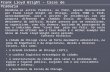
Page 1 of 2 Flow Cytometry Report PHYSICIAN PATIENT Patient: SAMPLE PATIENT DOB/Sex: MR/Chart #: Specimen: BONE MARROW Collected: 3/27/2012 Received: 3/28/2012 SAMPLE DOCTOR Copies: REPORT DATE: 3/28/2012 SPECIMEN Accession #: All Specimens: 2 BM NA HEP, 1 BM EDTA 1. Increased Myeloid Cells 2. Mild Increase in Myeloblasts with an Abnormal Immunophenotype INTERPRETATION: Comment: These abnormalities can be seen in a variety of myeloid neoplasms, including myeloproliferative, myeloproliferative/myelodysplastic and myelodysplastic disorders. Correlation with the CBC/Diff data and examination of a stained bone marrow aspirate smear suggest that a myeloproliferative neoplasm is most likely in this case. CD34 positive blasts account for 2% of total white blood cells and have an aberrant myeloid immunophenotype. Blasts espress CD34, CD38, dim CD33, bright HLA-DR, dim CD11b, dim CD117, dim CD45, without expression of CD13, CD56, CD7, CD2, CD16, CD14, CD41, CD64 and CD61. The viability of the specimen is 82%. The granulocytes (88% of the total white blood cells) and monocytes (1% of the total white blood cells) reveal no significant immunophenotypic abnormalities. B-cells comprise 8% of the lymphocytes and have a kappa:lambda ratio of 1.03. Approximately 78% of lymphocytes are T-cells with a CD4:CD8 ratio of 1.14 (Normal 1-4). NK cells account for 1% of the total white blood cells (14% of lymphocytes). Plasma cells are not detected. FLOW CYTOMETRY DIFFERENTIAL (% of total cells) LYMPHOCYTES 4 B CELLS 4 Kappa:Lambda Ratio 1.03 T CELLS 38 CD4:CD8 Ratio 1.14 Large Granular Lymphs 5 NK CELLS 7 MONOCYTES 1 GRANULOCYTES 88 BLASTS 2 PLASMA CELLS Negative VIABILITY 82 ANTIBODIES ANALYZED: Antibodies used for evaluation include: CD2,3,4,5,7,8,10,11b,13,16,19,20,33,34,38,45,56,57,7AAD, HLADR, kappa, lambda,14,41,61,64,117 Leukocytosis, thrombocytosis. CLINICAL HISTORY: The immunohistochemical assays were developed and their performance characteristics determined by OncoMetrix. They have not been cleared or approved by the U.S. Food and Drug Administration. The FDA has determined that such clearance or approval is not necessary. This testing is used for clinical purposes. It should not be regarded as investigational or for research. This laboratory is certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA) as qualified to perform high complexity clinical testing. IDC9 Codes: 288.60, 238.71 CPT Codes: 88184; 88185x27; 88189 (877) 670-HEME (4363) Dr. Douglas W. Kingma, Laboratory Director OncoMetrix CLIA #: 44D0915029, TN License #: 0000003284 150 Collins Street, Memphis, TN 38812 Poplar Healthcare, PLLC Howard L. Martin, III, M.D., Medical Director BONE MARROW ASPIRATE:

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript
-
Page 1 of 2
Flow Cytometry Report
PHYSICIANPATIENTPatient: SAMPLE PATIENTDOB/Sex:MR/Chart #:
Specimen: BONE MARROWCollected: 3/27/2012Received: 3/28/2012
SAMPLE DOCTORCopies:REPORT DATE: 3/28/2012
SPECIMEN
Accession #: All Specimens: 2 BM NA HEP, 1 BM EDTA
1. Increased Myeloid Cells2. Mild Increase in Myeloblasts with an Abnormal Immunophenotype
INTERPRETATION:
Comment:These abnormalities can be seen in a variety of myeloid neoplasms, including myeloproliferative,myeloproliferative/myelodysplastic and myelodysplastic disorders. Correlation with the CBC/Diff data andexamination of a stained bone marrow aspirate smear suggest that a myeloproliferative neoplasm is most likely inthis case.
CD34 positive blasts account for 2% of total white blood cells and have an aberrant myeloid immunophenotype.Blasts espress CD34, CD38, dim CD33, bright HLA-DR, dim CD11b, dim CD117, dim CD45, without expression ofCD13, CD56, CD7, CD2, CD16, CD14, CD41, CD64 and CD61. The viability of the specimen is 82%. Thegranulocytes (88% of the total white blood cells) and monocytes (1% of the total white blood cells) reveal nosignificant immunophenotypic abnormalities. B-cells comprise 8% of the lymphocytes and have a kappa:lambdaratio of 1.03. Approximately 78% of lymphocytes are T-cells with a CD4:CD8 ratio of 1.14 (Normal 1-4). NK cellsaccount for 1% of the total white blood cells (14% of lymphocytes). Plasma cells are not detected.
FLOW CYTOMETRY DIFFERENTIAL(% of total cells)
LYMPHOCYTES 4 B CELLS 4Kappa:Lambda Ratio 1.03 T CELLS 38CD4:CD8 Ratio 1.14 Large Granular Lymphs 5 NK CELLS 7MONOCYTES 1GRANULOCYTES 88BLASTS 2PLASMA CELLS NegativeVIABILITY 82
ANTIBODIES ANALYZED:Antibodies used for evaluation include: CD2,3,4,5,7,8,10,11b,13,16,19,20,33,34,38,45,56,57,7AAD, HLADR, kappa,lambda,14,41,61,64,117
Leukocytosis, thrombocytosis.CLINICAL HISTORY:
The immunohistochemical assays were developed and their performance characteristics determined by OncoMetrix. They have not been cleared or approved by the U.S. Food and DrugAdministration. The FDA has determined that such clearance or approval is not necessary. This testing is used for clinical purposes. It should not be regarded as investigational or forresearch. This laboratory is certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA) as qualified to perform high complexity clinical testing.
IDC9 Codes: 288.60, 238.71 CPT Codes: 88184; 88185x27; 88189(877) 670-HEME (4363)
Dr. Douglas W. Kingma, Laboratory Director
OncoMetrix
CLIA #: 44D0915029, TN License #: 0000003284
150 Collins Street, Memphis, TN 38812Poplar Healthcare, PLLC
Howard L. Martin, III, M.D., Medical Director
BONE MARROW ASPIRATE:
-
Page 2 of 2
Flow Cytometry Report
PHYSICIANPATIENTPatient: SAMPLE PATIENTDOB/Sex:MR/Chart #:
Specimen: BONE MARROWCollected: 3/27/2012Received: 3/28/2012
SAMPLE DOCTORCopies:REPORT DATE: 3/28/2012
SPECIMEN
Accession #: All Specimens: 2 BM NA HEP, 1 BM EDTA
IMAGES:
Increased myeloid cells; smallpopulation of blasts present.
Normal myeloid immunophenotype inmaturing myeloid cells.
Normal myeloid immunophenotype inmaturing myeloid cells.
Blasts show aberrant co-expressionof CD34 and CD11b.
Polyclonal B-cells. Normal CD4:CD8 ratio.
Mihaela Onciu, M.D.Hematopathologist
Electronically signed3/28/2012 4:43 PM by
The immunohistochemical assays were developed and their performance characteristics determined by OncoMetrix. They have not been cleared or approved by the U.S. Food and DrugAdministration. The FDA has determined that such clearance or approval is not necessary. This testing is used for clinical purposes. It should not be regarded as investigational or forresearch. This laboratory is certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA) as qualified to perform high complexity clinical testing.
IDC9 Codes: 288.60, 238.71 CPT Codes: 88184; 88185x27; 88189(877) 670-HEME (4363)
Dr. Douglas W. Kingma, Laboratory Director
OncoMetrix
CLIA #: 44D0915029, TN License #: 0000003284
150 Collins Street, Memphis, TN 38812Poplar Healthcare, PLLC
Howard L. Martin, III, M.D., Medical Director
Related Documents