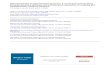
Fluctuations and Brownian Fluctuations and Brownian Motion Motion 2 2 fluorescent spheres in water (left) and DNA solution fluorescent spheres in water (left) and DNA solution (right) (right) (Movie Courtesy Professor Eric Weeks, Emory University: http://www.seas.harvard.edu/weitzlab/research/brownian.html) Brownian motion in water Brownian motion of DNA Brownian motion in water Brownian motion of DNA Copyright (c) Stuart Lindsay 2008

Fluctuations and Brownian Motion 2 fluorescent spheres in water (left) and DNA solution (right) (Movie Courtesy Professor Eric Weeks, Emory University:
Dec 21, 2015
Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

Fluctuations and Brownian MotionFluctuations and Brownian Motion
2 2 fluorescent spheres in water (left) and DNA solution (right) fluorescent spheres in water (left) and DNA solution (right)(Movie Courtesy Professor Eric Weeks, Emory University: http://www.seas.harvard.edu/weitzlab/research/brownian.html)
Brownian motion in water Brownian motion of DNABrownian motion in water Brownian motion of DNA
Copyright (c) Stuart Lindsay 2008

FluctuationsFluctuations
One set of collisions 5·105 set of collisions
Simulated distribution of speeds in a population of 23 atoms of an ideal gas: V=1.25·103 nm3 (50·50·50nm); T=300K.

Calculating FluctuationsCalculating Fluctuations
Zln
Z
EexpEE r
rr
rr
r EexpEZ
Z
Z
ZlnZln
1
2
2
2
2 111
Z
Z
Z.
Z
Z
Z
Z
Z
Zln
Taking the second derivative of dlnZ/d:

22
22
2
22
2
expexp11
EEZ
EE
Z
EEZ
Z
Z
Zr
rrr
rr
This is just the mean square thermal average of E:
222222 2 EEEEEEEEE
jj E
jj
E
jjj
jj eE
QeE
QPEE
11 222
Qln
EE
EQQ
1

VB CTkT
EkTE 222
22 ET
EkT
For an ideal gas:
NkCTNkE VB 2
3
2
3
NE
CkT
E
EV 122
The relative size of energy fluctuations scales as The relative size of energy fluctuations scales as N
1

T,V
ZlnkTN
In an open system:
222 NNN
NE
j,N
NE
j,Nj,N
j,N
eeNZ
kTeeN
ZPNN j,Nj,N222 1
2N
NkT
ZlnN
NkTNZ
Z
kT
V,T
NkTN
2

From thermodynamics:T,NV,T V
p
N
V
N
2
22 NV
kTN
T,Np
V
V
1 Isothermal compressibility
For an ideal gas:p
1
NN 2NN
N 12

• The result just obtained for energy holds for all quantities (extensive quantities) that, like energy, grow with N (T, E, P, V, S).
• In general the root-mean-square value of the fluctuations relative to the mean value of a quantity is given by
NX
X RMS 1
NN Relative fluctuationRelative fluctuation
10 31.6%
1000 3.2%
1026 10-13
Copyright (c) Stuart Lindsay 2008

Brownian motionBrownian motion
)t(Fvdt
dvm
Langevin equation:Langevin equation:
α (friction coefficient )
For a sphere of radius a in a medium of viscosity η: =6πηa
Stoke’s forceStoke’s force = friction exerted on the particle by the fluid. For small velocities, it is proportional to the velocity v.

0)( tF )tt(F)t(F)t(F
F(t) is a random force representing the constant molecular bombardment exerted by the surrounding fluid:
Average is zero! Finite only over duration of single “effective” collision
F(t) is independent of the velocity of the particle (v) and varies extremely rapidly compared to the variations in v.
There is no correlation between F(t) and F(t+Δt) even though Δt is expected to be very small.

)()( 2 txFxxxxxdt
dmxmx
Multiplying both sides of the Langevin eqn. by x and using
xv
Re-arranging and taking thermal averages:
)(2 txFxxxmxxdt
dm
Tkxm B2
1
2
1 2 = 0

so
xxmm
Tkxx
dt
d B
substituting
CBtexpAxx
yields
Tk
C Bm
B
To find A, note
2
2
1x
dt
dxx

with <x2>=0 at t=0:
Tk
A B
m
texp
Tkx
dt
d B
12
1 2
d
mdt
m
t
substitute
m
tmt
Tkx B
exp1
22
Integrate with <x2>=0 at t=0:

m
tmt
Tkx B
exp1
22
Long time solution:
m
t
a
tTktTkx BB
3
22
Mean square displacement increases with t!
Copyright (c) Stuart Lindsay 2008

• Now we see why the sphere in a viscous DNA solution moves more slowly!
2 fluorescent spheres in water (left) and DNA solution (right)
a
TtkTtkx BB
3
22
Copyright (c) Stuart Lindsay 2008
(Movie Courtesy Professor Eric Weeks, Emory University: http://www.seas.harvard.edu/weitzlab/research/brownian.html)

The Diffusion EquationThe Diffusion Equation
Flow of solute or heat under action of random forces:
J is flux per second per unit area
xA
txAJtxxJAC
)()(.
0, txx
txJ
t
txC
),(),(
The flux is the change in concentration across a surface multiplied by the speed with which particles arrive:
t
x
x
C
t
xCJ
2

Using: Dt
x
2
x
CDJ
From which
2
2 ),(),(
x
txCD
t
txC
DiffusionDiffusioncoefficientcoefficient
Fick’s first lawFick’s first law
Fick’s second lawFick’s second lawDiffusion EquationDiffusion Equation
In 3D: )t,r(CDr
)t,r(CD
t
)t,r(C 22
2
12 sm

The solution in 1-D for a solute initially added as a point source is:
Dt
x
Dt
AtxC
4exp),(
2
Dtx / 221
A=1
As t→∞, the distribution becomes uniform, the point of ‘half-maximum concentration’ x½ advancing with time according to:

Einstein-Smoluchowski RelationEinstein-Smoluchowski Relation
Dtx / 221
Fa
Fv
6The mobility is defined as the inverse of the friction coefficient:
a
TkD B
6
TkD BEinstein-Smoluchowski relationEinstein-Smoluchowski relation
a
TtkTtkx BB
3
22 Compare to
A surprising relation between thermal motion and driven motion: the diffusion constant is the ratio of kT to the friction constant!
A fundamental relation between energy dissipation and diffusion

Viscosity is not an equilibrium property, because viscous forces are generated only by movement that transfers energy from one part of the system to another.
Ex. Motion of a spherical large particle with respect to a large number of small molecules.
vaF 6
Forceradius
viscosity
speed
Stokes’ lawStokes’ law
The introduction of bulk viscosity requires that the small molecules rearrange themselves on very short times compared with the time scale of the motion of the sphere.

Diffusion, fluctuations and chemical reactionsDiffusion, fluctuations and chemical reactions
1. If the reactants are not already mixed they need to come together by diffusion.
2. Once together, they need to be jiggled by thermal fluctuations into a “transition state”
3. If the free energy of the products is lower than that of the reactants the products lose heat to the environment to form stable end products.

Haber process for AmmoniaHaber process for Ammonia
The transition state
Tk
G
h
Tkk
B
B exp1
Eyring transition state theory
Copyright (c) Stuart Lindsay 2008

The entropy adds a temperature-dependent component to the energy differences that determine the final equilibrium state of a system.

Kramers’ Theory of Chemical ReactionsKramers’ Theory of Chemical Reactions
• Noise driven escape:
Thermal fluctuations allow the particles in the well to rapidly equilibrate with the surroundings.The motion of the particles over the barrier is much slower.

The Kramers ModelThe Kramers Model
Reaction coordinate
Tk
Eexpk
B
bba
2
Microscopic description of the prefactor in terms of potential curvature
c,b,ax
c,b,a x
U
m
2
22 1

Unimolecular reactionsUnimolecular reactions
• Not very common, but can be described as a one step process by the Kramers theory
• Example is isomerism of isonitrile
E1 E2
k-
k+
CH3
NC
CH3
CN
211 EkEk
dt
Ed
Copyright (c) Stuart Lindsay 2008

Thermodynamic Potentials for NanosystemsThermodynamic Potentials for Nanosystems
• The Gibbs free energy in a multicomponent system is:
This equation contains no reference to system size (all quantities are extensive: doubling the volume of a system, doubles its free energy)
• Nanosystems at equilibrium derive their “stable” size from Nanosystems at equilibrium derive their “stable” size from surface and interface effects which are not extensive.surface and interface effects which are not extensive.
Ex. Self-assembly originates from a competition between bulk Ex. Self-assembly originates from a competition between bulk and surface energies of the phases that self-assemble (stability and surface energies of the phases that self-assemble (stability of colloidal systems).of colloidal systems).
NTSPVEG
Copyright (c) Stuart Lindsay 2008

• Hill has generalized thermodynamics to include a “subdivision potential”
dd
dEdNVdpTdSdE
The simplest approach add surface terms to the free energy.
Subdivision potential
number of independent parts of the system

Modeling nanosystems explicitly:Modeling nanosystems explicitly:Molecular DynamicsMolecular Dynamics
irij
ij rr
rUF
2
2 )(
TkVm Bzyxii 2
1
2
1 2,,,
Limited to s timescales by required time step/processor speed.
Interatomic vibrations are rapid (≈1013 Hz) so that the time step in the calculations must be much smaller (≈10-15 s).
Copyright (c) Stuart Lindsay 2008

• The number of calculations scales as N2 (it depends on the problem).
If all possible pair interactions need to be considered , the number of interactions would scale like 2N.
Limitations of MDLimitations of MD
• Timescale is limited to ns or s at best. This is fine for harmonic vibrations but nanosystems like enzymes work on ms timescales.
Ex. Viscously damped motion:
Ex. Activated transitions over barriers:
m
a
61
Tk
Ek
B
bba
exp2

Example of MD
Cyclic Sugar molecule being pulled over DNA
Qamar et al. Biophys J. (2008) 94, 1233
Movie link:13mer.avi

Thermal Fluctuations and Quantum Thermal Fluctuations and Quantum MechanicsMechanics
• The density matrix formulation of quantum mechanics allows description of the time evolution of a system subject to time dependent forces.
Copyright (c) Stuart Lindsay 2008
ˆ,Ht
ˆi
q*nnm aaqn

• Thermal fluctuations can be represented by the “random phase approximation” – quantum interference effects are destroyed.

Uncertainty Principle and fluctuationsUncertainty Principle and fluctuations
• Interaction energy, , will decay exponentially at large distances
• When kT
The time characteristic of a molecular vibration.
Copyright (c) Stuart Lindsay 2008
pseV.
seV
Et 1
0250
104 14
Related Documents