Extracellular Matrix 1 Contents 1 Extracellular Matrix 2 Introduction 2.1 Note 2.2 Archive 3 Objectives 4 ECM Function 5 ECM Structure 6 Shapes and Sizes ECM molecules 7 Collagen 8 Collagen Components 8.1 Collagen Structure 8.2 Collagen Fibers 8.3 Collagen Interactions 8.4 Collagen Type Functions 8.5 Collagen Proteins 8.6 Collagen Synthesis 9 Collagen Diseases 9.1 Collagen Diseases - Excess 9.2 Collagen Diseases - Insufficient 10 Elastin 10.1 Microfibrils 10.2 Elastin Structure 10.3 Elastin Function 10.4 Elastin Disorders 11 References 11.1 Recent Reviews 11.2 Textbooks 11.2.1 Essential Cell Biology 11.2.2 Molecular Biology of the Cell 11.2.3 Molecular Cell Biology 11.2.4 The Cell- A Molecular Approach 11.2.5 Essentials of Glycobiology, 2nd ed. 11.2.6 Search Online Textbooks 11.3 Books 11.4 PubMed 11.4.1 Search Pubmed 11.4.2 Reviews 11.4.3 Articles 12 Links 13 2017 Course Content Extracellular Matrix Introduction

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

Extracellular Matrix 1Contents
1 Extracellular Matrix2 Introduction
2.1 Note2.2 Archive
3 Objectives4 ECM Function5 ECM Structure6 Shapes and Sizes ECM molecules7 Collagen8 Collagen Components
8.1 Collagen Structure8.2 Collagen Fibers8.3 Collagen Interactions8.4 Collagen Type Functions8.5 Collagen Proteins8.6 Collagen Synthesis
9 Collagen Diseases9.1 Collagen Diseases - Excess9.2 Collagen Diseases - Insufficient
10 Elastin10.1 Microfibrils10.2 Elastin Structure10.3 Elastin Function10.4 Elastin Disorders
11 References11.1 Recent Reviews11.2 Textbooks
11.2.1 Essential Cell Biology11.2.2 Molecular Biology of the Cell11.2.3 Molecular Cell Biology11.2.4 The Cell- A Molecular Approach11.2.5 Essentials of Glycobiology, 2nd ed.11.2.6 Search Online Textbooks
11.3 Books11.4 PubMed
11.4.1 Search Pubmed11.4.2 Reviews11.4.3 Articles
12 Links13 2017 Course Content
Extracellular MatrixIntroduction

(/cellbiology/index.php/File:Skthick0021he.jpg)Skin
(/cellbiology/index.php/File:Cell_adhesion_summary.png)Cell Adhesion
(/cellbiology/index.php/File:09lungtem.jpg)Collagen (type I) Fibres
This lecture introduces the materials lying outside the cell, knowncollectively as the extracellular matrix (ECM). There is no one matrixthough, with different tissues having their own specific ECM, whichmay be dynamic or static in structure. In particular the ECM hassignificant roles in normal tissue development, function and disease.This matrix is manufactured by cells, secreted and modified outside thecell by several different enzymes.
This lecture introduces the ECM, describes the major fiber (fibre) andmatrix components and will then cover the major ECM glycoproteinsand experimental studies of ECM function.
Spelling - UK sulphate, US sulfate ; UK fibre, US fiber
NoteTake a look at the 2015 student group projects designed around ECM -2015 Extracellular Matrix: Group 1 - Small Leucine-RichProteoglycans (/cellbiology/index.php/2015_Group_1_Project) | Group2 - Integrins (/cellbiology/index.php/2015_Group_2_Project) | Group 3 -Elastic Fibres (/cellbiology/index.php/2015_Group_3_Project) | Group 4- Fibronectin (/cellbiology/index.php/2015_Group_4_Project) | Group 5 -Laminin (/cellbiology/index.php/2015_Group_5_Project) | Group 6 -Collagen (/cellbiology/index.php/2015_Group_6_Project) | Group 7 -Basement Membrane (/cellbiology/index.php/2015_Group_7_Project)
ArchiveMH - note that content listed below willnot match exactly current lecturestructure but has been selected ashaving similar content
2016
(https://cellbiology.med.unsw.edu.au/cellbiology/index.php?title=Extracellular_Matrix_1&oldid=77626) |2010 (/cellbiology/index.php/2010_Lecture_13) | 2009 (/cellbiology/index.php/2009_Lecture_14) | 2008(http://cellbiology.med.unsw.edu.au/units/pdf/08L12-13s1.pdf) | 2008 4 slides/page(http://cellbiology.med.unsw.edu.au/units/pdf/08L12-13s4.pdf) |

(/cellbiology/index.php/File:ECM_Collagen_triplehelix_sm.png)Collagen triplehelix
ObjectivesUnderstand the localisation and origin of extracellular matrixUnderstand the 3 major components
fibers, proteoglycans (matrix), adhesive glycoproteinsBroad understanding of structure and function
collagen fiberselastin fibersproteoglycans
ECM Function
Support for cellsPattern of ECMregulates
polaritycell divisionadhesionmotility
Developmentmigrationdifferentiationgrowthfactors
(/cellbiology/index.php/File:Evolution_of_metazoan_extracellular_matrix_PMID22431747.jpg)Evolution of metazoan extracellular matrix
ECM Features

[Expand]
stable and able to be reorganised?different for different tissues
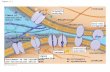
| Figure 19-61. How the extracellular matrix could, in principle, propagate order from cell to cell within atissue (http://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=mboc4&part=A3532&rendertype=figure&id=A3586)
Cell Walls/ECM?
ECM StructureGlycoproteinsFibers
Collagen- main fibersElastin
Hydrated MatrixProteoglycanshigh carbohydrate
AdhesiveLamininFibronectin
Shapes and Sizes ECM molecules|MBoC Figure 19-59. The comparative shapes and sizes of some of the major extracellular matrixmacromolecules (http://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=mboc4&part=A3532&rendertype=figure&id=A3582)Note the relative size and differential contribution of protein (green) and glycosaminoglycan (red)
Collagentensile strength and elasticity
TendonsCartilageBone
half total body proteins (by weight)
Collagen ComponentsInsoluble glycoprotein
protein + carbohydrateProtein
high glycinehigh prolinehydroxylysinehydroxyproline(gly-X-Y)n - any of 20 standard amino acids may occupy the X position and/or the Y position (Yposition- hydroxylysine, hydroxyproline)most frequently observed triplet is Gly-Pro-Hyp (10.5%) PMID 9724608
Carbohydrate
glucose

(/cellbiology/index.php/File:Collagen_cartoon2.jpg)Collagen Fiber structure and size
(/cellbiology/index.php/File:11lungtem.jpg)Collagen Type I
galactose
Collagen StructureCollagen Protein
3 polypeptide (a) chainsleft hand helix, forms fibersmany different (vertebrate) collagens by differentcombinations of a-chainsType I, II, III
main fibers, flexibleType I
bone, skin, tendons90% of all collagen
Type IIcartilage
Collagen FibersType I, II, III cross striated
e.g. tendons - type I fibrils, have a 67-nmperiod striations and are oriented longitudinally(direction of the stress)showing overlapping packing of individualcollagen moleculesreticular fibres type III, support individual cells
Type IV fine unstriatedsheet-like supportive meshworkmature basal laminaetracks for embryonic migrationbarriers for cell migration
Type V-XIIsmaller diameter fibers than I-IIIno striations
Collagen InteractionsCollagen fibril types can interact with a variety of non-fibrous collagen types (microfiber)
fibrous collagens—types I, II, III, and VCartilage - types II (fiber) and IX collagen microfibrilsTendons - type I fibrils bound and linked by type VI microfibrils.
MCB - Interactions of fibrous and nonfibrous collagens (http://www.ncbi.nlm.nih.gov/books/bv.fcgi?&rid=mcb.figgrp.6555)
MBoC Type IX collagen (http://www.ncbi.nlm.nih.gov/books/bv.fcgi?highlight=collagen&rid=mboc4.figgrp.3565)
Collagen Type FunctionsCollagen Type I - skin, tendon, vascular, ligature, organs, bone (main component of bone)Collagen Type II - cartilage (main component of cartilage)Collagen Type III - reticular fibers with type I.

[Expand]
(/cellbiology/index.php/File:Skthick0021he.jpg)Skin
(/cellbiology/index.php/File:Fibrillar_collagen_01.jpg)Fibrillar collagen
Collagen Type IV - forms bases of cell basement membraneCollagen Type V - hair and nail
You do not need to know the protein table below in detail, just themajor type/functions shown above.
Collagen ProteinsCollagen Types
Collagen Synthesis
Endoplasmic ReticulummRNA attached to ERprotein synthesized into ER lumencotranslational and post-translational modifications3 proto-a-chains form soluble procollagenmoved to golgi apparatus
Golgi Apparatus

[Expand]
packed into secretion vesiclesfuse with membrane
Outside Cellprocollagen processed by enzymes outside cellassemble into collagen fiberscollagen fibrils form lateral Interactions of triple helices
Link: MBoC - The intracellular and extracellular events in theformation of a collagen fibril(http://www.ncbi.nlm.nih.gov/books/bv.fcgi?&rid=mboc4.figgrp.3559)| MCB - Collagen synthesis(http://www.ncbi.nlm.nih.gov/books/bv.fcgi?&rid=mcb.figgrp.6551)
(/cellbiology/index.php/File:Collagen-synthesis.jpg)
(/cellbiology/index.php/File:Collagen-fibril-
em.jpg)
Collagen Assembly
Collagen in the ECM is modified/cleaved by matrix metalloproteinases (MMPs) - family of enzymes

(calcium-dependent zinc-containing endopeptidases)
Collagen DiseasesCollagen Diseases - Excess
fibrosislung- pulmonary fibrosisoverproduction of collagen Iliver- over consumption of alcoholarteries- atherosclerosis
Collagen Diseases - InsufficientEhlers-Danlos syndrome
defect in the synthesis of Type I or III collagenrubber-manskin and tendons easily stretchedcontortionists often suffer from this disease
Osteogenesis imperfectabrittle-bone syndromemutation in Type I procollagenfail to assemble triple helixdegrade imperfect collagenLeads to fragile bones
Scurvydietary Vitamin C deficiencyneeded for hydroxylationProline -> Hydroxyprolineform too few hydrogen bonds in collagenskin, bone, teeth weakness and malformationblood vessels weakened, bleeding
ElastinMBoC - Collagen and elastin (http://www.ncbi.nlm.nih.gov/books/bv.fcgi?&rid=mboc4.figgrp.435)
Elastin in Aorta (http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2679973/figure/F1/) PMID 18248974
elastin and elastic fibresuncoils into an extended conformation when the fiber is stretchedrecoils spontaneously as soon as the stretching force is relaxed
Elastic fibers are composed of a core of cross-linked elastin embedded within a peripheral mantle ofmicrofibrils.
Microfibrils

(/cellbiology/index.php/File:ECM_elastic_fibres_skin.jpg)Elastic Fibers Skin
(/cellbiology/index.php/File:Fibrilin-microfibrils.jpg)
may regulate assembly and organization of elastic fibers by acting as a scaffoldguiding tropoelastin depositionaggregates of threadlike filamentsperiodically spaced globular domains (beads) connected by multiple linear arms
beaded structure is parallel fibrillin monomers aligned head-to-tailfibulin-5 induces elastic fiber assembly and maturation by organizing tropoelastin and cross-linkingenzymes onto microfibrils PMID: 17371835 (http://www.ncbi.nlm.nih.gov/pubmed/17371835)
Links: MBoC - Collagen and elastin (http://www.ncbi.nlm.nih.gov/books/bv.fcgi?&rid=mboc4.figgrp.435)

(/cellbiology/index.php/File:ECM_elastic-fiber_synthesis.jpg)elastic fiber synthesis
(/cellbiology/index.php/File:ECM_elastic_fiber_assemby_model.jpg)elastic fiber assemby model
Elastin Structureprotein Mr 64 to 66 kDacomposed of the amino acids glycine, valine,alanine, and prolinecross-linked tropoelastin monomersfirst secreted as soluble precursors (tropoelastin)assembly and crosslinking of tropoelastinmonomersform insoluble elastin matrix into functional fibres
lysine residues in the cross-linking domain ofsecreted tropoelastin rapidly cross-linked (bothinter- and intra-molecularly by lysyl oxidase)hydrophobic segments - elastic propertiesα-helical segments (alanine- and lysine-rich) -form cross-links between adjacent molecules
Links: Nature - Fibulin-5 is an elastin-binding protein
(http://www.nature.com/nature/journal/v415/n6868/full/415168a.html)
Elastin Functionstructural integrity and function of tissuesrequiring reversible extensibility or deformabilityhigh levels in tissues that require elasticity
lung, skin, major blood vessels
Elastin DisordersWilliams Syndrome (http://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=gene&part=williams#williams)
ReferencesRecent Reviews

Adam Byron, Margaret C Frame Adhesion protein networks reveal functions proximal and distal tocell-matrix contacts. Curr. Opin. Cell Biol.: 2016, 39;93-100 PubMed 26930633(http://www.ncbi.nlm.nih.gov/pubmed/26930633)
Lauren E Tracy, Raquel A Minasian, E J Caterson Extracellular Matrix and Dermal Fibroblast Function inthe Healing Wound. Adv Wound Care (New Rochelle): 2016, 5(3);119-136 PubMed 26989578(http://www.ncbi.nlm.nih.gov/pubmed/26989578)
Kristen M Kwan Coming into focus: the role of extracellular matrix in vertebrate optic cupmorphogenesis. Dev. Dyn.: 2014, 243(10);1242-8 PubMed 25044784(http://www.ncbi.nlm.nih.gov/pubmed/25044784)
TextbooksEssential Cell Biology
Essential Cell Biology Chapter 19 p594-604
Molecular Biology of the CellAlberts, Bruce; Johnson, Alexander; Lewis, Julian; Raff, Martin; Roberts, Keith; Walter, Peter New York andLondon: Garland Science; c2002
Molecular Biology of the Cell 4th ed. - V. Cells in Their Social Context Chapter 19. Cell Junctions, CellAdhesion, and the Extracellular MatrixThe Extracellular Matrix of Animals (http://www.ncbi.nlm.nih.gov:80/books/bv.fcgi?db=Books&rid=mboc4.section.3532)
Molecular Cell BiologyLodish, Harvey; Berk, Arnold; Zipursky, S. Lawrence; Matsudaira, Paul; Baltimore, David; Darnell, James E.New York: W. H. Freeman & Co.; c1999
Molecular Cell Biology - Chapter 22. Integrating Cells into TissuesCell-Matrix Adhesion (http://www.ncbi.nlm.nih.gov:80/books/bv.fcgi?db=Books&rid=mcb.section.6526)
The Cell- A Molecular ApproachCooper, Geoffrey M. Sunderland (MA): Sinauer Associates, Inc.; c2000
The Cell- A Molecular ApproachThe Cell - A Molecular Approach - III. Cell Structure and Function Chapter 12. The Cell SurfaceThe Extracellular Matrix (http://www.ncbi.nlm.nih.gov:80/books/bv.fcgi?db=Books&rid=cooper.section.2035#2045)
Essentials of Glycobiology, 2nd ed.Varki, A.; Cummings, R.D.; Esko, J.D.; Freeze, H.H.; Stanley, P.; Bertozzi, C.R.; Hart, G.W.; Etzler, M.E.,editors Plainview (NY): Cold Spring Harbor Laboratory Press; 2008
Proteoglycans and Sulfated Glycosaminoglycans (http://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=glyco2&part=ch16)hyaluronan-binding proteins (http://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=glyco2&part=ch15&rendertype=figure&id=ch15.f3)
Search Online Textbooks"extracellular matrix" Molecular Biology of the Cell (http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=extracellular+matrix+mboc4) | Molecular Cell Biology(http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=extracellular+matrix+mcb) |

The Cell- A molecular Approach (http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=extracellular+matrix+cooper) | Bookshelf(http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=extracellular+matrix)"collagen" Molecular Biology of the Cell (http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=collagen+mboc4) | Molecular Cell Biology(http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=collagen+mcb) | The Cell- Amolecular Approach (http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=collagen+cooper) | Bookshelf(http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=collagen)"elastin" Molecular Biology of the Cell (http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=elastin+mboc4) | Molecular Cell Biology(http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=elastin+mcb) | The Cell- Amolecular Approach (http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=elastin+cooper) | Bookshelf (http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=elastin)"glycosaminoglycan" Molecular Biology of the Cell (http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=glycosaminoglycan+mboc4) | Molecular Cell Biology(http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=glycosaminoglycan+mcb) |The Cell- A molecular Approach (http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=glycosaminoglycan+cooper) | Bookshelf(http://www.ncbi.nlm.nih.gov/sites/entrez?db=Books&cmd=search&term=glycosaminoglycan)
Books
PubMedPubMed is a service of the U.S. National Library of Medicine that includes over 18 million citationsfrom MEDLINE and other life science journals for biomedical articles back to 1948. PubMed includeslinks to full text articles and other related resources. PubMed(http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed)PubMed Central (PMC) is a free digital archive of biomedical and life sciences journal literature at theU.S. National Institutes of Health (NIH) in the National Library of Medicine (NLM) allowing all users freeaccess to the material in PubMed Central. PMC (http://www.ncbi.nlm.nih.gov/sites/entrez?db=PMC)Online Mendelian Inheritance in Man (OMIM) is a comprehensive compendium of human genes andgenetic phenotypes. The full-text, referenced overviews in OMIM contain information on all knownmendelian disorders and over 12,000 genes. OMIM (http://www.ncbi.nlm.nih.gov/sites/entrez?db=omim)Entrez is the integrated, text-based search and retrieval system used at NCBI for the major databases,including PubMed, Nucleotide and Protein Sequences, Protein Structures, Complete Genomes,Taxonomy, and others Entrez (http://www.ncbi.nlm.nih.gov/sites/gquery?itool=toolbar)
Search Pubmed"extracellular matrix" Entrez all databases (http://www.ncbi.nlm.nih.gov/sites/gquery?itool=toolbar&cmd=search&term=extracellular+matrix)"collagen" Entrez all databases (http://www.ncbi.nlm.nih.gov/sites/gquery?itool=toolbar&cmd=search&term=collagen)"elastin" Entrez all databases (http://www.ncbi.nlm.nih.gov/sites/gquery?itool=toolbar&cmd=search&term=elastin)"glycosaminoglycan" Entrez all databases (http://www.ncbi.nlm.nih.gov/sites/gquery?itool=toolbar&cmd=search&term=glycosaminoglycan)"proteoglycan" Entrez all databases (http://www.ncbi.nlm.nih.gov/sites/gquery?itool=toolbar&cmd=search&term=proteoglycan)

(http://moodle.telt.unsw.edu.au/course/view.php?id=25143)Moodle
note the spelling differences when carrying out other related ECM searches: UK sulphate, USsulfate ; UK fibre, US fiber
ReviewsCanty EG, Kadler KE. Procollagen trafficking, processing and fibrillogenesis. J Cell Sci. 2005 Apr 1;118(Pt7):1341-53. Review. PMID: 15788652 (http://www.ncbi.nlm.nih.gov/pubmed/15788652) JCS Link(http://jcs.biologists.org/cgi/content/full/118/7/1341)
Hay ED. The extracellular matrix in development and regeneration. An interview with Elizabeth D. Hay.Int J Dev Biol. 2004;48(8-9):687-94. No abstract available. PMID: 15558460(http://www.ncbi.nlm.nih.gov/pubmed/15558460)Hay ED. Extracellular matrix. J Cell Biol. 1981 Dec;91(3 Pt 2):205s-223s. Review. No abstract available.PMID: 6172429 (http://www.ncbi.nlm.nih.gov/pubmed/6172429)Sandberg LB, Soskel NT, Leslie JG. Elastin structure, biosynthesis, and relation to disease states. NEngl J Med. 1981 Mar 5;304(10):566-79. Review. PMID: 7005671(http://www.ncbi.nlm.nih.gov/pubmed/7005671)Wagenseil JE, Mecham RP. New insights into elastic fiber assembly. Birth Defects Res C EmbryoToday. 2007 Dec;81(4):229-40. Review. PMID: 18228265(http://www.ncbi.nlm.nih.gov/pubmed/18228265)
ArticlesSandberg LB, Soskel NT, Leslie JG. Elastin structure, biosynthesis, and relation to disease states. NEngl J Med. 1981 Mar 5;304(10):566-79. Review.
PMID: 7005671 (http://www.ncbi.nlm.nih.gov/pubmed/7005671?dopt=Abstract)
LinksMarfan syndrome is an autosomal dominant disorder that has been linked to the FBN1 gene onchromosome 15. FBN1 encodes a protein called fibrillinCommunication between the intra- and extracellular compartment(http://multimedia.mcb.harvard.edu/anim_innerlife.html)
2017 Course ContentLectures: Cell Biology Introduction (/cellbiology/index.php/Cell_Biology_Introduction) |Cells Eukaryotes and Prokaryotes(/cellbiology/index.php/Cells_Eukaryotes_and_Prokaryotes) | Cell Membranes andCompartments (/cellbiology/index.php/Cell_Membranes_and_Compartments) | CellNucleus (/cellbiology/index.php/Cell_Nucleus) | Cell Export - Exocytosis(/cellbiology/index.php/Cell_Export_-_Exocytosis) | Cell Import - Endocytosis(/cellbiology/index.php/Cell_Import_-_Endocytosis) | Cytoskeleton Introduction(/cellbiology/index.php/Cytoskeleton_Introduction) | Cytoskeleton - Microfilaments(/cellbiology/index.php/Cytoskeleton_-_Microfilaments) | Cytoskeleton - Microtubules(/cellbiology/index.php/Cytoskeleton_-_Microtubules) | Cytoskeleton - Intermediate Filaments(/cellbiology/index.php/Cytoskeleton_-_Intermediate_Filaments) | Cell Mitochondria(/cellbiology/index.php/Cell_Mitochondria) | Cell Junctions (/cellbiology/index.php/Cell_Junctions) |Extracellular Matrix 1 | Extracellular Matrix 2 (/cellbiology/index.php/Extracellular_Matrix_2) | Cell Cycle(/cellbiology/index.php/Cell_Cycle) | Cell Division (/cellbiology/index.php/Cell_Division) | Cell Death 1(/cellbiology/index.php/Cell_Death_1) | Cell Death 2 (/cellbiology/index.php/Cell_Death_2) | Signal 1(/cellbiology/index.php/Signal_1) | Signal 2 (/cellbiology/index.php/Signal_2) | Stem Cells 1(/cellbiology/index.php/Stem_Cells_1) | Stem Cells 2 (/cellbiology/index.php/Stem_Cells_2) | Development(/cellbiology/index.php/Development) | 2017 Revision (/cellbiology/index.php/2017_Revision)

2017 Laboratories: Introduction to Lab (/cellbiology/index.php/2017_Lab_1) | Fixation and Staining(/cellbiology/index.php/2017_Lab_2) |
2017 Projects (/cellbiology/index.php/ANAT3231_Projects_2017): Group 1 - Delta(/cellbiology/index.php/2017_Group_1_Project) | Group 2 - Duct(/cellbiology/index.php/2017_Group_2_Project) | Group 3 - Beta(/cellbiology/index.php/2017_Group_3_Project) | Group 4 - Alpha(/cellbiology/index.php/2017_Group_4_Project)
Dr Mark Hill 2015, UNSW Cell Biology - UNSW CRICOS Provider Code No. 00098G
Categories (/cellbiology/index.php/Special:Categories): Science-Undergraduate (/cellbiology/index.php/Category:Science-Undergraduate)2017ANAT3231 (/cellbiology/index.php/Category:2017ANAT3231)
This page was last modified on 25 April 2017, at 12:06.
(//www.mediawiki.org/)
Related Documents