newman JG, Cohen NA. “Laser-Welded Endoscopic Endoluminal Repair of Iatrogenic Esophageal Perforation: An Animal Model.” Otolaryngology – Head and Neck Surgery. 2008;139:713-717.

THANK YOU!THANK YOU!Special Thanks To:Special Thanks To:- My Faculty Mentors- My Faculty Mentors
- Dr. O’Malley- Dr. O’Malley- Dr. Cohen, Dr. Palmer, Dr. Bleier & - Dr. Cohen, Dr. Palmer, Dr. Bleier &
The Penn Rhinology LabThe Penn Rhinology Lab- Perry DeRitis & VA ARF Staff- Perry DeRitis & VA ARF Staff
- VA Neurology Service- VA Neurology Service
Related Documents