Class Functional gp Suffix Example Formula Alkane C - C - ane ethane C n H 2n+2 H H ׀ ׀H - C – C – H ׀ ׀H H H ׀H - C – H ׀H H H H ׀ ׀ ׀H - C – C – C – H ׀ ׀ ׀H H H H H H H ׀ ׀ ׀ ׀H - C – C – C – C – H ׀ ׀ ׀ ׀H H H H Number carbon Word IUPAC name Structure formula Molecular formula 1 Meth Methane CH 4 CH 4 2 Eth Ethane CH 3 CH 3 C 2 H 6 3 Prop Propane CH 3 CH 2 CH 3 C 3 H 8 4 But Butane CH 3 (CH 2 ) 2 CH 3 C 4 H 10 5 Pent Pentane CH 3 (CH 2 ) 3 CH 3 C 5 H 12 6 Hex Hexane CH 3 (CH 2 ) 4 CH 3 C 6 H 14 7 Hept Heptane CH 3 (CH 2 ) 5 CH 3 C 7 H 16 8 Oct Octane CH 3 (CH 2 ) 6 CH 3 C 8 H 18 9 Non Nonane CH 3 (CH 2 ) 7 CH 3 C 9 H 20 10 Dec Decane CH 3 (CH 2 ) 8 CH 3 C 10 H 22 methane ethane propane butane Saturated hydrocarbon (C – C single bond) Chemical rxn Alkane Reactivity for Alkanes Combustion rxn Complete combustion – produce CO 2 + H 2 O • C 2 H 6 + 7/2O 2 → 2CO 2 + 3H 2 O Incomplete combustion – produce C, CO, CO 2 , H 2 O • 2C 3 H 8 + 7O 2 → 2C + 2CO + 8H 2 O + 2CO 2 Free Radical Substitution rxn Free Radical Substitution Mechanism - Homolytic fission- bond break by radical form. - Covalent bond split, each atom obtain one electron (unpair e) - UV needed - Radical react with molecule - Radical + radical → molecule CH 4 + CI 2 → CH 3 CI + HCI • Low reactivity - Strong stable bond bet C - C, C - H • Low reactivity - Low polarity of C - H bond • Saturated hydrocarbon – Non polar bond Initiation Propagation Radical (dot) Termination homolytic fission Radical recycle again 1 2

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

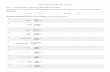
Class Functional gp Suffix Example Formula
Alkane C - C - ane ethane CnH2n+2
H H
׀ ׀
H - C – C – H
׀ ׀
H H
H ׀
H - C – H
׀ H
H H H
׀ ׀ ׀
H - C – C – C – H
׀ ׀ ׀
H H H
H H H H
׀ ׀ ׀ ׀
H - C – C – C – C – H
׀ ׀ ׀ ׀
H H H H
Number carbon
Word IUPAC name
Structure formula Molecular formula
1 Meth Methane CH4 CH4
2 Eth Ethane CH3CH3 C2H6
3 Prop Propane CH3CH2CH3 C3H8
4 But Butane CH3(CH2)2CH3 C4H10
5 Pent Pentane CH3(CH2)3CH3 C5H12
6 Hex Hexane CH3(CH2)4CH3 C6H14
7 Hept Heptane CH3(CH2)5CH3 C7H16
8 Oct Octane CH3(CH2)6CH3 C8H18
9 Non Nonane CH3(CH2)7CH3 C9H20
10 Dec Decane CH3(CH2)8CH3 C10H22
methane ethane propane butane
Saturated hydrocarbon (C – C single bond)
Chemical rxn Alkane Reactivity for Alkanes
Combustion rxn
Complete combustion – produce CO2 + H2O • C2H6 + 7/2O2 → 2CO2 + 3H2O Incomplete combustion – produce C, CO, CO2, H2O • 2C3H8 + 7O2 → 2C + 2CO + 8H2O + 2CO2
Free Radical Substitution rxn
Free Radical Substitution Mechanism
- Homolytic fission- bond break by radical form. - Covalent bond split, each atom obtain one electron (unpair e) - UV needed
- Radical react with molecule
- Radical + radical → molecule
CH4 + CI2 → CH3CI + HCI
• Low reactivity - Strong stable bond bet C - C, C - H • Low reactivity - Low polarity of C - H bond • Saturated hydrocarbon – Non polar bond
Initiation
Propagation
Radical (dot)
Termination
homolytic fission
Radical recycle again
1
2

H H
׀ ׀
C = C
׀ ׀
H H
H H H
׀ ׀ ׀
C = C – C - H
׀ ׀
H H
H H H H
׀ ׀ ׀ ׀
C = C – C – C - H
׀ ׀ ׀
H H H
Unsaturated hydrocarbon (C = C double bond)
H H H H H
׀ ׀ ׀ ׀ ׀
C = C – C – C – C - H
׀ ׀ ׀ ׀
H H H H
ethene propene butene pentene
Reactivity for Alkene
- High reactivity - Unstable bond bet C = C - High reactivity – Weak pi bond overlap bet p orbital - Unsaturated hydrocarbon – ᴨ bond overlap
Combustion rxn
Chemical rxn Alkane
Complete combustion – produce CO2 + H2O C2H4 + 3O2 → 2CO2 + 2H2O Incomplete combustion – produce C, CO, CO2, H2O 2C2H4 + 7/2O2 → 2C + CO + 4H2O + CO2
CH2 = CH2 + Br2 → CH2BrCH2Br CH2 = CH2 + HCI → CH3CH2CI CH2 = CH2 + H2O → CH3CH2OH
Addition rxn
H H
׀ ׀
C = C
׀ ׀
H H
H H
׀ ׀
H - C – C – H
׀ ׀
CI CI
Class Functional Suffix Example Formula
Alkene Alkenyl - ene ethene CnH2n
H H
׀ ׀
H - C – C – H
׀ ׀
Br Br
H H
׀ ׀
H - C – C – H
׀ ׀
H CI
H H
׀ ׀
H - C – C – H
׀ ׀
H OH
1
2
Polymerization (Addition rxn) 3
Polymers are long chains molecules (plastics) • Join repeat units call monomers • Addition and condensation polymerization
• Monomers double bond (unsaturated) • Repeat units join together by covalent bond without loss of any molecule
ethene polyethene
add monomer
polymer
propene polypropylene
add monomer
H CH3 H CH3
monomer
monomer
chloroethene polychloroethene (PVC)
tetrafluoroethene polytetrafluoroethene (PTFE)
H CI H CI
F F
F F
F F
F F
polymerization
polymer
Alkene decolourize brown liq Br2

OH
׀ CH3-C – CH3 + [O] No product ׀ CH3
OH O
‖ ׀ CH3- C–CH3 + [O] CH3- C – CH3 + H2O
H
׀
CH3 – C – OH
׀
H
Class Functional Suffix Example Formula
Alcohol Hydroxyl - ol methanol CnH2n+1OH
Number carbon
IUPAC name Structure formula Molecular
formula
1 Methanol CH3OH CH3OH
2 Ethanol CH3CH2OH C2H5OH
3 Propanol CH3CH2CH2OH C3H7OH
4 Butanol CH3(CH2)2CH2OH C4H9OH
methanol ethanol propanol butanol
H
׀
H - C – OH
׀
H
H H
׀ ׀
H - C – C – OH
׀ ׀
H H
H H H
׀ ׀ ׀
H - C – C – C – OH
׀ ׀ ׀
H H H
H H H H
׀ ׀ ׀ ׀
H - C – C – C – C – OH
׀ ׀ ׀ ׀
H H H H
Hydrocarbon skeleton Functional gp
Chemical rxn Alcohol Reactivity for Alcohol
Primary 1 0
1 alkyl /R gp bond to C attach to OH
CH3 H
׀ ׀
CH3 – C – C – OH
׀ ׀
CH3 H
Combustion rxn
Complete combustion–produce CO2 + H2O C2H6OH + 3O2 → 2CO2 + 3H2O Incomplete combustion-produce C, CO, CO2, + H2O 2C2H5OH + 4O2 → C + 2CO + 6H2O + CO2
Oxidation rxn
Secondary 2 0
2 alkyl/R gp bond to C attach to OH
H
׀
CH3 – C – OH
׀
CH3
H H H
׀ ׀ ׀
H - C – C – C – H
׀ ׀ ׀
H OH H
Tertiary 3 0
3 alkyl/R gp bond to C attach to OH
CH3
׀
CH3 – C – OH
׀
CH3
R
׀
R – C – OH
׀
R
H ׀ CH3-CH2-OH + [O] CH3- C = O + H2O MnO4
- /H
+
K2Cr2O7/H+
Primary 10 – Oxidised to Aldehyde and Carboxylic acid
H OH ׀ ׀ CH3- C= O + [O] CH3-C= O
Secondary 20 - Oxidised to Ketone
Tertiary 30 - Cannot be Oxidised
MnO4- /H
+
K2Cr2O7/H+
MnO4- /H
+
K2Cr2O7/H+
MnO4- /H
+
K2Cr2O7/H+
1
1
Esterification rxn 3
O H
׀ ‖
H - C – O – C – H + H2O
׀
H
H
׀
H- O – C – H
׀
H
O
‖
H - C – O-H +

Chemical rxn Alcohol
Oxidation rxn – oxidized carbon attach to OH
Primary 10 – Oxidised to Aldehyde and Carboxylic acid
Secondary 20 - Oxidised to Ketone Tertiary 30 - Cannot be Oxidised
OH
׀ CH3-C – CH3 + [O] No product ׀ CH3
MnO4- /H
+
K2Cr2O7/H+
MnO4- /H
+
K2Cr2O7/H+
MnO4- /H
+
K2Cr2O7/H+
Alcohol to Aldehyde (Distillation) 1. Acidified dichromate(VI)/permanganate(VII)
2.Warm it , collect distillate (Distillation)
Aldehyde Carboxylic acid
-1 + 1
ON carbon increase Alcohol
H OH ׀ ׀
CH3- C= O + [O] CH3- C =O
H H ׀ ׀
CH3- C -O-H + [O] CH3- C = O
׀ H
+ 1 + 3
ON carbon increase Aldehyde
Primary 10 – Oxidised to Aldehyde and Carboxylic acid
Alcohol to Carboxylic acid (Reflux) 1. Acidified dichromate(VI)/permanganate(VII)
2.Warm it , collect distillate (Distillation)
Alcohol oxidize to Aldehyde • MnO4
- reduce from purple (Mn7+) to pink (Mn2+) • Cr2O7
2- reduce from orange (Cr6+) to green (Cr3+)
0 + 2
ON carbon increase Alcohol Ketone
Alcohol to Ketone (Reflux) 1. Acidified dichromate(VI)/permanganate(VII)
2.Warm it , collect distillate (Distillation) Click here oxidation alcohol
RCH2OH + [O] → RCHO + H2O RCH2OH + 2[O] → RCOOH + H2O RCH(OH)R + [O] → RCOR + H2O
Oxidation eqn (addition of O)
Aldehyde Alcohol
Alcohol
Alcohol
Carboxylic acid
Ketone
Alcohol oxidize to Carboxylic acid • MnO4
- reduce from purple (Mn7+) to pink (Mn2+) • Cr2O7
2- reduce from orange (Cr6+) to green (Cr3+)
distillation
reflux
Aldehyde turn to carboxylic acid
Aldehyde Alcohol
reflux
Alcohol turn to ketone
OH O
‖ ׀
CH3- C – CH3 + [O] CH3- C – CH3 + H2O

Class Functional Suffix Formula
Ester Ester - oate R –COO-R
Number carbon
IUPAC name Structure formula Molecular formula
1 Methyl methanoate HCOOCH3 R–COO-R
2 Methyl ethanoate CH3COOCH3 R–COO-R
3 Methyl propanoate CH3CH2COOCH3 R–COO-R
4 Methyl butanoate CH3CH2CH2COOCH3 R–COO-R
methyl methanoate methyl ethanoate methyl propanoate
O H
׀ ‖
H - C – O – C - H
׀
H
H O H
׀ ‖ ׀
H - C - C – O - C - H
׀ ׀
H H
H H O H
׀ ‖ ׀ ׀
H - C – C – C – O - C - H
׀ ׀ ׀
H H H
Hydrocarbon skeleton Functional gp
Esterification
O
‖
H - C – O-H
H
׀
H- O – C – H
׀
H
O H
׀ ‖
H - C – O – C – H + H2O
׀
H
Ester
Condensation rxn
↔ +
Methanoic acid Methanol Methyl methanoate
Esterification (reversible rxn) After reflux – reach equilibrium
Acid and alcohol (reflux) Conc H2SO4 (catalyst) used
Water produced
condensation
reflux
Ester purified and distill Click here ester preparation
H O H
׀ ‖ ׀
H - C - C – O - C – H + H2O
׀ ׀
H H
H
׀
H- O – C – H
׀
H
H O
‖ ׀
H - C - C – OH
׀
H
CH3COOH + CH3OH → CH3COOCH3 + H2O
H O H H
׀ ׀ ‖ ׀
H – C – C– O - C–C-H
׀ ׀ ׀
H H H
+
Ethanoic acid Methanol Methyl ethanoate
↔
H H
׀ ׀
H- O- C– C – H
׀ ׀
H H
H O
‖ ׀
H – C – C - OH
׀
H
condensation
CH3COOH + CH3CH2OH → CH3COOCH2CH3 + H2O
+
condensation
↔
Ethanoic acid Ethanol Ethyl ethanoate
+ H2O

H
׀
CH3 – C – CI
׀
H
H
׀
H - C – CI
׀
H
H H
׀ ׀
H - C – C – CI
׀ ׀
H H
H H H
׀ ׀ ׀
H - C – C – C – CI
׀ ׀ ׀
H H H
Hydrocarbon skeleton Functional gp
Primary 1 0
1 alkyl /R gp bond to C attach to CI
Secondary 2 0
2 alkyl/R gp bond to C attach to CI
H
׀
CH3 – C – CI
׀
CH3
H H H
׀ ׀ ׀
H - C – C – C – H
׀ ׀ ׀
H CI H
Tertiary 3 0
3 alkyl/R gp bond to C attach to CI
CH3
׀
CH3 – C – CI
׀
CH3
R
׀
R – C – CI
׀
R
Reactivity for Halogenoalkane
Class Functional Prefix Example
Halogenoalkane F, CI, Br, I - chloro chloroethane
Number carbon
IUPAC name Structure formula Molecular formula
1 chloromethane CH3CI CH3CI
2 chloroethane CH3CH2CI C2H5CI
3 chloropropane CH3CH2CH2CI C3H7CI
4 chlorobutane CH3(CH2)2CH2CI C4H9CI
chloromethane chloroethane chloropropane
Reactivity for halogenoalkane • Carbon bond to halogen – F, CI, Br, I • High electronegativity on halogen gp • High reactivity – due to polarity of C+- Br -
Nucleophile – Lone pair electron – Donate electron pair (Lewis base)
Chemical rxn Halogenoalkane
C - Br ᵟ+ ᵟ-
electron
Electron deficient
carbon
O–H ..
..
ᵟ- ᵟ+
C ᵟ+
Substitution rxn
CH3CH2CI + OH- → CH3CH2OH + CI-
H H
׀ ׀
H - C – C – CI
׀ ׀
H H
+ OH- ᵟ+ ᵟ-
H H
׀ ׀
H - C – C – OH + CI-
׀ ׀
H H
H Br H
׀ ׀ ׀
H - C – C – C – H
׀ ׀ ׀
H H H
CH3CHBrCH3 + OH- → CH3CHOHCH3 + Br-
+ OH-
H OH H
׀ ׀ ׀
H - C – C – C – H + Br-
׀ ׀ ׀
H H H
ᵟ+
ᵟ-
CH3 H
׀ ׀
CH3 – C – C – CI
׀ ׀
CH3 H

Electrophile - Electron deficient - Accept lone pair - Positive charge - Lewis Acid
C - Br
Reactivity for halogenoalkane • Carbon bond to halogen – F, CI, Br, I • High electronegativity on halogen gp • High reactivity – due to polarity of C+- CI -
C - Br ᵟ+ ᵟ-
electron
Electron deficient carbon
OH ..
ᵟ- ᵟ+
Nucleophilic Substitution rxn
CH3CH2CI + OH- → CH3CH2OH + CI-
H H
׀ ׀
H - C – C – CI
׀ ׀
H H
+ OH- ᵟ+ ᵟ-
H H
׀ ׀
H - C – C – OH + CI-
׀ ׀
H H
H Br H
׀ ׀ ׀
H - C – C – C – H
׀ ׀ ׀
H H H
CH3CHBrCH3 + OH- → CH3CHOHCH3 + Br-
+ OH-
H OH H
׀ ׀ ׀
H - C – C – C – H + Br-
׀ ׀ ׀
H H H
ᵟ+ ᵟ-
Nucleophile and Substitution Electrophile and Addition
vs Reactivity of Alkene - High reactivity - Unstable bond bet C = C - High reactivity – Weak pi bond overlap bet p orbital - Unsaturated hydrocarbon – ᴨ bond overlap
C = C Electron rich π electron
ᵟ- ᵟ-
H ᵟ+
C = C ᵟ- ᵟ-
E ᵟ+
E+ Electron deficient Nu
ᵟ-
ᵟ-
Nucleophile – Lone pair electron – Donate electron pair - Lewis Base
H H ׀ ׀
C = C
׀ ׀ H H
CH2=CH2 + Br2 → CH2BrCH2Br
+ Br – Br ᵟ- ᵟ+
H H ׀ ׀
H - C – C – H
׀ ׀ Br Br
vs
CH2=CH2 + HCI → CH3CH2CI
H H ׀ ׀
C = C
׀ ׀ H H
ᵟ- + H – CI
ᵟ+
H H ׀ ׀
H - C – C – H
׀ ׀ H CI
Electrophilic Addition rxn

Electrophile - Electron deficient - Accept lone pair - Positive charge - Lewis Acid
ᵟ-
Electron rich region
Electrophilic Substitution rxn
C6H6 + Br2 C6H5Br + HBr
+ Br-Br ᵟ+
+ NO2+
ᵟ+
Electrophile and Substitution Electrophile and Addition
vs
C = C Electron rich π electron
ᵟ- ᵟ-
ᵟ+
C = C ᵟ- ᵟ-
E ᵟ+
E+ Electron deficient
E ᵟ+
H H ׀ ׀
C = C
׀ ׀ H H
CH2=CH2 + Br2 → CH2BrCH2Br
+ Br – Br ᵟ- ᵟ+
H H ׀ ׀
H - C – C – H
׀ ׀ Br Br
vs
CH2=CH2 + HCI → CH3CH2CI
H H ׀ ׀
C = C
׀ ׀ H H
ᵟ- + H – CI ᵟ+
H H ׀ ׀
H - C – C – H
׀ ׀ H CI
Electrophilic Addition rxn
E
Electrophile - Electron deficient - Accept lone pair - Positive charge - Lewis Acid
ᵟ+ + H E
+ H
Electron rich region
H Br
+ HBr
C6H6 + HNO3 C6H5NO2 + HCI
AICI3 dry ether
warm/Conc H2SO4
H NO2
Reactivity of Alkene - High reactivity - Unstable bond bet C = C - High reactivity – Weak pi bond overlap bet p orbital - Unsaturated hydrocarbon – ᴨ bond overlap
Reactivity of Benzene (Unreactive) - Delocalization of electron in ring - Stability due to delocalized π electron - Substitution instead of Addition
ethene decolourize brown Br2(I)
benzene –stable (unreactive) toward addition rxn
H
C6H6 – no rxn with brown Br2(I)

Electrophile - Electron deficient - Accept lone pair - Positive charge - Lewis Acid
C - Br OH .. ᵟ- ᵟ+
Nucleophile Electrophile
ᵟ+
C = C ᵟ-
Nucleophile – Lone pair electron – Donate electron pair - Lewis Base
Organic Rxn
Addition rxn Substitution rxn
Nucleophilic Substitution
Free Radical Substitution
Electrophilic Substitution Electrophilic Addition rxn
Free radicle
CI CI
CI CI . .
:
Radical (unpair electron)
uv radiation
H H ׀ ׀
C = C
׀ ׀ H H
+ Br – Br
H H ׀ ׀
H - C – C – H
׀ ׀ Br Br
ᵟ+
ᵟ-
H H
׀ ׀
H - C – C – CI
׀ ׀
H H
+ OH-
H H
׀ ׀
H - C – C – OH + CI-
׀ ׀
H H
ᵟ- ᵟ+ H
E + + H
E ᵟ+
H H
׀ ׀
C = C
׀ ׀
H H
H H
׀ ׀
H - C – C – H
׀ ׀
CI CI
H H
׀ ׀
H - C – C – H
׀ ׀
H CI
H H
׀ ׀
H - C – C – H
׀ ׀
H OH
Add HCI
CI2 / UV
H H
׀ ׀
H - C – C – CI
׀ ׀
H H
H H
׀ ׀
H - C – C – OH + CI-
׀ ׀
H H
H H
׀ ׀
H - C – C – NH2 + CI-
׀ ׀
H H
H H
׀ ׀
H - C – C – CN + CI-
׀ ׀
H H
NH3
OH-
CN-
H
׀
H - C – H
׀
H
H
׀
H - C – CI + H
׀
H
CI2 → 2 CI•
CH3• + CI2 → CH3CI + CI•
CI• + CH4 → HCI + CH3•
H

Electrophile - Electron deficient - Accept lone pair - Positive charge - Lewis Acid
C - Br OH .. ᵟ- ᵟ+
Nucleophile Electrophile
H ᵟ+
C = C ᵟ-
Nucleophile – Lone pair electron – Donate electron pair - Lewis Base
Free radicle
CI CI
CI CI . .
:
Radical (unpair electron)
uv radiation
H H
׀ ׀
C = C
׀ ׀
H H
H H
׀ ׀
H - C – C – H
׀ ׀
CI CI
H H
׀ ׀
H - C – C – H
׀ ׀
H CI
H H
׀ ׀
H - C – C – H
׀ ׀
H OH
Add HCI
CI2 / UV
H H
׀ ׀
H - C – C – CI
׀ ׀
H H
H H
׀ ׀
H - C – C – OH + CI-
׀ ׀
H H
H H
׀ ׀
H - C – C – NH2 + CI-
׀ ׀
H H
H H
׀ ׀
H - C – C – CN + CI-
׀ ׀
H H
NH3
OH-
CN-
H
׀
H - C – H
׀
H
H
׀
H - C – CI + H
׀
H
CI2 → 2 CI•
CH3• + CI2 → CH3CI + CI•
CI• + CH4 → HCI + CH3•
Alkene – Addition rxn Halogenoalkane – Substitution rxn Alkane - Radical substitution
H OH
׀ ׀
H - C – C – H
׀ ׀
H H
H O
‖ ׀
H - C – C – H
׀
H
H O
‖ ׀
H - C – C – OH
׀
H
H O H
׀ ‖ ׀
H - C – C – C – H
׀ ׀
H H
H OH H
׀ ׀ ׀
H - C – C – C – H
׀ ׀ ׀
H H H
H OH H
׀ ׀ ׀
H - C – C – C – H
׀ ׀ ׀
H CH3 H
Alcohol – Oxidation rxn
10 alcohol 20 alcohol 30 alcohol
carboxylic acid aldehyde ketone
no reaction

׀ ׀ C- C –OH ׀ ׀
O ‖ C – C – C
O ‖ C – C – H
O ‖ C – C – OH
O ‖ C –C – C– O – C – C
O H ׀ ‖ C – C – N – C – C
No reaction
1o alcohol
[O]/Cr2O7/H+
Aldehyde
Ketone Carboxylic Acid
Free radical substitution
CI2/ UV
Halogenoalkane
Alkane
3o alcohol
[O]/ Cr2O7/H+
Substitution
warm / OH-
Alcohol
Substitution / CN-
Amine
Nitrile
Alcohol
Condensation
Amide
Amine
Carboxylic Acid
Alkene
Elimination
100C /Conc alcoholic OH-
Alkane Halogenoalkane Dihalogenoalkane
Condensation
Ester
Addition
Polymerisation
X
׀ ׀ C – C – CI ׀ ׀
׀ ׀ C = C ׀ ׀
׀ ׀ ׀ ׀ C – C – C – C ׀ ׀ ׀ ׀
׀ ׀ C – C ׀ ׀ H CI
׀ ׀ C – C ׀ ׀ CI CI
׀ ׀ C – C ׀ ׀ Br Br
׀ ׀ C – C ׀ ׀
׀ ׀ C – C – OH ׀ ׀
׀ ׀ C – C – CN ׀ ׀
׀ ׀ C – C – NH2 ׀ ׀ ׀ ׀ ׀
C – C – C –NH2 ׀ ׀ ׀
׀ ׀ C – C – COOH ׀ ׀
Start here
PolyAlkene
׀ ׀ C – C ׀ ׀ H H

H
׀
CH3 – C – Br
׀
H
CH3 H
׀ ׀
CH3 – C – C – Br
׀ ׀
CH3 H
Reactivity for halogenoalkane • Carbon bond to halogen – F, CI, Br, I • High electronegativity on halogen • High reactivity – polarity of C+- Br -
Nucleophile – Lone pair electron – Donate electron pair - (Lewis base)
Chemical rxn Halogenoalkane
C - Br ᵟ+ ᵟ-
electron
Electron deficient
carbon
O–H ..
.. ᵟ-
C ᵟ+
H H
׀ ׀
H - C – C – Br
׀ ׀
H H
+ OH- ᵟ+ ᵟ-
H H
׀ ׀
H - C – C – OH + Br-
׀ ׀
H H
Nucleophilic Substitution
Primary 10 - SN2
Primary 10 - SN2
- Experimentally rate expression = k [CH3CH2Br][OH-] - Rate dependent on conc- CH3CH2Br and OH-
- Molecularity = 2 - No bulky alkyl gp, less steric effect - Allow nucleophile to attack electron deficient carbon from opposite site (Inversion of configuration)
CH3CH2Br + OH- → CH3CH2OH + Br-
SN2 Substitution
Bimolecular collision bet 2 molecule
Nucleophilic
Bimolecular Nucleophilic Substitution
OH- + CH3CH2Br [ HO---CH2(CH3)---Br]- CH3CH2OH + Br-
HO-
Bond breaking and making in transition state
+ Br-
One step mechanism – Bond break and making in transition state
nucleophile attack
leaving gp
Click here to view SN2
slow step (RDS)
fast step
slow step (RDS) fast step
✓ 1ₒ SN2

Hydrolysis bromoethane (1o)
H
׀
OH- + CH3 – C – Br
׀
H
Bond Breaking and Making at transition state Br leaving gp substituted with OH-
H H
׀ ׀ CH3 - C – Br + OH- CH3 – C – OH + Br - ׀ ׀ H H
Nucleophile collide with bromoethane
CH3CH2Br + OH- → CH3CH2OH + Br- Single step
Nucleophilic Substitution
Click here view SN2
SN2 Substitution
Nucleophilic
Bimolecular Nucleophilic Substitution
Bimolecular collision bet 2 molecule
- Experimentally rate expression = k [CH3CH2Br][OH-] - Rate dependent on conc = CH3CH2Br and OH-
- Molecularity = 2 - No bulky alkyl gp, less steric effect - Allow nucleophile to attack electron deficient carbon from the opposite site (Inversion of configuration)
Formation of ethanol
1 step mechanism (concerted)
SN2 1ₒ

Nucleophile – Lone pair electron – Donate electron pair - (Lewis base)
CH3
׀
CH3 – C – Br
׀
CH3
CH3
׀
CH3 – C – Br
׀
CH3
R
׀
R – C – Br
׀
R
Reactivity for halogenoalkane • Carbon bond to halogen gp – F, CI, Br, I • High electronegativity on halogen gp • High reactivity – polarity of C+- Br -
Chemical rxn Halogenoalkane
C - Br ᵟ+ ᵟ-
electron
Electron deficient
carbon
O–H ..
.. ᵟ-
C ᵟ+
+ OH- ᵟ+ ᵟ-
Nucleophilic Substitution
Tertiary 30 – SN1
Tertiary 30 – SN1
- Experimentally rate expression = k [(CH3)3CBr] - Rate dependent on conc - (CH3)3CBr
- Molecularity = 1 - 3 Bulky alkyl gp, Steric hindrance effect - 30 carbocation more stable due to inductive effect • 3 alkyl gp stabilize carbocation by inductive effect push electron to carbocation (reducing positive charge) making it more stable
SN1 Substitution
Unimolecular (1 molecule)
Nucleophilic
Unimolecular Nucleophilic Substitution
+ :OH-
carbocation (Intermediate)
+ Br-
1st step mechanism – carbocation formation
nucleophile attack
Click here to view SN1
(CH3)3CBr + OH- → (CH3)3COH + Br-
CH3
׀
CH3 – C – OH + Br -
׀
CH3
slow step (RDS)
heterolytic fission Br leaving gp
fast step
2nd step mechanism – OH attack carbocation
(CH3)3CBr → (CH3)3C+ + Br- 1st step (slow)
(CH3)3C+ + OH- → (CH3)3COH 2nd step (fast)
✓ 3ₒ SN1

Formation of 2 methylpropan-2-ol
Hydrolysis 2-bromo- 2- methylpropane (3o)
CH3 │
CH3 - C – Br
│
CH3
Carbocation formation (Intermediate) Nucleophile OH- attack carbocation
Heterolytic fission - Carbocation and Br- form
(CH3)3CBr → (CH3)3C+ + Br- 1st step (slow)
(CH3)3C+ + OH- → (CH3)3COH 2nd step (fast)
CH3 CH3
׀ ׀ CH3 - C – Br + OH- CH3 –C – OH + Br - ׀ ׀ CH3 CH3
Nucleophilic Substitution
Click here to view
- 3 Bulky alkyl gp - Steric hindrance effect - 30 carbocation more stable due to inductive effect • 3 alkyl gp stabilize carbocation by inductive effect push electron to carbocation (reducing positive charge) making it more stable
SN1 Unimolecular (1 molecule)
Substitution
Nucleophilic
Unimolecular Nucleophilic Substitution
2 step mechanism
3ₒ SN1

H Br H
׀ ׀ ׀
H - C – C – C – H
׀׀ ׀ ׀ ׀
H H H
+ :OH- ᵟ+
Nucleophilic Substitution
Secondary 20 - SN1 and SN2
- Experimentally rate expression = k [CH3CHBrCH3][OH-] - Rate dependent conc = CH3CHBrCH3 and OH-
- Molecularity = 2 - No bulky alkyl gp, less steric effect - Allow nucleophile to attack electron deficient carbon from opposite site (Inversion of configuration)
SN2 Substitution
Bimolecular collision bet 2 molecule
Nucleophilic
Bimolecular Nucleophilic Substitution
HO-
Bond breaking and making in transition state
+ Br-
One step mechanism – Bond break and making in transition state
nucleophile attack
leaving gp
slow step (RDS)
fast step
CH3CHBrCH3 + OH- → CH3CH(OH)CH3 + Br-
H OH H
׀ ׀ ׀
H - C – C – C – H + Br -
׀ ׀ ׀
H H H
CH3 CH3 CH3
SN1 Substitution
Nucleophilic
Unimolecular (1 molecule)
Unimolecular Nucleophilic Substitution
heterolytic fission Br leaving gp
slow step (RDS)
carbocation (Intermediate)
+ Br-
nucleophile attack
+ :OH-
CH3
1st step mechanism – carbocation formation
fast step
+
+
2nd step mechanism – OH attack carbocation
CH3
Click here SN1 vs SN2
1 step mechanism (concerted)
CH3CHBrCH3 → CH3CH+ CH3 + Br- 1st step (slow)
CH3CH+ CH3 + OH- → CH3CHOHCH3 2nd step (fast)
2 step mechanism CH3CHBrCH3 + OH- → CH3CH(OH)CH3 + Br-
Click here SN1 vs SN2 Khan academy
✓
2ₒ SN1 SN2

Electrophile - Electron deficient - Accept lone pair - Positive charge - Lewis Acid
C - Br OH .. ᵟ- ᵟ+
Nucleophile Electrophile
H ᵟ+
C = C ᵟ-
Nucleophile – Lone pair electron – Donate electron pair - Lewis Base
Free radicle
CI CI
CI CI . .
:
Radical (unpair electron)
uv radiation
H+ Br+ NO2+ :OH- :CN- H2O: :NH3
Homolytic fission Heterolytic fission
CI CI : uv radiation
CI CI . . fish hook arrow Single electron movement
A B :
A B :
A – B A + :B
Double headed arrow pair electron movement
Control by electronic factor (charges)
vs vs
vs
Nucleophilic Substitution
Primary 10 - SN2 Secondary 20 -SN1 and SN2 Tertiary 30 – SN1
SN1
SN2
Control by steric factor (alkyl gp)
SN2 SN1
Favour 10 30
Nature mechanism
1 step (transition state)
2 step (carbocation)
Rate lower higher
Solvent Polar aprotic Polar protic
Reaction profile
Click here SN1 vs SN2

Factor affecting Rate of Nucleophilic Substitution
• Bond polarity decrease ↓ • Bond strength decrease ↓
• Rate fastest (Halogen leave easily) Iodo > Bromo > Chloro > Fluoro
Nucleophilic Substitution
• SN 1 > SN 2 mechanism • 3o > 2o > 1o
• 3o – SN 1 - Carbocation - faster • 1o - SN 2 – Transition state - slower
Nature of solvent
Nature of Halogen
CH3
׀
CH3 – C – Br
׀
CH3
H
׀
CH3 – C – Br
׀
CH3
H
׀
CH3 – C – Br
׀
H
> > CH3CH2 – I > CH3CH2 – CI > CH3CH2 – F
fastest slowest
weak bond strong bond
C - Br OH Nucleophile
ᵟ-
H bond to O or N H2 bonding/donate H+
H2O, NH3 CH3OH, CH3CH2OH Able to solvate cation and anion
Polar protic Polar aprotic
Lack acidic H, no H2 Bonding Acetone/CH3COCH3, DMSO, CH3CN
Solvate cation–nucleophile free for SN2
H H
׀ ׀
H - C – C – OH
׀ ׀
H H
H
׀
H -– C – OH
׀
H
ᵟ+
Nature of Halogenoalkane
SN1
polar + H2 bonding
:O:
‖
CH3 – C – CH3
:O:
‖
CH3 – S – CH3
polar only
SN2
Rate of hydrolysis of halogenoalkane
C4H9CI + H2O → C4H9OH + H+ + CI-
C4H9Br + H2O → C4H9OH + H+ + Br-
C4H9I + H2O → C4H9OH + H+ + I-
Reaction Time ppt to appear Observation
1-chlorobutane slowest white ppt 1-bromobutane cream ppt
1-iodobutane fastest yellow ppt
Method:
- Prepare 3 test tube contain 2 ml of ethanol each
- Pipette 0.1ml of chloro, bromo and iodobutane to each test tube
- Leave 3 test tube in 60C bath.
- Add 1ml AgNO3, mix and record time ppt to form
Ag+ react CI- → AgCI (white ppt) Ag+ react Br- → AgBr (cream ppt) Ag+ react I- → AgI (yellow ppt)
fastest slowest
1-iodobutane 1-chlorobutane ✓
+ Ag+

Factor affecting Rate of Nucleophilic Substitution
Click here protic/aprotic solvent
Nucleophilic Substitution
Nature of solvent
H bond to O or N H2 bonding/donate H+
H2O, NH3 CH3OH, CH3CH2OH Able to solvate cation and anion
+ Br-
Polar protic Polar aprotic
Lack acidic H, no H2 Bonding Acetone/CH3COCH3, DMSO
Solvate cation–nucleophile free for SN2
NaOH → Na+ + OH-
SN1 SN2
H2O solvate carbocation and Br- form Stabilize it – exist in intermediate state
H H
׀ ׀
H - C – C – Br
׀ ׀
H H
+ OH-
H H
׀ ׀
H - C – C – OH + Br-
׀ ׀
H H
H H
׀ ׀
H - C – C – OH
׀ ׀
H H
CH3 │
CH3 - C – Br
│
CH3
carbocation solvated by H2O
anion solvated by H2O
H
׀
H -– C – OH
׀
H
Acetone solvate cation – nucleophile free for SN2 No H2 bond- unable to solvate anion/nucleophile
:O:
‖
CH3 – C – CH3
: O
:
‖
CH
3 –
C –
CH
3
Na+ solvated by CH3COCH3
nucleophile free to attack
C - Br OH Nucleophile
ᵟ+ ᵟ-
Click here protic/aprotic solvent
:O:
‖
CH3 – C – CH3
:O:
‖
CH3 – S – CH3
Click here expt protic/aprotic solvent
Related Documents