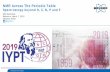
IUPAC Periodic Table of the Isotopes 1 2 H [1.007 84; 1.008 11] hydrogen 1 7 6 Li [6.938; 6.997] lithium 3 9 Be 9.012 182(3) beryllium 4 11 10 B [10.806; 10.821] boron 5 12 13 C [12.0096; 12.0116] carbon 6 14 15 N [14.006 43; 14.007 28] nitrogen 7 16 18 17 O [15.999 03; 15.999 77] oxygen 8 19 F 18.998 4032(5) fluorine 9 20 22 21 Ne 20.1797(6) neon 10 23 Na 22.989 769 28(2) sodium 11 24 26 25 Mg 24.3050(6) magnesium 12 27 Al 26.981 5386(8) aluminium (aluminum) 13 28 29 30 Si [28.084; 28.086] silicon 14 31 P 30.973 762(2) phosphorus 15 32 34 33 36 S [32.059; 32.076] sulfur 16 35 37 Cl [35.446; 35.457] chlorine 17 40 36 38 Ar 39.948(1) argon 18 39 41 40 K 39.0983(1) potassium 19 40 44 42 48 43 46 Ca 40.078(4) calcium 20 45 Sc 44.955 912(6) scandium 21 48 46 47 49 50 Ti 47.867(1) titanium 22 51 50 V 50.9415(1) vanadium 23 52 53 50 54 Cr 51.9961(6) chromium 24 55 Mn 54.938 045(5) manganese 25 56 54 57 58 Fe 55.845(2) iron 26 59 Co 58.933 195(5) cobalt 27 58 60 62 61 64 Ni 58.6934(4) nickel 28 63 65 Cu 63.546(3) copper 29 64 66 68 67 70 Zn 65.38(2) zinc 30 69 71 Ga 69.723(1) gallium 31 74 72 70 76 73 Ge 72.63(1) germanium 32 75 As 74.921 60(2) arsenic 33 80 78 76 82 77 74 Se 78.96(3) selenium 34 79 81 Br 79.904(1) bromine 35 84 86 82 83 80 78 Kr 83.798(2) krypton 36 85 87 Rb 85.4678(3) rubidium 37 88 86 87 84 Sr 87.62(1) strontium 38 89 Y 88.905 85(2) yttrium 39 90 94 92 91 96 Zr 91.224(2) zirconium 40 93 Nb 92.906 38(2) niobium 41 98 96 95 92 100 97 94 Mo 95.96(2) molybdenum 42 Tc technetium 43 102 104 101 99 100 96 98 Ru 101.07(2) ruthenium 44 103 Rh 102.905 50(2) rhodium 45 106 108 105 110 104 102 Pd 106.42(1) palladium 46 107 109 Ag 107.8682(2) silver 47 114 112 111 110 113 116 106 108 Cd 112.411(8) cadmium 48 115 113 In 114.818(3) indium 49 120 118 116 119 117 124 122 112 114 115 Sn 118.710(7) tin 50 121 123 Sb 121.760(1) antimony 51 130 128 126 125 124 122 123 120 Te 127.60(3) tellurium 52 127 I 126.904 47(3) iodine 53 132 129 131 134 136 130 128 124 126 Xe 131.293(6) xenon 54 133 Cs 132.905 4519(2) caesium (cesium) 55 138 137 136 135 134 130 132 Ba 137.327(7) barium 56 139 138 La 138.905 47(7) lanthanum 57 140 142 138 136 Ce 140.116(1) cerium 58 141 Pr 140.907 65(2) praseodymium 59 142 144 146 143 145 148 150 Nd 144.242(3) neodymium 60 Pm promethium 61 152 154 147 149 148 150 144 Sm 150.36(2) samarium 62 153 151 Eu 151.964(1) europium 63 158 160 156 157 155 154 152 Gd 157.25(3) gadolinium 64 159 Tb 158.925 35(2) terbium 65 164 162 163 161 160 158 156 Dy 162.500(1) dysprosium 66 165 Ho 164.930 32(2) holmium 67 166 168 167 170 164 162 Er 167.259(3) erbium 68 169 Tm 168.934 21(2) thulium 69 174 172 173 171 176 170 168 Yb 173.054(5) ytterbium 70 175 176 Lu 174.9668(1) lutetium 71 180 178 177 179 176 174 Hf 178.49(2) hafnium 72 181 180 Ta 180.947 88(2) tantalum 73 184 186 182 183 180 W 183.84(1) tungsten 74 187 185 Re 186.207(1) rhenium 75 192 190 189 188 187 186 184 Os 190.23(3) osmium 76 193 191 I r 192.217(3) iridium 77 195 194 196 198 192 190 Pt 195.084(9) platinum 78 197 Au 196.966 569(4) gold 79 202 200 199 201 198 204 196 Hg 200.59(2) mercury 80 205 203 Tl [204.382; 204.385] thallium 81 208 206 207 204 Pb lead 82 209 Bi 208.980 40(1) bismuth 83 Po polonium 84 At astatine 85 Rn radon 86 Fr francium 87 Ra radium 88 lanthanoids 57 - 71 actinoids 89 - 103 Ac actinium 89 232 Th 232.038 06(2) thorium 90 231 Pa 231.035 88(2) protactinium 91 238 235 234 U 238.028 91(3) uranium 92 Np neptunium 93 Pu plutonium 94 Am americium 95 Cm curium 96 Bk berkelium 97 Cf californium 98 Es einsteinium 99 Fm fermium 100 Md mendelevium 101 No nobelium 102 Lr lawrencium 103 Rf rutherfordium 104 Db dubnium 105 Sg seaborgium 106 Bh bohrium 107 Hs hassium 108 Mt meitnerium 109 Ds darmstadtium 110 Rg roentgenium 111 Cn copernicium 112 Uut ununtrium 113 Fl flerovium 114 Uup ununpentium 115 Lv livermorium 116 Uus ununseptium 117 Uuo ununoctium 118 June 20, 2012 www.ciaaw.org Element Background Color Key [Standard atomic weights are determined using all stable isotopes and selected radioactive isotopes (having relatively long half-lives and characteristic isotopic abundances) in natural terrestrial substances. Isotopes are considered stable (non-radioactive) if evidence for radioactive decay has not been detected experimentally.] 1 18 2 13 14 15 16 17 3 4 5 6 7 8 9 10 11 12 4 3 He 4.002 602(2) helium 2 114 112 111 110 106 108 Cd 112.411(8) cadmium 48 { uncertainty in last digit (112.411 ± 0.008) element name element symbol atomic number (number of protons) standard atomic weight isotope mass number (number of protons + neutrons) 113 116 , black indicates isotope is stable isotopic abundance (mole fraction , red indicates isotope is radioactive of isotope) International Union of Pure and Applied Chemistry (IUPAC) Project 2007-038-3-200, "Development of an isotopic periodic table for the educational community" Element has two or more isotopes that are used to determine its standard atomic weight. The isotopic abundances and atomic weights vary in natural terrestrial substances. These variations are well known, and the standard atomic weight is given as lower and upper bounds within square brackets, [ ]. Element has two or more isotopes that are used to determine its standard atomic weight. The isotopic abundances and atomic weights vary in natural terrestrial substances, but upper and lower bounds of the standard atomic weight have not been assigned by IUPAC or the variations may be too small to affect the standard atomic weight value. Thus, the standard atomic weight is given as a single value with an uncertainty that includes both measurement uncertainty and uncertainty due to isotopic abundance variations. Element has only one isotope that is used to determine its standard atomic weight. Thus, the standard atomic weight is invariant and is given as a single value with an IUPAC evaluated measurement uncertainty. Element has no standard atomic weight because all of its isotopes are radioactive and, in natural terrestrial substances, no isotope occurs with a characteristic isotopic abundance from which a standard atomic weight can be determined. . .

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript
IUPAC Periodic Table of the Isotopes
1
2H
[1.007 84; 1.008 11]
hydrogen
1
7
6Li
[6.938; 6.997]
lithium
39Be
9.012 182(3)
beryllium
4
11
10B
[10.806; 10.821]
boron
5
12
13C
[12.0096; 12.0116]
carbon
6
14
15N
[14.006 43; 14.007 28]
nitrogen
7
16
1817O
[15.999 03; 15.999 77]
oxygen
819F
18.998 4032(5)
fluorine
9
20
2221Ne
20.1797(6)
neon
10
23Na
22.989 769 28(2)
sodium
11
24
2625
Mg
24.3050(6)
magnesium
1227
Al26.981 5386(8)
aluminium(aluminum)
1328
2930Si
[28.084; 28.086]
silicon
1431P
30.973 762(2)
phosphorus
15
32
3433 36
S
[32.059; 32.076]
sulfur
16
35
37Cl
[35.446; 35.457]
chlorine
17
40
36 38Ar
39.948(1)
argon
18
39
4140K
39.0983(1)
potassium
19
40
4442 48
4346Ca
40.078(4)
calcium
2045Sc
44.955 912(6)
scandium
21
48
46 47 4950Ti
47.867(1)
titanium
22
51
50V
50.9415(1)
vanadium
23
52
53 5054Cr
51.9961(6)
chromium
2455Mn
54.938 045(5)
manganese
25
56
5457
58Fe
55.845(2)
iron
2659Co
58.933 195(5)
cobalt
27
58
6062
6164Ni
58.6934(4)
nickel
28
63
65Cu
63.546(3)
copper
29
6466
68 6770Zn
65.38(2)
zinc
30
69
71Ga
69.723(1)
gallium
31
7472
70 7673Ge
72.63(1)
germanium
3275As
74.921 60(2)
arsenic
33
80
78
7682 77
74Se
78.96(3)
selenium
34
79
81Br
79.904(1)
bromine
35
8486
8283
8078Kr
83.798(2)
krypton
36
85
87Rb
85.4678(3)
rubidium
37
88
86 8784Sr
87.62(1)
strontium
3889Y
88.905 85(2)
yttrium
39
90
94
92 9196Zr
91.224(2)
zirconium
4093Nb
92.906 38(2)
niobium
4198
9695
92
100
97 94Mo
95.96(2)
molybdenum
42Tctechnetium
43
102104
101
99100 9698Ru
101.07(2)
ruthenium
44103Rh
102.905 50(2)
rhodium
45106
108105
110104
102Pd
106.42(1)
palladium
46
107
109Ag
107.8682(2)
silver
47114
112111
110
113 116106 108
Cd
112.411(8)
cadmium
48
115
113In
114.818(3)
indium
49120
118116
119
117124
122112 114
115Sn
118.710(7)
tin
50
121
123Sb
121.760(1)
antimony
51130
128
126
125124
122 123120Te
127.60(3)
tellurium
52127I
126.904 47(3)
iodine
53132
129
131
134136130 128
124126Xe
131.293(6)
xenon
54
133
Cs132.905 4519(2)
caesium(cesium)
55138
137136
135
134130
132Ba
137.327(7)
barium
56
139
138La
138.905 47(7)
lanthanum
57
140
142
138136Ce
140.116(1)
cerium
58141Pr
140.907 65(2)
praseodymium
59142
144146
143
145148 150Nd
144.242(3)
neodymium
60Pmpromethium
61152
154147
149
148150
144Sm
150.36(2)
samarium
62
153
151Eu
151.964(1)
europium
63158
160156
157
155154 152Gd
157.25(3)
gadolinium
64159Tb
158.925 35(2)
terbium
65164
162
163
161160
158 156Dy
162.500(1)
dysprosium
66165Ho
164.930 32(2)
holmium
67166
168
167
170
164162Er
167.259(3)
erbium
68169Tm
168.934 21(2)
thulium
69174
172173
171
176170
168Yb
173.054(5)
ytterbium
70
175
176Lu
174.9668(1)
lutetium
71
180
178
177
179176
174Hf
178.49(2)
hafnium
72
181
180Ta
180.947 88(2)
tantalum
73184
186
182
183
180W
183.84(1)
tungsten
74
187
185Re
186.207(1)
rhenium
75 192
190
189
188187
186 184Os
190.23(3)
osmium
76
193
191Ir
192.217(3)
iridium
77 195
194
196198
192 190Pt
195.084(9)
platinum
78
197Au
196.966 569(4)
gold
79202
200199
201
198 204196Hg
200.59(2)
mercury
80
205
203Tl
[204.382; 204.385]
thallium
81
208
206
207 204Pblead
82209Bi
208.980 40(1)
bismuth
83Popolonium
84Atastatine
85Rnradon
86
Frfrancium
87Raradium
88
lanthanoids
57 - 71
actinoids
89 - 103
Acactinium
89232Th
232.038 06(2)
thorium
90231Pa
231.035 88(2)
protactinium
91
238
235234U
238.028 91(3)
uranium
92Npneptunium
93Puplutonium
94Amamericium
95Cmcurium
96Bkberkelium
97Cfcalifornium
98Eseinsteinium
99Fmfermium
100Mdmendelevium
101Nonobelium
102Lrlawrencium
103
Rfrutherfordium
104Dbdubnium
105Sgseaborgium
106Bhbohrium
107Hshassium
108Mtmeitnerium
109Dsdarmstadtium
110Rgroentgenium
111Cncopernicium
112Uutununtrium
113Flflerovium
114Uupununpentium
115Lvlivermorium
116Uusununseptium
117Uuoununoctium
118
June 20, 2012 www.ciaaw.org
Element Background Color Key[Standard atomic weights are determined using all stable isotopes and selected radioactive isotopes (having relatively long half-lives and characteristic isotopic abundances) in natural terrestrial substances. Isotopes are considered stable (non-radioactive) if evidence for radioactive decay has not been detected experimentally.]
1 18
2 13 14 15 16 17
3 4 5 6 7 8 9 10 11 12
4
3He
4.002 602(2)
helium
2
114
112111
110
113 116106 108
Cd
112.411(8)
cadmium
48
{
uncertainty in last digit (112.411 ± 0.008)
element name
element symbol
atomic number(number of protons)
standard atomic weight
isotope mass number(number of protons +neutrons)
113 116 , black indicates isotope is stable
isotopic abundance(mole fraction
, red indicates isotope is radioactive
of isotope)
International Union of Pure and Applied Chemistry (IUPAC) Project 2007-038-3-200, "Development of an isotopic periodic table for the educational community"
Element has two or more isotopes that are used to determine its standard atomic weight. The isotopic abundances and atomic weights vary in natural terrestrial substances. These variations are well known, and the standard atomic weight is given as lower and upper bounds within square brackets, [ ].
Element has two or more isotopes that are used to determine its standard atomic weight. The isotopic abundances and atomic weights vary in natural terrestrial substances, but upper and lower bounds of the standard atomic weight have not been assigned by IUPAC or the variations may be too small to affect the standard atomic weight value. Thus, the standard atomic weight is given as a single value with an uncertainty that includes both measurement uncertainty and uncertainty due to isotopic abundance variations.
Element has only one isotope that is used to determine its standard atomic weight. Thus, the standard atomic weight is invariant and is given as a single value with an IUPAC evaluated measurement uncertainty.
Element has no standard atomic weight because all of its isotopes are radioactive and, in natural terrestrial substances, no isotope occurs with a characteristic isotopic abundance from which a standard atomic weight can be determined.
.
.
Related Documents



![IUPAC Periodic Table of the Isotopes - · PDF fileIUPAC Periodic Table of the Isotopes 1 H 2 [1.007 84; 1.008 11] hydrogen 1 7 Li 6 [6.938; 6.997] lithium 3 Be 9 9.012 182(3) beryllium](https://static.cupdf.com/doc/110x72/5a7ac52f7f8b9a563b8b90e2/iupac-periodic-table-of-the-isotopes-periodic-table-of-the-isotopes-1-h-2-1007.jpg)