-
8/13/2019 Clase 2 Chemistry of Cell .ppt
1/73
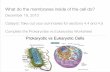
Basic Chemistry
MatterElements.
AtomsCompounds
Molecules
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
2/73
A. Matter
Material that takes up space.1. Elements
Pure chemical substances composed of atoms.
Examples?
How many elements exist?
How many of these elements are essential to life?
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
3/73
Periodic Table of Elements
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
4/73
2. AtomThe smallest piece of an element that retains the
characteristics of that element.
Composed of 3 subatomic particles:
Protons
Neutrons
Electrons
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
5/73
Characteristics of Subatomic Particles
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
6/73
Model of an Atom
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
7/73
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
8/73
Atomic symbol
Atomic number
Atomic mass (weight).
ATOMS
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
9/73
CHEMICAL REACTIONS
Electron shells
Valence (outer)
shellOctet rule
Ionic bonds
Covalent bonds
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
10/73
Atoms will strive to fill their
outer (valence) orbitals
If an atom has 1 valence e-,
its happy to give it awayExample: Sodium
1 valence e-
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
11/73
Atoms will strive to fill their
outer (valence) orbitals
ANDIf an atom has 7
valence e-, its happy to
pick up one
Ex. Chlorine
7 valence e-
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
12/73
Ionic Bonds
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
13/73
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
14/73
Ions can have important biological
functions.
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
15/73
Covalent Bonding
Carbon (C)4
CC H
Hydrogen (H)1
CCH
H
H
H
Methane CH4
Each pair of electrons
represents the formation
of a covalent bond.
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
16/73
Aside from single covalent bonds, double,
or triple covalent bonds can form.
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
17/73
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
18/73
Review
Ionic bonds occur when e- are shared in a+/- arrangement
Covalent bonds exist when one atom shares1 or more e- with another atom.
One covalent bond means 2 e- are involved.Always involves a pair of e-
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
19/73
Oxidation/Reduction
Oxidation----loss of electron
Reduction----Gain of electron
Examples?
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
20/73
Water is the most abundant molecule inliving things.
Water has special traits that make itimportant to life.
Water and Life
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
21/73
Water molecules: 2-H covalently bonded
to 1-O. Covalent=shared e-
. But issharing equal?
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
22/73
Chemistry of Water
Amazing stuff!!!
Should not be a liquidat room temp
Highly Polar
molecule.
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
23/73
WATER
Hydrogen bonds
Polarity
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
24/73
Hydrogen Bonding
Positively charged hydrogen end
Negatively charged oxygen end Hydrogen Bonds
Covalent bonds between hydrogen and oxygen
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
25/73
Characteristics of water:
1. liquid at room temperature2. universal solvent for polar molecules
3. water molecules are cohesive
4. water molecules are adhesive5. temperature of water changes slowly
6. absorbs heat upon vaporization
7. releases heat upon freezing
8. frozen water is less dense
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
26/73
B. The Importance of Water
1. Properties
Cohesion- the attraction of water moleculesfor each other.
Adhesion- the attraction of water moleculesfor other compounds.
High heat capacitytakes a great deal ofheat to raise the temperature of water.
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
27/73
High heat of vaporization- a lot of heat isrequired to evaporate water.
Exists as solid, liquid or gas - solid (ice) is lessdense than liquid.
2. Solutions
A solution is a mixture of one or more solutesdissolvedin a solvent.
If solvent is water, then it is an aqueous solution.
Water is a strong solvent because it separates charged
atoms or molecules.
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
28/73
Water as a
SolventHow itWorks
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
29/73
Dissociation of Water Molecules
Water dissociates and releases hydrogen ions(H+) and hydroxide ions (OH-).
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
30/73
ACIDS AND BASESAcids- release hydrogen ions(H+) in solution.HCl H+ + Cl-
Bases- take up hydrogen ions(H+) or give off hydroxide ions
(OH-) in solution.NaOH Na+ + OH-
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
31/73
pHa Measure of Acidity
If ph7, base
7=neutral
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
32/73
Buffers --substances thathelp to resist change in pH.
Why important?
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
33/73
WaterFinal Notes
Hydrophilic--polar
Hydrophobicnon-polar
Amphipathic---containhydrophilic and hydrophobicparts
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
34/73
Organic molecules are found in livingthings.
The chemistry of carbon accounts forthe chemistry of organic molecules.
Macromolecules (Polymers)
Organic Moleculescarbon based
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
35/73
Organic Molecules
organic monomer
polymer
M l l ( l )
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
36/73
Macromolecules (polymers)and monomers.
Polymer Monomer
carbohydrate monosaccharidesprotein amino acid
nucleic acid nucleotide
D h d i S h i
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
37/73
Dehydration Synthesis orCondensation Reactions
vsHydrolysis
A BOH H+ + HOH
or
H2O
A B
covalent bonds
If the reaction is carried out multiple times you create a polymer!
+ H2O molecules
Condensation
Hydrolysis
Dehydration (condensation) builds. Hydrolysis breaks down.
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
38/73
What do they do?
Structure
Act as enzymesto speed reactionsServe as carriers
Act as antibodies
Transporters and channels
Proteins
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
39/73
Proteins --polymers of amino acids.
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
40/73
Peptide Bonds
Proteins--Levels of Organization
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
41/73
Proteins--Levels of Organization.
TERTIARY (3) STRUCTURE 3D
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
42/73
TERTIARY (3) STRUCTURE3D 1 structure and
environment
FactorsR-groups
Hydrogen bonding
Hydrophobic interactions
Ionic (salt) bridges
Disulfide bonds
Native vs Denatured
REMEMBER: APROTEINS FUNCTIONIS A RESULT OF ITSFORM!!
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
43/73
Catalyst?
Speeds up reaction
ProteinsThe Model:
EnzymesBiological Catalysts
E+S ES E+P
E=Enzyme
S=Substrate
P=Product
Enzymes Lower Energy of
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
44/73
Enzymes Lower Energy ofActivation
Enzymes-- lower the energy barrier
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
45/73
EA
barrier
Reactants
1 Products 2
En
zyme
Figure 5.5A
Enzymes lower the energy barrier
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
46/73
Enzyme-Substrate Complexes
Every reaction in a cell requires a specificenzyme.
Enzymes are named for their substrates:
Substrate EnzymeLipid Lipase
Urea Urease
Maltose MaltaseRibonucleic acid Ribonuclease
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
47/73
Cell Chemistry
Enzymesinvolved in almost allchemical reactions.
AnabolismThe building
reactions. EX. Protein synthesis
CatabolismThe breakdown
reactions. EX. Protein digestionMetabolismsum of all reactions
E C f t
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
48/73
Enzyme Cofactors
Cofactors---may be necessary forsome enzymes to carry out theirfunctions. Two types:
Metal ions. EX copper (Cu+2) or iron(Fe+2)
CoenzymesOrganic molecules,must be present for other enzymesto function. EX vitamins
L l f P t i St t
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
49/73
Levels of Protein StructureQuaternary (4) Structure
Multiple chains:interactions of morethan one polypeptide
chain to form thecomplete, functionalprotein. Ex.Hemoglobin andantibodies
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
50/73
Generally, C:H:O in a 1:2:1 ratio(CH2O)n
Functions
quick energy and short-term energystorage. EX glycogen, starch
Structure. EX. Cellulose
Polymer=polysaccharides EX. Starch Monomers= monosaccharides EX.
glucose
Carbohydrates
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
51/73
NOMENCLATURE
Based on number of carbons
3C==triose
4C==tetrose
5C==pentose, etc
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
52/73
Structure of Glucose
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
53/73
Disaccharides
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
54/73
Polymerization of Carbohydrates
O S CC S
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
55/73
POLYSACCHARIDES
STARCH
GLYCOGEN
CELLULOSE
LIPIDS
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
56/73
LIPIDS
NON-POLAR BIOLOGICAL MOLECULESFUNCTIONS
Energy storage: Fats and oils.
Waterproofing: Waxes and oils Insulation: Fat layers (blubber)
Cushioning: Fat layers (soles of your feet)
Regulating metabolism: Steroids
Component of cell membranes: Phospholipids
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
57/73
Lipid structure (Triglyceride)
Glycerol backbone
Fatty acid tails
Fats vs oils X3
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
58/73
Triglyceride formation
H
H-COH
H-COH
H-COH
H
GLYCEROL
O H H H H H
HO-C-C-C-C-C-C-H
H H H H H
O H H H H H
HO-C-C-C-C-C-C-H
H H H H H
O H H H H H
HO-C-C-C-C-C-C-HH H H H H
FATTY ACIDS
H
H-CO
H-CO
H-CO
H
O H H H H H
-C-C-C-C-C-C-H
H H H H HO H H H H H
-C-C-C-C-C-C-H
H H H H H
O H H H H H
-C-C-C-C-C-C-H
H H H H H
+ H2
0
+ 3 H20TRIGLYCERIDE
What type of reaction forms a triglyceride? Dehydration Synthesis
+ H20
+ H20
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
59/73
Saturated vs. Unsaturated Fats
Saturated vs Unsaturated Fats
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
60/73
Saturated vs. Unsaturated Fats
F tt A id i t
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
61/73
Fatty Acids: cis vs. trans
-linolenicacid
Trans -linolenic acid
Ph h li id
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
62/73
Phospholipids
The majorstructuralcomponent
of cellmembranes
Ph h li id B h i
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
63/73
Phospholipid Behavior
Phospholipids
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
64/73
Phospholipids
S d E l ifi
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
65/73
Soaps and Emulsifiers
Steroids-Hormones and
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
66/73
Steroids Hormones andStructure
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
67/73
N l tid St t
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
68/73
Nucleotide Structure
DNA RNA
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
69/73
DNA vs RNA
The Double Helix of DNA-
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
70/73
e oub e e oComplementary base pairing
The Central Dogma ATGAGTAACGCGTACTCATTGCGCDNA-gene
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
71/73
Transcription---
Translation---
Replication---
RNA
Protein
DNA
duplication of DNA using DNA as the template
synthesis of RNA using DNA as the template
synthesis of proteins using RNA as the template
TACTCATTGCGC
ATGAGTAACGCGTACTCATTGCGC
ATGAGTAACGCG
TACTCATTGCGC
+
AUGAGUAACGCG
MetSerAsnAla
mRNA-message
Protein--end product
DNA gene
M t ti DNA Ch
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
72/73
Mutation-DNA Change
d h h
-
8/13/2019 Clase 2 Chemistry of Cell .ppt
73/73
Adenosine Triphosphate-ATP
Adenosine triphosphate(ATP) is
the energy currency of cells