Chondroblastoma CHONDROBLASTOMA General Information Benign neoplasm of immature cartilage cell (chondroblast) proliferation Cells resemble chondrocytes/chondroblasts Marked predilection for arising from the epiphysis Usually occurs in skeletally immature patients <1% of osseous neoplasms High propensity for local recurrence Codman Tumor (old historical name for chondroblastoma) Cartilage Containing Giant Cell Tumor o Kolodney 1972 Calcifying Giant Cell Tumor o Ewing 1928 Clinical Data Rare; 1-2% all bone tumors Male predilection (2:1) Children and young adults; 90% 5-25 yrs. old Benign Aggressive Tumor with High Propensity for Local Recurrence Very rare cases that metastasize to the lungs Location: Almost all cases arise from the epiphysis of the bone Epiphysis only 40% of cases Epiphysis and metaphysis 55% of cases Metaphysis only 4%

Chondroblastoma
Sep 26, 2015
1
Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript
Chondroblastoma
CHONDROBLASTOMA
General Information
Benign neoplasm of immature cartilage cell (chondroblast) proliferation
Cells resemble chondrocytes/chondroblasts
Marked predilection for arising from the epiphysis
Usually occurs in skeletally immature patients
Female 1.4:1
Age:
Range 3 - 72 years
95% of cases occur between the ages 5 and 25
Most cases occur in adolescents between 10 and 20 years of age
Sites:
Predilection for distal femur, proximal tibia & humerus
98% located in epiphysis, 30% in knee area
May also occur in calcaneus, talus and temporal bone
Most Common Sites:
Proximal Femur 23%
Distal Femur 20%
Head and Neck 16%
Trochanter 7%
Proximal Tibia 17%
Proximal Humerus 17%
Hands and Feet 10%
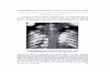
Radiographic Presentation
Presents as a highly defined/well circumscribed geographic oval/round lytic defect
Surrounded by rim of sclerotic bone
Usually in epiphyseal region
Lesion ranges from 3 cm to 6 cm diameter
Usually radiolucent
May have fine trabeculae and irregular calcifications
Calcifications are often better detected with a CT scan but are not uniformly present
Lesions may expand the bone and new periosteal bone may form
Bony end plate, cortex, bone contour are unaffected
Plain X-rays:
Geographic lytic lesion IA/IB margin of sclerosis
Usually Eccentric more often than Central in the bone
Rarely expansile (rarely penetrates the cortex)
Calcified chondroid matrix 30%-50% of cases
Often better detected with a CT Scan
Periosteal Reaction 30-50% of cases
Usually occurs in Adjacent Diaphysis/Metaphysis since epiphysis is intraarticular and not surrounded by periosteum
MRI:
Geographic, well circumscribed lesion in the epiphysis
Intermediate Signal on T1
High signal on T2 mixed with low signal areas (low signal areas proposed to be secondary to lysosomal content of highly cellular areas)
Fluid/Fluid levels demonstrated in tumors that have undergone ABC change (aneurysmal bone cyst change)
Extensive Surrounding edema is common
Joint effusion in 30-50% of cases
CT scan:
Most useful for detecting subtle mineralization that is not apparent on X-rays
Useful for identifying intact periosteum around any expansile soft tissue component that appears as a surrounding thin reactive shell of bone/mineralization (Egg Shell Rim of Calcification). This helps place the tumor in a benign category.
Can help evaluate bony quality, extent of bone and cortical destruction and whether the subchondral plate of bone adjacent to the joint cartilage has been destroyed or is intact.
Bone Scan:
Chondroblastomas demonstrate intense increased uptake on a bone scan
Roll over the images for more information
Gross Pathology
Grossly variable appearance
Grey/yellow/brown and gritty if has interspersed calcifications
Interspersed red areas from hemorrhagic necrosis
May be blue-grey areas from the chondroid matrix
Rim of sclerotic bone is visible in totally resected specimens
Lesion may be fully cystic with solid foci of tumor tissue at periphery
May undergo aneurysmal bone cyst change (ABC change)
Roll over the images for more information
Microscopic Pathology
Variable appearance depending on percentage of cells, necrosis, cartilage matrix formation and ABC change
Hypercellular Tumor; Minimal Pleomorphism; Occasional Mitoses but no Abnormal Mitoses; No Atypia
Chondroid matrix in up to 15% of tumor
ABC component 5-15% of tumors
The tumor is composed of chondroblasts that have a distinct, thick cell membrane. The thick cell membrane gives it a "Chicken Wire Fence Appearance" especially when the cell membranes are calcified. "Chicken Wire Calcifications"
Cytoplasm of chondroblasts is plump, clear, eosinophilic
Nucleus is centrally or eccentrically round/oval with indentations
Coffee Bean Shaped Nucleus
Nucleus exhibits clefts, grooves, invaginations
Cells are closely packed together
Osteoclast-like giant cells are interspersed
Calcification is an important diagnostic sign and deposits itself along cell membranes. This gives a pattern referred to as a "Chicken Wire pattern of Calcification" because the appearance is similar to a chicken wire fence.
Chondroblasts stain positive for S-100
Roll over the images for more information
Biological Behavior
Chondroblastomas are benign aggressive tumors. They grow aggressively and destroy the bone.
Chondroblastomas can destroy the cortex and grow into the soft tissues. They are contained by the periosteum (this differs from a malignant tumor that destroys the cortex)
There are extremely rare cases where chondroblastomas metastasize to the lungs and may not appear for 30 years
Metastases may remain stable or may progress and cause death
Recurrences may occur in the bone or adjacent soft tissue
Rare cases of multifocal chondroblastomas have been documented
Synchronous involvement of several sites
Secondary aneurysmal bone cyst frequently correlated with chondroblastoma
Chondroblastomas have been reported to transform into fibrosarcoma or osteosarcoma years after being treated with radiation.
Treatment
Intralesional curettage resection and bone grafting is the most common treatment. Cement and internal fixation may also be used to fill the defect after removal for selected patients.
High risk of local recurrence after curettage alone
Local adjuvants such as cryosurgery (liquid nitrogen application) may be considered to decrease the risk of local recurrence.
Local recurrence results in further bony destruction
Rarely chondroblastomas that have grown out of control have required amputations for treatment because they have completely destroyed the bone and/or adjacent joint.
In patients who are skeletally immature (still growing) there is always a risk of growth plate failure from the chondroblastoma since it usually grows adjacent to the growth plate and may damage it.
CT Guided Radiofrequency Ablation (Minimally Invasive Approach)
May be indicated for selected small tumors
Mostly performed in specialized centers
Roll over the images for more information
Prognosis
Chondroblastomas are benign aggressive tumors that grow and destroy the bone and joint as it grows.
Most patients are cured with the first surgery
There is a significant risk of local recurrence (up to 30% with an intralesional curettage alone without an additional local adjuvant such as cryosurgery). Microscopic tumor cells can grow back after the tumor is removed.
My preferred method is to perform curettage and cryosurgery whenever feasible in appropriate cases in order to help eradicate microscopic disease and decrease the risk of local recurrence (decrease the risk of the tumor coming back in the bone after surgery)
Radiofrequency has been successful in the treatment of very selected small tumors.
This is a minimally invasive approach
Rare cases of pulmonary metastases have been reported.
Pulmonary metastases may be stable or may progress and cause death
Pulmonary metastases have extremely rarely been reported to develop 30 years after initial treatment.
Related Documents

![CASE REPORT Open Access Chondroblastoma associated with aneurysmal cyst … · 2017. 8. 26. · cuboid bones [3,6,7]. To ourknowledge, chondroblas-toma of the navicular bone has not](https://static.cupdf.com/doc/110x72/60f9c9be29546e1364712a6e/case-report-open-access-chondroblastoma-associated-with-aneurysmal-cyst-2017-8.jpg)