Digest Paper Design of supramolecular chiral ligands for asymmetric metal catalysis Kohsuke Ohmatsu a , Takashi Ooi a,b,⇑ a Institute of Transformative Bio-Molecules (WPI-ITbM), and Department of Applied Chemistry, Graduate School of Engineering, Nagoya University, Chikusa, Nagoya 464-8603, Japan b CREST, Japan Science and Technology Agency (JST), Chikusa, Nagoya 464-8603, Japan article info Article history: Received 26 December 2014 Revised 17 February 2015 Accepted 17 February 2015 Available online 5 March 2015 Keywords: Supramolecular catalysis Metal catalysis Chiral ligand Asymmetric synthesis Combinatorial screening abstract Three strategies for the development of supramolecular chiral ligands for asymmetric metal catalysis are outlined. The basic ideas, advantages, and examples of each strategy are described. Ó 2015 Published by Elsevier Ltd. This is an open access article under the CC BY-NC-ND license (http:// creativecommons.org/licenses/by-nc-nd/4.0/). Contents Introduction.......................................................................................................... 2043 Supramolecular bidentate chiral ligands by assembly of two monodentate ligands ................................................ 2044 Assembly of chiral ligands with achiral building blocks....................................................................... 2045 Achiral ligands with chiral subcomponents ................................................................................ 2046 Conclusions .......................................................................................................... 2048 Acknowledgements .................................................................................................... 2048 References and notes .................................................................................................. 2048 Introduction Asymmetric metal catalysis has been regarded as one of the most powerful tools for the preparation of stereochemically pure chiral organic molecules. 1 Over the last few decades, a myriad of chiral metal complexes have been elaborated, paving the way for the development of various catalytic asymmetric transformations. 2 A chiral metal catalyst generally comprises a central metal atom and a chiral ligand, and its reactivity and selectivity are heavily depen- dent on the ligand attributes. Despite the deep insight gained into manifold reaction mechanisms and the advancement in theoretical chemistry, however, rational design of chiral ligands that can attain sufficient levels of catalytic efficiency and stereoselectivity in a tar- get transformation remains very difficult. Therefore, a process for the discovery of an effective chiral ligand for a given asymmetric reaction still involves numerous trial-and-error attempts. Conventional chiral ligands rely on a covalently constructed, single chiral molecule embedded with coordinative functional groups. Their syntheses from each single molecule with the requi- site chirality often require multi-step operation and can be compli- cated. This intrinsic problem hinders the iterative design, evaluation, and structural optimization processes in the identifica- tion of an optimal ligand. Thus, an alternative strategy that allows the simple and rapid preparation of structurally diverse chiral ligands would be of significant importance. Meanwhile, nature has evolved a wide variety of enzymes from a relatively small number of building blocks, and a multitude of http://dx.doi.org/10.1016/j.tetlet.2015.02.096 0040-4039/Ó 2015 Published by Elsevier Ltd. This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/). ⇑ Corresponding author. Tel.: +81 52 789 4501; fax: +81 52 789 3338. E-mail address: [email protected] (T. Ooi). Tetrahedron Letters 56 (2015) 2043–2048 Contents lists available at ScienceDirect Tetrahedron Letters journal homepage: www.elsevier.com/locate/tetlet

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript
-
Tetrahedron Letters 56 (2015) 2043–2048
Contents lists available at ScienceDirect
Tetrahedron Letters
journal homepage: www.elsevier .com/ locate/ tet le t
Digest Paper
Design of supramolecular chiral ligands for asymmetric metalcatalysis
http://dx.doi.org/10.1016/j.tetlet.2015.02.0960040-4039/� 2015 Published by Elsevier Ltd.This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/).
⇑ Corresponding author. Tel.: +81 52 789 4501; fax: +81 52 789 3338.E-mail address: [email protected] (T. Ooi).
Kohsuke Ohmatsu a, Takashi Ooi a,b,⇑a Institute of Transformative Bio-Molecules (WPI-ITbM), and Department of Applied Chemistry, Graduate School of Engineering, Nagoya University, Chikusa, Nagoya 464-8603, Japanb CREST, Japan Science and Technology Agency (JST), Chikusa, Nagoya 464-8603, Japan
a r t i c l e i n f o a b s t r a c t
Article history:Received 26 December 2014Revised 17 February 2015Accepted 17 February 2015Available online 5 March 2015
Keywords:Supramolecular catalysisMetal catalysisChiral ligandAsymmetric synthesisCombinatorial screening
Three strategies for the development of supramolecular chiral ligands for asymmetric metal catalysis areoutlined. The basic ideas, advantages, and examples of each strategy are described.� 2015 Published by Elsevier Ltd. This is an open access article under the CC BY-NC-ND license (http://
creativecommons.org/licenses/by-nc-nd/4.0/).
Contents
Introduction. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2043Supramolecular bidentate chiral ligands by assembly of two monodentate ligands . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2044Assembly of chiral ligands with achiral building blocks. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2045Achiral ligands with chiral subcomponents . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2046Conclusions . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2048Acknowledgements . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2048References and notes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2048
Introduction
Asymmetric metal catalysis has been regarded as one of the mostpowerful tools for the preparation of stereochemically pure chiralorganic molecules.1 Over the last few decades, a myriad of chiralmetal complexes have been elaborated, paving the way for thedevelopment of various catalytic asymmetric transformations.2 Achiral metal catalyst generally comprises a central metal atom anda chiral ligand, and its reactivity and selectivity are heavily depen-dent on the ligand attributes. Despite the deep insight gained intomanifold reaction mechanisms and the advancement in theoreticalchemistry, however, rational design of chiral ligands that can attain
sufficient levels of catalytic efficiency and stereoselectivity in a tar-get transformation remains very difficult. Therefore, a process forthe discovery of an effective chiral ligand for a given asymmetricreaction still involves numerous trial-and-error attempts.
Conventional chiral ligands rely on a covalently constructed,single chiral molecule embedded with coordinative functionalgroups. Their syntheses from each single molecule with the requi-site chirality often require multi-step operation and can be compli-cated. This intrinsic problem hinders the iterative design,evaluation, and structural optimization processes in the identifica-tion of an optimal ligand. Thus, an alternative strategy that allowsthe simple and rapid preparation of structurally diverse chiralligands would be of significant importance.
Meanwhile, nature has evolved a wide variety of enzymes froma relatively small number of building blocks, and a multitude of
http://crossmark.crossref.org/dialog/?doi=10.1016/j.tetlet.2015.02.096&domain=pdfhttp://creativecommons.org/licenses/by-nc-nd/4.0/http://creativecommons.org/licenses/by-nc-nd/4.0/http://dx.doi.org/10.1016/j.tetlet.2015.02.096http://creativecommons.org/licenses/by-nc-nd/4.0/mailto:[email protected]://dx.doi.org/10.1016/j.tetlet.2015.02.096http://www.sciencedirect.com/science/journal/00404039http://www.elsevier.com/locate/tetlet
-
conventional chiral ligand
chiralscaffold L
(a) two monodentate ligands
L = coordinating group
L
L
(b) chiral ligand with achiral subcomponents
L
(c) achiral ligand withchiral subcomponent
L
= non-covalent interaction
Figure 1. Three strategies toward the development of supramolecular chiralligands.
OP
OO
N1
Ph
Ph
Ph
Zn NN
NN
R
2a: R = H
Zn
Rh(acac)(CO) 2
H2/CO
= R
2b
Zn
O O
Zn
1 +
Zn
O O
ZnN N
O OP PRh
H
CO
COO
O OO
**3
Scheme 1. Self-assembly of bidentate chiral ligand through the coordination ofpyridine nitrogens to zinc porphyrins.
Ph
3H2/CO
toluene, 25 °CPh
CHO
33% ee
[6% ee with HRh(1·2a )2(CO)2]
Scheme 2. Hydroformylation of styrene using a supramolecular chiral rhodiumcomplex.
NN
NN
Zn
Ph
PhPh
O P
O
O
N P
[Rh]
4
O
O
N PPh
Ph
4aO
O=
N P=
Figure 2. Chiral rhodium complex with non-symmetric bidentate ligand.
2044 K. Ohmatsu, T. Ooi / Tetrahedron Letters 56 (2015) 2043–2048
enzyme libraries have proven to be a versatile implement forasymmetric catalysis.3,4 Inspired by such a biological system,supramolecular chiral ligands, which are assembled from smallmolecules through non-covalent interactions and behave as sin-gle-molecule chiral ligands, have recently attracted increasingattention. These supramolecular chiral ligands have several advan-tages over the conventional counterparts: (i) the synthesis of eachsmall component is in principle much easier than that of the morecomplex conventional chiral ligands, (ii) the diverse combinationsof ligand components enable facile and rapid access to sufficientlylarge libraries, (iii) the steric and electronic properties of theligands can be tuned easily by changing each component, and(iv) the screening of the ligands can be considerably streamlinedthrough combinatorial methods.5–7
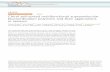
Several reviews have recently been published, covering varioustopics related to supramolecular catalysis.8,9 In the present digest,we will focus on selected reports on the three approaches tosupramolecular chiral ligands (Fig. 1). The first approach is thegeneration of self-assembled bidentate ligands from two mon-odentate ligand components (Fig. 1a). The second is based on theassembly of chiral ligands with achiral building blocks to constructligands that are more structurally complex (Fig. 1b). The third uti-lizes the association of achiral ligands with chiral subcomponents(Fig. 1c). Within the scope defined above, the design principlesand synthetic applications of supramolecular chiral ligands aredescribed.
Supramolecular bidentate chiral ligands by assembly of twomonodentate ligands (Fig. 1a)
In the 1970’s, chelating bidentate chiral ligands were found tobe effective for asymmetric hydrogenation,10 and a variety of cata-lysts bearing bidentate chiral ligands have been developed.11 Thepreparation of bidentate chiral ligands through the supramolecularassembly of monodentate ligands is an attractive strategy for read-ily generating efficacious chiral ligands. The first example of such asupramolecular bidentate chiral ligand was introduced by Reekand co-workers.12 Their system featured the assembly of two mon-odentate ligands 1 with bisporphyrin template 2b through thecoordinative interaction between the pyridine nitrogen of 1 andthe zinc center of each porphyrin ring in 2b (Scheme 1).13
Compared to the corresponding monodentate congener, whichwas a complex generated from monodentate ligand 1, porphyrin2a, and Rh(acac)(CO)2, supramolecular catalyst 3 exhibited higherenantioselectivity in the hydroformylation of styrene (Scheme 2).
Although C2-symmetric chiral ligands have proven effective in awide range of metal-catalyzed asymmetric transformations, therehave been reactions that are promoted with higher efficiency andstereocontrol by the catalysts bearing non-symmetric ligandsrather than C2-symmetric ones.11 One of the distinct advantages
of supramolecular bidentate ligands is the facile preparation ofnon-symmetric chiral ligands from two different ligand compo-nents. Reek and co-workers applied the zinc porphyrin–pyridinecoordination system to the design of non-symmetric supramolecu-lar ligands.14 They developed porphyrin-functionalized chiralphosphite ligands and nitrogen-containing phosphine ligands.The simple mixing of these two ligand components with rhodiumsalt in solution led to the spontaneous formation of a complex witha non-symmetric chiral ligand via self-assembly of the comple-mentary functionalities of each component (Fig. 2). This type ofsupramolecular chiral ligand is suitable for the rapid preparationof diverse catalyst libraries. For instance, a library of 48 bidentateligands can be generated from 4 porphyrin phosphites and 12nitrogen-containing phosphines. The power of this strategy wasdemonstrated by achieving the rhodium-catalyzed, highly enan-tioselective hydrogenation of cyclic enamide 5 through the high-throughput screening of the ligand library (Scheme 3).15
Another example of a non-symmetric supramolecular bidentateligand that harnessed zinc–nitrogen interaction was independentlyreported by Takacs et al., who utilized a chiral bisoxazoline (box)framework and Zn(II) metal for the ligand assembly.16 Mixing aracemic mixture of box ligand 7 with Zn(OAc)2 resulted in thepreferential formation of heteroleptic complex (SS,RR)-8;
-
NO
ON
Ph
Ph
BnH
(S,S)-7
+N
O
ON
Ph
Ph
BnH
(R,R)-7
Zn(OAc) 2N
O
ON
Ph
Ph
BnN
O
ON
Ph
Ph
BnZn
(SS,RR)-8
Scheme 4. Selective formation of heteroleptic (box)2 Zn complex from racemicoxazoline.
N
N
NN
NH
H
OHN
NMe
O
A
T
NNH
Piv
OHN
NNH
Piv
OHN
La
Lb
[M] La
Lb[M]+
10
La, Lb = PAr2
Ar1 OP
O
R
R
Scheme 5. Selective formation of heteroleptic bidentate ligand through comple-mentary hydrogen bonding.
AcHN CO2Me
Ph [Rh(11a)(12)]BF4 (0.5 mol%)H2 (10 bar)
CH2Cl2, r.t. AcHN CO2Me
Ph
99% ee
CO2Me
[Rh(11b)(12)]BF4 (0.5 mol%)H2 (10 bar)
CH2Cl2, r.t.99% ee
O
MeO
Me
CO2Me
O
MeO
NH
OP
O
Me
MeO
NH
O
P
Me
Me
11a 11b
S
N OP
O
HNPiv
12
NH
La
O
N NH
La
S
NLb
HNPiv
N LbHN
Piv
120 combinations optimal ligands were identified within 17 experiments
Scheme 6. Identification of the optimal ligand combinations for asymmetrichydrogenations.
HN
Ac 4
H2 (12 bar)iPr2NEt (20 equiv)
CH2Cl2, 25 °C
∗HN
Ac
5up to 94% ee
6
Scheme 3. Asymmetric hydrogenation of N-(3,4-dihydro-2-naphthalenyl)ac-etamide 5.
K. Ohmatsu, T. Ooi / Tetrahedron Letters 56 (2015) 2043–2048 2045
generation of homoleptic (SS,SS)- and (RR,RR)-complexes was notobserved (Scheme 4). Based on this observation, they developednovel non-symmetric chiral ligands of type 9 consisting of two chi-ral phosphites with the pendent box subunits (Fig. 3). This strategyalso suited for the preparation of a combinatorial ligand library,and the screening of the chiral ligands enabled the highly enan-tioselective hydroboration of alkenes and the asymmetric hydro-genation of amino acid precursors.17
In addition to metal-heteroatom coordination, hydrogen-bondinginteraction has been exploited in the assembly of supramolecularligands. Breit and co-workers reported a self-assembly strategy basedon complementary hydrogen bonding similar to that found in an A–Tbase pair analog, which afforded structurally defined non-symmetricbidentate ligands.18 For instance, aminopyridine and isoquinolonepossessing chiral phosphorus ligand moieties, upon treatment witha metal salt, generated a new class of heterobidentate supramolecularchiral complexes 10 (Scheme 5). This system was successfully appliedto rhodium-catalyzed asymmetric hydrogenation and palladium-cat-alyzed asymmetric allylic amination.19
Based on the exclusive formation of heterobidentatesupramolecular complexes 10, Breit and co-workers subsequentlydeveloped an efficient method for combinatorial high-throughputscreening of catalyst libraries. For the rhodium-catalyzed asym-metric hydrogenation, the iterative deconvolution strategy allowedthem to identify the optimal catalyst from a library of 120 hetero-bidentate combinations within 17 experiments.20 The classicalindividual examination of all 120 self-assembled catalysts con-firmed that the catalyst identified through the iterative deconvolu-tion process was indeed the best of the 120 catalysts. By using thismethod, highly selective rhodium catalysts could be found for theasymmetric hydrogenation of several alkene substrates (Scheme 6)and the number of experiments required to identify the optimalcatalyst was dramatically reduced.
NO
ON
Ph
Ph
NO
ON
Ph
Ph
Zn
OP
O
O
PhPh
PhPh
O O
Me Me
OPO
O
PhPh
PhPh
O O
Me Me
Figure 3. Self-assembled non-symmetric chiral diphosphate 9.
Other families of chiral ligands based on the judicious use ofhydrogen-bonding donor/acceptor functionalities, such as pep-tides,21 ureas,22 amides,23 sulfonamides,24 and phosphoramidites,25
were also developed. Furthermore, other types of non-covalentinteractions, such as acid-base interactions26 or cation-crown ethercomplexations,27 were found to be suitable for the preparation ofself-assembled ligands. These approaches significantly contributedto the successful application of supramolecular bidentate chiralligands toward highly efficient and enantioselective hydrogenationwith a broad substrate scope.
Assembly of chiral ligands with achiral building blocks (Fig. 1b)
The structural modification of privileged chiral ligands, espe-cially through the introduction of various substituents, representsone of the most commonly employed yet reliable tactics for thedevelopment of metal-catalyzed asymmetric transformations. Forexample, the 2,20-disubstituted 1,10-binaphthyl skeleton has beenextensively explored as a key structure in chiral catalysts, and theintroduction and modification of substituents at the 3,30-positions
-
OP
ONMe2
N
N
Ph
Ph
Ph
Ph
Zn NN
NN
Zn=O
PO
NMe2
N
N
Zn
Zn
Scheme 7. Supramolecular approach for the modification of chiralphosphoramidite.
OHOH
P
P
O
O
O
O
O
O
Me
Me
Me
Me
14
(HO)2B
CF3
CF3
(1)
(
H2O (100 mol%)CH2Cl2/THF, r.t.
(2) 4Å MS, 99%
93%2%0%
(endo/exo = 6/94)(endo/exo = 12/88)
Scheme 9. Anomalous endo-selective asymmetric Diels–Alder reaction.
formation of the conformationally flexible supramolecular com-plex. Borane 16 acted as a bulky functional group to form a narrowand deep cavity around the central boron atom. Moreover, thestrong electron-accepting nature of 16 increased the Lewis acidityof the boron atom through the conjugated bonds, thus takingadvantage of Lewis acid-assisted chiral Lewis acid (LLA) catalysts.31
With supramolecular complex 13, they accomplished highlyenantioselective Diels–Alder reactions with anomalous endo/exoselectivities (Scheme 9).
Achiral ligands with chiral subcomponents (Fig. 1c)
The underlying idea in the design of supramolecular chiralligands is to divide the structurally complex chiral ligands intosimpler components, and the way of dividing them largely definesthe characteristics of the respective design principles. In thisregard, a new approach based on the division of chiral ligands intoachiral ligands and chiral subcomponents has recently been intro-duced. This is useful, especially for the preparation of phosphineligands, because the easily oxidizable nature of phosphines gener-ally makes their syntheses labor-intensive. In addition, simpli-fication of the phosphine structure itself and its modificationwith the aid of readily available chiral molecules would allowthe rapid and systematic preparation of structurally diverse arraysof supramolecular ligands, thereby significantly facilitating the dis-covery of effective catalysts.
The feasibility of this idea was demonstrated by the Reek group.Upon combining an achiral phosphine ligand bearing a pyridinemoiety with a chiral Zn(II) porphyrin (Fig. 4), they observed asym-metric induction in the palladium-catalyzed allylic alkylation of1,3-diphenylallyl acetate with dimethyl malonate, albeit with aninsufficient level (up to 10% ee).28
Reek and co-workers subsequently reported the utilization of anachiral diphosphine ligand that binds with carboxylate-containingchiral molecules (Fig. 5).32 NMR experiments showed that, uponbinding of the chiral molecules in the pocket of the free diphos-phine ligand, the phosphorus atoms and the indole and amideNHs of the ligand became diastereotopic, confirming that chiroge-nesis was induced by chirality transfer from the carboxylate tothe ligand through supramolecular interactions. The screening ofmany different chiral carboxylate derivatives revealed complexesthat could promote the asymmetric hydrogenation of a variety ofalkenes with excellent enantioselectivities (Scheme 10). They also
Figure 4. Achiral pyridyl based phosphine ligand with chiral Zn(II) porphyrin.
Et
NH HN
Me Me
NH HNO O
PPh2 Ph2P
+
iPr
NH
O
OS
NHR
Figure 5. Achiral diphosphine ligand with chiral acetate.
-
NMe N
O
PPh2NMe
Et
N
O
PPh2
Rh(nbd)HH
HH
AcHN
CH2
CO2Me (1 mol%)
CH2Cl2H2, r.t., 16 h
AcHN CO2Me
93%, 98% eeMe
O
O
R
Scheme 10. Asymmetric hydrogenation using supramolecular Rh complexincorporating chiral carboxylate.
O2N CO2tBu
Me
+
Ph OCO2Me
Pd2(dba) 3(Pd 2.5 mol%)19 (5 mol%)
toluene/H 2O0 °C
PhCO2tBu
Me NO297%, 94% ee
Scheme 13. Enantioselective allylation of a-nitrocarboxylate.
OO
Bn
+
MeO2COMe
CO2tBu
Pd2(dba) 3·CHCl3(Pd 2.5 mol%)20 (5 mol%)
mesitylene/H2O0 °C
MeCO2tBu
O
Bn
O99%, E/Z = >20:1, 94% ee
Ar22P
Me3N
Ar1
Ar120 (Ar1 = 2-Me-4-MeOC6H3, Ar2 = 4-ClC6H4)
OP
O OO
Scheme 14. Asymmetric allylation of benzofuranone with 1,2-disubstituted allyliccarbonate.
K. Ohmatsu, T. Ooi / Tetrahedron Letters 56 (2015) 2043–2048 2047
adopted the iterative deconvolution screening strategy for the iden-tification of the optimal catalyst. The library of 24 chiral carboxy-lates was divided into two subgroups, and was subjected toasymmetric hydrogenation to identify the superior subgroup. Byfollowing the superior set of carboxylates and further dividing theminto two smaller subgroups, the best chiral carboxylate was identi-fied through 9 experiments instead of 24.
From a similar standpoint, van Leeuwen et al. developedsupramolecular chiral ligand 17, which was assembled from anachiral ligand and a chiral diol–titanium complex (Scheme 11).33
The Schiff base-phosphine hybrid ligands were treated with vari-ous chiral diols in the presence of Ti(OiPr)4 to generate a libraryof chiral bidentate ligands. The synthetic value of this library wasassessed in the rhodium-catalyzed asymmetric hydrogenation ofolefins, and high enantioselectivities were obtained through theappropriate choice of phosphine and diol.
The electrostatic interaction between positively and negativelycharged ions also offers a unique means for the design ofsupramolecular chiral ligands. In conjunction with our research
Ti(O iPr)4
OO
HO OHPhPh
PhPh
Me Me
OO
Me Me
OTi
OPhPh
PhPh
iPrO O iPr
+
Ph2P
N
HO
MeO
O
O
N
Ph2P
TiO O
N
OO
OO MeMe
Ph
Ph
PhPh
Ph2P
Me
Me
17
Scheme 11. The supramolecular ligand based on Ti, achiral ligand, and TADDOL.
Me2N
(1) n-BuLi(2) Ar2PCl
Et2O
Me2N
Ar2P CH3CN
MeOTf Me3N
Ar2P
TfO
18
O
OH Ar2P
Me3N
β-Naph
β-Naph
(1) amberlyst A-26 (OH—)
MeOH
(2) (R)-BINOLderivative
19 (Ar = 4-ClC6H4, R = tBu)
R R R
R
Scheme 12. Preparation of ion-paired chiral ligands.
on organic ion-pair catalysis, we developed novel ligands assem-bled through the ion-pairing between readily preparable achiralphosphines incorporating ammonium ion moieties and chiralanions. The simple two-step transformations starting from com-mercially available benzylic amines can give rise to ammoniumphosphines 18. The subsequent ion-exchange process with BINOLderivatives furnished supramolecular chiral ligands 19, termedion-paired chiral ligands (Scheme 12).34 The vast potential of thisnew class of chiral ligands was clearly demonstrated through thedifficult-to-control palladium-catalyzed asymmetric allylations ofprochiral carbon nucleophiles. In particular, excellent enantiose-lectivity was attained in the allylic alkylation of a wide range ofa-nitrocarboxylates by taking advantage of the structural modu-larity of the ion-paired ligands (Scheme 13).
The ion-paired ligand served as a powerful tool for developinghitherto difficult asymmetric bond-forming reactions by fullyexploiting a multitude of combinations of ammonium-phosphinesand easily accessible chiral anions. For instance, the employmentof appropriate chiral ligand 20 featuring a BINOL-derived chiralphosphate ion35,36 allowed us to successfully develop an unprece-dented highly E-selective and enantioselective allylation of 3-sub-stituted benzofuran-2(3H)-ones with 1,2-disubstituted allyliccarbonates (Scheme 14).37 The origin of the high E-selectivity could
SO
Bn
+MeO2CO CO2tBu
Pd2(dba) 3·CHCl3(Pd 2.5 mol%)
21 (total 5 mol%)22 (total 5 mol%)
K2CO3 (10 mol%)toluene/H 2O
0 °C
up to 99%up to 94% ee
Ar22P
Me3N
Ar1
Ar1
22
OP
O OHO
SO
Bn CO2tBu
RR
21
HSO4
RS
144 combinations
Scheme 15. Iterative deconvolution screening for asymmetric allylation ofbenzylbenzothiophenone.
-
2048 K. Ohmatsu, T. Ooi / Tetrahedron Letters 56 (2015) 2043–2048
be ascribed to the ability of ion-paired ligand 20 to control eitherthe distribution of syn and anti p-allyl palladium complexes orthe relative rate of their bond formations.
Our group subsequently established a method for the in situgeneration of ion-paired chiral ligands from hydrogen sulfate saltsof ammonium phosphines and chiral Brønsted acids under phase-transfer conditions. This technique was effectively utilized for theiterative deconvolution screening of ion-paired chiral ligands inasymmetric palladium catalysis. After only 16 experiments, thebest of 144 ligands could be identified to achieve the first highlyenantioselective allylation of benzothiophenones (Scheme 15).38
Conclusions
Although miscellaneous chiral supramolecular ligands havebeen developed, it would be fair to state that this field is still inits infancy and there remains much room for further development.First, the synthesis of each component for self-assembly is ratherdifficult in many cases. Second, the search for non-covalent inter-actions suitable for creating supramolecular chiral ligands is animportant task. Finally, most of supramolecular strategies areapplied toward well-established asymmetric transformations, suchas asymmetric hydrogenation of alkenes. Therefore, using self-assembled chiral ligands to challenge hitherto difficult asymmetricbond-forming reactions is essential to expand the frontiers ofsupramolecular catalysis.
Acknowledgements
This work was supported by CREST from JST, Grants-in-Aid forScientific Research (C) from JSPS, Program for Leading GraduateSchools ‘Integrative Graduate Education and Research Program inGreen Natural Sciences’ in the Nagoya University, the TatematsuFoundation, and the Uehara Memorial Foundation.
References and notes
1. Comprehensive Asymmetric Catalysis, and Supplements 1 and 2; Jacobsen, E. N.,Pfaltz, A., Yamamoto, H., Eds.; Springer, 1999, 2004.
2. Yoon, T. P.; Jacobsen, E. N. Science 2003, 299, 1691.3. Groeger, H. In Catalytic Asymmetric Synthesis; Ojima, I., Ed.; John Wiley & Sons:
New York, 2008.4. Selected recent reviews on asymmetric enzymatic catalysis, see: (a) Reetz, M. T.
J. Org. Chem. 2009, 74, 5767; (b) Reetz, M. T. Angew. Chem., Int. Ed. 2011, 50, 138;(c) Müller, M. Adv. Synth. Catal. 2012, 354, 3161.
5. (a) Crabtree, R. H. Chem. Commun. 1999, 1611; (b) Gennari, C.; Piarulli, U. Chem.Rev. 2003, 103, 3071; (c) Jäkel, C.; Paciello, R. Chem. Rev. 2006, 106, 2912.
6. For reviews on mass spectrometry-based high-throughput screening, see: (a)Chen, P. Angew. Chem., Int. Ed. 2003, 42, 2832; (b) Müller, C. A.; Markert, C.;Teichert, A. M.; Pfaltz, A. Chem. Commun. 2009, 1607; (c) Schröder, D. Acc. Chem.Res. 2012, 45, 1521.
7. For selected reports on high-throughput screening, see: (a) Taylor, S. J.;Morken, J. P. Science 1998, 280, 267; (b) Jarvo, E. R.; Evans, C. A.; Copeland, G. T.;Miller, S. J. J. Org. Chem. 2001, 66, 5522; (c) Evans, M. A.; Morken, J. P. J. Am.Chem. Soc. 2002, 124, 9020; (d) Reetz, M. T.; Tielmann, P.; Eipper, A.; Ross, A.;Schlotterbeck, G. Chem. Commun. 2004, 1366; (e) Miller, C. A.; Pfaltz, A. Angew.Chem., Int. Ed. 2008, 47, 3363; (f) Wassenaar, J.; Jansen, E.; van Zeist, W.-J.;Bickelhaupt, F. M.; Siegler, M. A.; Spek, A. L.; Reek, J. N. H. Nat. Chem. 2010, 2,417; (g) Ebner, C.; Müller, C. A.; Markert, C.; Pfaltz, A. J. Am. Chem. Soc. 2011,133, 4710; (h) Lichtor, P. A.; Miller, S. J. ACS Comb. Sci. 2011, 13, 321.
8. Supramolecular Catalysis; van Leeuwen, P. W. N. M., Ed.; Wiley-VCH: Weinheim,2008.
9. (a) Wilkinson, M. J.; van Leeuwen, P. W. N. M.; Reek, J. N. H. Org. Biomol. Chem.2005, 3, 2371; (b) Breit, B. Angew. Chem., Int. Ed. 2005, 44, 6816; (c) Reetz, M. T.Angew. Chem., Int. Ed. 2008, 47, 2556; (d) Meeuwissen, J.; Reek, J. N. H. Nat.Chem. 2010, 2, 615; (e) Carboni, S.; Gennari, C.; Pignataro, L.; Piarulli, U. DaltonTrans. 2011, 40, 4355; (f) Bellini, R.; van der Vlugt, J. I.; Reek, J. N. H. Isr. J. Chem.2012, 52, 613; (g) Kirkorian, K.; Ellis, A.; Twyman, L. J. Chem. Soc. Rev. 2012, 41,6138; (h) Kataev, E. A.; Müller, C. Tetrahedron 2014, 70, 137; (i) Raynal, M.;Ballester, P.; Vidal-Ferran, A.; van Leeuwen, P. W. N. M. Chem. Soc. Rev. 2014, 43,
1660; (j) Raynal, M.; Ballester, P.; Vidal-Ferran, A.; van Leeuwen, P. W. N. M.Chem. Soc. Rev. 2014, 43, 1734.
10. (a) Dang, T.-P.; Kagan, H. B. J. Chem. Soc. D 1971, 481; (b) Kagan, H. B.; Dang, T.-P. J. Am. Chem. Soc. 1972, 94, 6429; (c) Poulin, J.-C.; Dang, T.-P.; Kagan, H. B. J.Organomet. Chem. 1975, 84, 87.
11. Privileged Chiral Ligands and Catalysts; Zhou, Q.-L., Ed.; Wiley-VCH: Weinheim,Germany, 2011.
12. Slagt, V. F.; van Leeuwen, P. W. N. M.; Reek, J. N. H. Chem. Commun. 2003, 2474.13. (a) Walter, C. J.; Anderson, H. L.; Sanders, J. K. M. J. Chem. Soc., Chem. Commun.
1993, 458; (b) Mackay, L. G.; Wylie, R. S.; Sanders, J. K. M. J. Am. Chem. Soc.1994, 116, 3141; (c) Morris, G. A.; Nguyen, S. T.; Hupp, J. T. J. Mol. Catal. A 2001,174, 15; (d) Merlau, M. L.; del Pilar Mejia, M.; Nguyen, S. T.; Hupp, J. T. Angew.Chem., Int. Ed. 2001, 40, 4239.
14. (a) Slagt, V. F.; Röder, M.; Kamer, P. C. J.; van Leeuwen, P. W. N. M.; Reek, J. N. H.J. Am. Chem. Soc. 2004, 126, 4056; (b) Reek, J. N. H.; Röder, M.; Goudriaan, P. E.;Kamer, P. C. J.; van Leeuwen, P. W. N. M.; Slagt, V. F. J. Organomet. Chem. 2005,690, 4505.
15. Jiang, X.-B.; Lefort, L.; Goudriaan, P. E.; de Vries, A. H. M.; van Leeuwen, P. W. N.M.; de Vries, J. G.; Reek, J. N. H. Angew. Chem., Int. Ed. 2006, 45, 1223.
16. Takacs, J. M.; Reddy, D. S.; Moteki, S. A.; Wu, D.; Palencia, H. J. Am. Chem. Soc.2004, 126, 4494.
17. (a) Takacs, J. M.; Chaiseeda, K.; Moteki, S. A.; Reddy, D. S.; Wu, D.; Chandra, K.Pure Appl. Chem. 2006, 78, 501; (b) Moteki, S. A.; Liu, Z.; Ma, J.; Holmes, A. E.;Takacs, J. M. Chem. Commun. 2012, 263; (c) Moteki, S. A.; Takacs, J. M. Angew.Chem., Int. Ed. 2008, 47, 894.
18. Weis, M.; Waloch, C.; Seiche, W.; Breit, B. J. Am. Chem. Soc. 2006, 128, 4188.19. (a) Birkholz, M.-N.; Dubrovina, N. V.; Jiao, H.; Michalik, D.; Holz, J.; Paciello, R.;
Breit, B.; Börner, A. Chem. Eur. J. 2007, 13, 5896; (b) Birkholz, M.-N.; Dubrovina,N. V.; Shuklov, I. A.; Holz, J.; Paciello, R.; Waloch, C.; Breit, B.; Börner, A.Tetrahedron: Asymmetry 2007, 18, 2055.
20. Wieland, J.; Breit, B. Nat. Chem. 2010, 2, 832.21. (a) Laungani, A. C.; Breit, B. Chem. Commun. 2008, 844; (b) Laungani, A. C.;
Slattery, J. M.; Krossing, I.; Breit, B. Chem. Eur. J. 2008, 14, 4488.22. (a) Sandee, A. J.; van der Burg, A. M.; Reek, J. N. H. Chem. Commun. 2007, 864;
(b) Meeuwissen, J.; Kuil, M.; van der Burg, A. M.; Sandee, A. J.; Reek, J. N. H.Chem. Eur. J. 2009, 15, 10272; (c) Meeuwissen, J.; Detz, R.; Sandee, A. J.; deBruin, B.; Siegler, M. A.; Spek, A. L.; Reek, J. N. H. Eur. J. Inorg. Chem. 2010, 2992;(d) Meeuwissen, J.; Sandee, A. J.; de Bruin, B.; Siegler, M. A.; Spek, A. L.; Reek, J.N. H. Organometallics 2010, 29, 2413.
23. Pignataro, L.; Carboni, S.; Civera, M.; Colombo, R.; Piarulli, U.; Gennari, C.Angew. Chem., Int. Ed. 2010, 49, 6633.
24. (a) Patureau, F. W.; Kuil, M.; Sandee, A. J.; Reek, J. N. H. Angew. Chem., Int. Ed.2008, 47, 3180; (b) Patureau, F. W.; de Boer, S.; Kuil, M.; Meeuwissen, J.; Breuil,P.-A. R.; Siegler, M. A.; Spek, A. L.; Sandee, A. J.; de Bruin, B.; Reek, J. N. H. J. Am.Chem. Soc. 2009, 131, 6683.
25. (a) Liu, Y.; Sandoval, C. A.; Yamaguchi, Y.; Zhang, X.; Wang, Z.; Kato, K.; Ding, K.J. Am. Chem. Soc. 2006, 128, 14212; (b) Breuil, P.-A. R.; Patureau, F. W.; Reek, J.N. H. Angew. Chem., Int. Ed. 2009, 48, 2162.
26. Pignataro, L.; Lynikaite, B.; Cvengroš, J.; Marchini, M.; Piarulli, U.; Gennari, C.Eur. J. Org. Chem. 2009, 2539.
27. (a) Hattori, G.; Hori, T.; Miyake, Y.; Nishibayashi, Y. J. Am. Chem. Soc. 2007, 129,12930; (b) Li, Y.; Feng, Y.; He, Y.-M.; Chen, F.; Pan, J.; Fan, Q.-H. Tetrahedron Lett.2008, 49, 2878; (c) Li, Y.; Ma, B.; He, Y.; Zhang, F.; Fan, Q.-H. Chem. Asian J. 2010,5, 2454.
28. Slagt, V. F.; Kaiser, P.; Berkessel, A.; Kuil, M.; Kluwer, A. M.; van Leeuwen, P. W.N. M.; Reek, J. N. H. Eur. J. Inorg. Chem. 2007, 4653.
29. (a) Bellini, R.; Chikkali, S. H.; Berthon-Gelloz, G.; Reek, J. N. H. Angew. Chem., Int.Ed. 2011, 50, 7342; (b) Gadzikwa, T.; Bellini, R.; Dekker, H. L.; Reek, J. N. H. J. Am.Chem. Soc. 2012, 134, 2860.
30. Hatano, M.; Mizuno, T.; Izumiseki, A.; Usami, R.; Asai, T.; Akakura, M.; Ishihara,K. Angew. Chem., Int. Ed. 2011, 50, 12189.
31. (a) Ishihara, K.; Kobayashi, J.; Inanaga, K.; Yamamoto, H. Synlett 2001, 394; (b)Futatsugi, K.; Yamamoto, H. Angew. Chem., Int. Ed. 2005, 44, 1484.
32. Dydio, P.; Rubay, C.; Gadzikwa, T.; Lutz, M.; Reek, J. N. H. J. Am. Chem. Soc. 2011,133, 17176.
33. van Leeuwen, P. W. N. M.; Rivillo, D.; Raynal, M.; Freixa, Z. J. Am. Chem. Soc.2011, 133, 18562.
34. (a) Ohmatsu, K.; Ito, M.; Kunieda, T.; Ooi, T. Nat. Chem. 2012, 4, 473; See also:(b) Ohmatsu, K.; Imagawa, N.; Ooi, T. Nat. Chem. 2014, 6, 47; (c) Ohmatsu, K.;Kawai, S.; Imagawa, N.; Ooi, T. ACS Catal. 2014, 4, 4304.
35. Chiral phosphoric acids have been widely used as efficient catalysts forasymmetric reactions. For reviews, see: (a) Akiyama, T. Chem. Rev. 2007, 107,5744; (b) Terada, M. Chem. Commun. 2008, 4097; (c) Terada, M. Synthesis 2010,1929.
36. Recent reviews on asymmetric catalysis with chiral anions, see: (a) Phipps,R. J.; Hamilton, G. L.; Toste, F. D. Nat. Chem. 2012, 4, 603; (b) Mahlau, M.;List, B. Angew. Chem., Int. Ed. 2013, 52, 518; For a review on asymmetric ion-pairing catalysis, see: (c) Brak, K.; Jacobsen, E. N. Angew. Chem., Int. Ed. 2013,52, 534.
37. (a) Ohmatsu, K.; Ito, M.; Ooi, T. Chem. Commun. 2014, 4554; See also: (b)Ohmatsu, K.; Ito, M.; Kunieda, T.; Ooi, T. J. Am. Chem. Soc. 2013, 135, 590.
38. Ohmatsu, K.; Hara, Y.; Ooi, T. Chem. Sci. 2014, 5, 3645.
http://refhub.elsevier.com/S0040-4039(15)00377-9/h0005http://refhub.elsevier.com/S0040-4039(15)00377-9/h0005http://refhub.elsevier.com/S0040-4039(15)00377-9/h0010http://refhub.elsevier.com/S0040-4039(15)00377-9/h0015http://refhub.elsevier.com/S0040-4039(15)00377-9/h0015http://refhub.elsevier.com/S0040-4039(15)00377-9/h0020http://refhub.elsevier.com/S0040-4039(15)00377-9/h0020http://refhub.elsevier.com/S0040-4039(15)00377-9/h0025http://refhub.elsevier.com/S0040-4039(15)00377-9/h0030http://refhub.elsevier.com/S0040-4039(15)00377-9/h0035http://refhub.elsevier.com/S0040-4039(15)00377-9/h0040http://refhub.elsevier.com/S0040-4039(15)00377-9/h0040http://refhub.elsevier.com/S0040-4039(15)00377-9/h0045http://refhub.elsevier.com/S0040-4039(15)00377-9/h0050http://refhub.elsevier.com/S0040-4039(15)00377-9/h0050http://refhub.elsevier.com/S0040-4039(15)00377-9/h0055http://refhub.elsevier.com/S0040-4039(15)00377-9/h0055http://refhub.elsevier.com/S0040-4039(15)00377-9/h0060http://refhub.elsevier.com/S0040-4039(15)00377-9/h0060http://refhub.elsevier.com/S0040-4039(15)00377-9/h0065http://refhub.elsevier.com/S0040-4039(15)00377-9/h0065http://refhub.elsevier.com/S0040-4039(15)00377-9/h0070http://refhub.elsevier.com/S0040-4039(15)00377-9/h0070http://refhub.elsevier.com/S0040-4039(15)00377-9/h0075http://refhub.elsevier.com/S0040-4039(15)00377-9/h0075http://refhub.elsevier.com/S0040-4039(15)00377-9/h0080http://refhub.elsevier.com/S0040-4039(15)00377-9/h0080http://refhub.elsevier.com/S0040-4039(15)00377-9/h0085http://refhub.elsevier.com/S0040-4039(15)00377-9/h0085http://refhub.elsevier.com/S0040-4039(15)00377-9/h0090http://refhub.elsevier.com/S0040-4039(15)00377-9/h0090http://refhub.elsevier.com/S0040-4039(15)00377-9/h0090http://refhub.elsevier.com/S0040-4039(15)00377-9/h0095http://refhub.elsevier.com/S0040-4039(15)00377-9/h0095http://refhub.elsevier.com/S0040-4039(15)00377-9/h0100http://refhub.elsevier.com/S0040-4039(15)00377-9/h0105http://refhub.elsevier.com/S0040-4039(15)00377-9/h0105http://refhub.elsevier.com/S0040-4039(15)00377-9/h0110http://refhub.elsevier.com/S0040-4039(15)00377-9/h0110http://refhub.elsevier.com/S0040-4039(15)00377-9/h0115http://refhub.elsevier.com/S0040-4039(15)00377-9/h0120http://refhub.elsevier.com/S0040-4039(15)00377-9/h0120http://refhub.elsevier.com/S0040-4039(15)00377-9/h0125http://refhub.elsevier.com/S0040-4039(15)00377-9/h0125http://refhub.elsevier.com/S0040-4039(15)00377-9/h0130http://refhub.elsevier.com/S0040-4039(15)00377-9/h0130http://refhub.elsevier.com/S0040-4039(15)00377-9/h0135http://refhub.elsevier.com/S0040-4039(15)00377-9/h0135http://refhub.elsevier.com/S0040-4039(15)00377-9/h0140http://refhub.elsevier.com/S0040-4039(15)00377-9/h0140http://refhub.elsevier.com/S0040-4039(15)00377-9/h0145http://refhub.elsevier.com/S0040-4039(15)00377-9/h0150http://refhub.elsevier.com/S0040-4039(15)00377-9/h0150http://refhub.elsevier.com/S0040-4039(15)00377-9/h0150http://refhub.elsevier.com/S0040-4039(15)00377-9/h0155http://refhub.elsevier.com/S0040-4039(15)00377-9/h0155http://refhub.elsevier.com/S0040-4039(15)00377-9/h0160http://refhub.elsevier.com/S0040-4039(15)00377-9/h0165http://refhub.elsevier.com/S0040-4039(15)00377-9/h0165http://refhub.elsevier.com/S0040-4039(15)00377-9/h0170http://refhub.elsevier.com/S0040-4039(15)00377-9/h0170http://refhub.elsevier.com/S0040-4039(15)00377-9/h0175http://refhub.elsevier.com/S0040-4039(15)00377-9/h0175http://refhub.elsevier.com/S0040-4039(15)00377-9/h0180http://refhub.elsevier.com/S0040-4039(15)00377-9/h0185http://refhub.elsevier.com/S0040-4039(15)00377-9/h0185http://refhub.elsevier.com/S0040-4039(15)00377-9/h0190http://refhub.elsevier.com/S0040-4039(15)00377-9/h0190http://refhub.elsevier.com/S0040-4039(15)00377-9/h0195http://refhub.elsevier.com/S0040-4039(15)00377-9/h0195http://refhub.elsevier.com/S0040-4039(15)00377-9/h0200http://refhub.elsevier.com/S0040-4039(15)00377-9/h0200http://refhub.elsevier.com/S0040-4039(15)00377-9/h0205http://refhub.elsevier.com/S0040-4039(15)00377-9/h0205http://refhub.elsevier.com/S0040-4039(15)00377-9/h0210http://refhub.elsevier.com/S0040-4039(15)00377-9/h0210http://refhub.elsevier.com/S0040-4039(15)00377-9/h0210http://refhub.elsevier.com/S0040-4039(15)00377-9/h0215http://refhub.elsevier.com/S0040-4039(15)00377-9/h0215http://refhub.elsevier.com/S0040-4039(15)00377-9/h0220http://refhub.elsevier.com/S0040-4039(15)00377-9/h0220http://refhub.elsevier.com/S0040-4039(15)00377-9/h0225http://refhub.elsevier.com/S0040-4039(15)00377-9/h0225http://refhub.elsevier.com/S0040-4039(15)00377-9/h0230http://refhub.elsevier.com/S0040-4039(15)00377-9/h0230http://refhub.elsevier.com/S0040-4039(15)00377-9/h0235http://refhub.elsevier.com/S0040-4039(15)00377-9/h0235http://refhub.elsevier.com/S0040-4039(15)00377-9/h0240http://refhub.elsevier.com/S0040-4039(15)00377-9/h0245http://refhub.elsevier.com/S0040-4039(15)00377-9/h0245http://refhub.elsevier.com/S0040-4039(15)00377-9/h0250http://refhub.elsevier.com/S0040-4039(15)00377-9/h0250http://refhub.elsevier.com/S0040-4039(15)00377-9/h0250http://refhub.elsevier.com/S0040-4039(15)00377-9/h0255http://refhub.elsevier.com/S0040-4039(15)00377-9/h0260http://refhub.elsevier.com/S0040-4039(15)00377-9/h0265http://refhub.elsevier.com/S0040-4039(15)00377-9/h0265http://refhub.elsevier.com/S0040-4039(15)00377-9/h0270http://refhub.elsevier.com/S0040-4039(15)00377-9/h0275http://refhub.elsevier.com/S0040-4039(15)00377-9/h0275http://refhub.elsevier.com/S0040-4039(15)00377-9/h0280http://refhub.elsevier.com/S0040-4039(15)00377-9/h0280http://refhub.elsevier.com/S0040-4039(15)00377-9/h0285http://refhub.elsevier.com/S0040-4039(15)00377-9/h0285http://refhub.elsevier.com/S0040-4039(15)00377-9/h0290http://refhub.elsevier.com/S0040-4039(15)00377-9/h0290http://refhub.elsevier.com/S0040-4039(15)00377-9/h0295http://refhub.elsevier.com/S0040-4039(15)00377-9/h0295http://refhub.elsevier.com/S0040-4039(15)00377-9/h0300http://refhub.elsevier.com/S0040-4039(15)00377-9/h0300http://refhub.elsevier.com/S0040-4039(15)00377-9/h0300http://refhub.elsevier.com/S0040-4039(15)00377-9/h0305http://refhub.elsevier.com/S0040-4039(15)00377-9/h0305http://refhub.elsevier.com/S0040-4039(15)00377-9/h0310http://refhub.elsevier.com/S0040-4039(15)00377-9/h0310http://refhub.elsevier.com/S0040-4039(15)00377-9/h0315http://refhub.elsevier.com/S0040-4039(15)00377-9/h0315http://refhub.elsevier.com/S0040-4039(15)00377-9/h0320http://refhub.elsevier.com/S0040-4039(15)00377-9/h0320http://refhub.elsevier.com/S0040-4039(15)00377-9/h0325http://refhub.elsevier.com/S0040-4039(15)00377-9/h0325http://refhub.elsevier.com/S0040-4039(15)00377-9/h0330http://refhub.elsevier.com/S0040-4039(15)00377-9/h0330http://refhub.elsevier.com/S0040-4039(15)00377-9/h0335http://refhub.elsevier.com/S0040-4039(15)00377-9/h0335http://refhub.elsevier.com/S0040-4039(15)00377-9/h0340http://refhub.elsevier.com/S0040-4039(15)00377-9/h0340http://refhub.elsevier.com/S0040-4039(15)00377-9/h0345http://refhub.elsevier.com/S0040-4039(15)00377-9/h0345http://refhub.elsevier.com/S0040-4039(15)00377-9/h0350http://refhub.elsevier.com/S0040-4039(15)00377-9/h0350http://refhub.elsevier.com/S0040-4039(15)00377-9/h0355http://refhub.elsevier.com/S0040-4039(15)00377-9/h0360http://refhub.elsevier.com/S0040-4039(15)00377-9/h0360http://refhub.elsevier.com/S0040-4039(15)00377-9/h0365http://refhub.elsevier.com/S0040-4039(15)00377-9/h0365http://refhub.elsevier.com/S0040-4039(15)00377-9/h0370http://refhub.elsevier.com/S0040-4039(15)00377-9/h0370http://refhub.elsevier.com/S0040-4039(15)00377-9/h0375http://refhub.elsevier.com/S0040-4039(15)00377-9/h0380http://refhub.elsevier.com/S0040-4039(15)00377-9/h0380http://refhub.elsevier.com/S0040-4039(15)00377-9/h0385http://refhub.elsevier.com/S0040-4039(15)00377-9/h0385http://refhub.elsevier.com/S0040-4039(15)00377-9/h0390http://refhub.elsevier.com/S0040-4039(15)00377-9/h0390http://refhub.elsevier.com/S0040-4039(15)00377-9/h0390http://refhub.elsevier.com/S0040-4039(15)00377-9/h0395http://refhub.elsevier.com/S0040-4039(15)00377-9/h0400http://refhub.elsevier.com/S0040-4039(15)00377-9/h0400http://refhub.elsevier.com/S0040-4039(15)00377-9/h0405http://refhub.elsevier.com/S0040-4039(15)00377-9/h0405http://refhub.elsevier.com/S0040-4039(15)00377-9/h0410http://refhub.elsevier.com/S0040-4039(15)00377-9/h0410http://refhub.elsevier.com/S0040-4039(15)00377-9/h0415http://refhub.elsevier.com/S0040-4039(15)00377-9/h0415http://refhub.elsevier.com/S0040-4039(15)00377-9/h0415http://refhub.elsevier.com/S0040-4039(15)00377-9/h0420http://refhub.elsevier.com/S0040-4039(15)00377-9/h0425http://refhub.elsevier.com/S0040-4039(15)00377-9/h0425http://refhub.elsevier.com/S0040-4039(15)00377-9/h0430
Design of supramolecular chiral ligands for asymmetric metal catalysisIntroductionSupramolecular bidentate chiral ligands by assemAssembly of chiral ligands with achiral buildingAchiral ligands with chiral subcomponents (Fig. ConclusionsAcknowledgementsReferences and notes
Related Documents