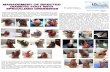
* Dressing A = Mepilex® Border Post-Op (10x25cm) Dressing D = Opsite Post-Op (10x25cm) Dressing B = Aquacel Surgical (9x25cm) Dressing E = Tegaderm™+Pad (9x25cm) Dressing C = Opsite Post-Op Visible (10x25cm) A LABORATORY EVALUATION OF THE BLOOD ABSORPTION PROPERTIES OF SURGICAL DRESSINGS AUTHOR: Malin Holmén (Research & Development Department, Mölnlycke Health Care, Gothenburg, Sweden) INTRODUCTION Surgical wounds can be associated with substantial blood loss at a rapid rate. Consequently, post-operative wound dressings with high absorption capacities and good dispersion characteristics should be selected in order to minimize the frequency of costly dressing changes. Each dressing change risks exposing the post- operative wound to contamination and therefore a risk of superficial surgical site infection (SSI). Furthermore, dressings should minimize the risk of exudates leakage and moisture-related damage (e.g. maceration). AIMS The poster describes the results of an in vitro study that was undertaken to compare the blood absorption capacity and dispersion ability of different surgical dressings. The study was designed in such a way as to assess the ability of the dressings to handle large amounts of blood quickly over a relatively short period of time and to distribute fluid within them. METHOD Materials The test equipment consisted of a Plexiglas® plane with an adjustable inclination. From underneath the middle of the plane, a tube was connected. The tube was connected to a syringe pump from which liquid was supplied at a controlled and even flow rate. Methodology The dressings were attached to the Plexiglas® plane at an inclination of 60 degrees. Bovine blood (in sodium citrate solution to prevent it from coagulation) was added at a high constant flow rate (3ml/min) into the centre of each dressing. The volume of blood added was recorded until it reached the bottom edges of the wound dressing (indicator of absorption capacity against gravity) and until leakage from the wound pad occurred (indicator of retention capacity). Please see chart for results. Photographs of the dispersion patterns were taken at regular intervals (i.e. after the initial addition of 5ml of blood and then after each subsequent 5ml addition). Please see photo matrix for results. Tests were terminated when blood was observed to be leaking from the dressing, or when it was heavy accumulated under the dressing border, making it impossible to continue. The tests were undertaken on five samples of each dressing and the photographs shown are representative of each sample tested.

EWMA 2014 - EP502 A LABORATORY EVALUATION OF THE BLOOD ABSORPTION PROPERTIES OF SURGICAL DRESSINGS
May 24, 2015
Holmén Malin
Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

* Dressing A = Mepilex® Border Post-Op (10x25cm) Dressing D = Opsite Post-Op (10x25cm)
Dressing B = Aquacel Surgical (9x25cm) Dressing E = Tegaderm™+Pad (9x25cm)
Dressing C = Opsite Post-Op Visible (10x25cm)
A LABORATORY EVALUATION OF THE BLOOD ABSORPTION
PROPERTIES OF SURGICAL DRESSINGS AUTHOR: Malin Holmén (Research & Development Department, Mölnlycke Health Care, Gothenburg, Sweden)
INTRODUCTION
Surgical wounds can be associated with substantial blood loss at a rapid rate. Consequently, post-operative
wound dressings with high absorption capacities and good dispersion characteristics should be selected in
order to minimize the frequency of costly dressing changes. Each dressing change risks exposing the post-
operative wound to contamination and therefore a risk of superficial surgical site infection (SSI).
Furthermore, dressings should minimize the risk of exudates leakage and moisture-related damage (e.g.
maceration).
AIMS
The poster describes the results of an in vitro study that was undertaken to compare the blood absorption
capacity and dispersion ability of different surgical dressings. The study was designed in such a way as to
assess the ability of the dressings to handle large amounts of blood quickly over a relatively short period of
time and to distribute fluid within them.
METHOD
Materials
The test equipment consisted of a Plexiglas® plane with an adjustable inclination. From underneath the middle of the plane, a tube was connected. The tube was connected to a syringe pump from which liquid was supplied at a controlled and even flow rate.
Methodology
The dressings were attached to the Plexiglas® plane at an inclination of 60 degrees. Bovine blood (in sodium citrate solution to prevent it from coagulation) was added at a high constant flow rate (3ml/min) into the centre of each dressing. The volume of blood added was recorded until it reached the bottom edges of the wound dressing (indicator of absorption capacity against gravity) and until leakage from the wound pad occurred (indicator of retention capacity). Please see chart for results. Photographs of the dispersion patterns were taken at regular intervals (i.e. after the initial addition of 5ml of blood and then after each subsequent 5ml addition). Please see photo matrix for results. Tests were terminated when blood was observed to be leaking from the dressing, or when it was heavy accumulated under the dressing border, making it impossible to continue. The tests were undertaken on five samples of each dressing and the photographs shown are representative of each sample tested.

* Dressing A = Mepilex® Border Post-Op (10x25cm) Dressing D = Opsite Post-Op (10x25cm)
Dressing B = Aquacel Surgical (9x25cm) Dressing E = Tegaderm™+Pad (9x25cm)
Dressing C = Opsite Post-Op Visible (10x25cm)
A LABORATORY EVALUATION OF THE BLOOD ABSORPTION
PROPERTIES OF SURGICAL DRESSINGS AUTHOR: Malin Holmén (Research & Development Department, Mölnlycke Health Care, Gothenburg, Sweden)
RESULTS
Dressing A* was able to handle more blood compared to the other surgical dressings tested (Dressings B-
E*), both in terms of dispersion against gravity and absorption capacity. Compared to the other test
dressings, Dressing A* was the most efficient at being able to disperse blood within it and absorb the
largest amount of blood without causing any leakage from either the wound pad or the dressing border.
DISCUSSION
The results indicate that there are significant differences in blood absorption properties between the
surgical dressings included in this evaluation. Since surgical wounds can be associated with substantial
amounts of bleeding, the results of this study are of relevance to the selection of dressings in the clinical
setting.
CONCLUSION
Based on the results of this in vitro evaluation, the blood absorbing properties of Dressing A* are superior
to those of the other surgical dressings tested. The results indicate that Dressing A* will minimize the need
for frequent dressing changes and therefore better protect the wound from outside contamination, the
result of which could be superficial SSI.
19
7,6 7,2
9,4 7,6
27,4
17,2
8,8 11,8 10
Dressing A* Dressing B* Dressing C* Dressing D* Dressing E*
Volume of blood added when blood was first observed at the bottom edge of the wound pad (ml)
Volume of blood added when leakage from the wound pad was first observed (ml)
Dressing A*
Dressing B* Dressing C* Dressing D* Dressing E*

* Dressing A = Mepilex® Border Post-Op (10x25cm) Dressing D = Opsite Post-Op (10x25cm)
Dressing B = Aquacel Surgical (9x25cm) Dressing E = Tegaderm™+Pad (9x25cm)
Dressing C = Opsite Post-Op Visible (10x25cm)
A LABORATORY EVALUATION OF THE BLOOD ABSORPTION
PROPERTIES OF SURGICAL DRESSINGS AUTHOR: Malin Holmén (Research & Development Department, Mölnlycke Health Care, Gothenburg, Sweden)
Dressing A*
Dressing B* Dressing C* Dressing D* Dressing E*
15 ml
25 ml
35 ml
Dressing A* 40 / 45 / 50 ml
Related Documents