Cell Theory • The cell is the basic unit of _______in living organisms • All organisms are made of ________ cells • All cells ________ from other cells • There are two types of cells: ___________ (small, simple, no nucleus, single chromosome, bacteria) and ___________ (nucleus, “typical” cell, anything other than bacteria) cells tissue organs organ system organism Examples? life 1 or more come prokaryote s eukaryotes

Cell Theory The cell is the basic unit of _______in living organisms All organisms are made of ________ cells All cells ________ from other cells There.
Jan 18, 2016
Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript
-
Cell TheoryThe cell is the basic unit of _______in living organismsAll organisms are made of ________ cellsAll cells ________ from other cellsThere are two types of cells: ___________ (small, simple, no nucleus, single chromosome, bacteria) and ___________ (nucleus, typical cell, anything other than bacteria)cellstissueorgansorgan systemorganismExamples?life1 or morecomeprokaryoteseukaryotes
-
Match The Following Terms
-
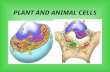
Animal CellGolgi ApparatusCytoplasmMitochondriaNucleusRibosomesCell MembraneNuclear MembraneSmooth ERRough ER
-
Plant CellMitochondriaChloroplastNucleusNucleolusCytoplasmVacuoleRough ERSmooth ERCell WallCell MembraneGolgi ApparatusRibosomesLysosomeNuclear Membrane
-
Energy TransformationLaw of Conservation of Matter: During an ordinary chemical change, there is no detectable change in the _________ of matter
Law of Conservation of Energy: energy cannot be _________ or _________, but can change in form
Carbohydrates, proteins, lipids, and nucleic acids are the organic macromolecules. They are composed of __________ and ___________ with additional elements and are the building blocks of all living things.amountcreateddestroyedcarbonhydrogen
-
Organic MacromoleculesenergyenergyhereditaryDisaccharidepolypeptidesupportRice, bread, potatoesMeat, cheese, beansButter, oilDNA/RNAMeat, fruit, vegetables
-
EcologyMatter and energy change forms by means of food chains and food webs (a series of interconnected food chains).Producers come first in a food chain. Producers are also called __autotrophs___ and include plants and other organisms that make their own food (usually through a process known as __photosynthesis_______________).Consumers (or _heterotrophs__) are shown in the order in which they consume their food or prey. Consumers can obtain their food through __predation___ or __scavenging___.Decomposers are organisms that break down dead organisms and allow nutrients to be recycled. They come last in a food chain.Example of a food chain:
-
EcologyEcology is the study of the interaction among organisms and between organisms and their environment. Levels of ecological organization:The environment is made up of living components (_biotic_ factors) and nonliving components (__abiotic__ factors)Organisms that live closely with other organisms are in a symbiotic relationship.Mutualism a. one organism benefits while the other is harmedCommensalismb. both organisms benefitParasitismc. one organism benefits while the other is neither helped nor harmed
-
Biomeshabitatnichesuccession
-
DNAGenetics is the scientific study of ___heredity__, or the passing of traits from parents to offspring. Traits are passed by means of _genes_, or deoxyribonucleic acid. DNA contains segments called __codons_ which code for certain __proteins___. These proteins control the traits of an organism. More about DNA:DNA makes up the _chromosomes___ that are found in the nucleus of a cell.DNA is a type of nucleic acid.Its shape is a twisted ladder, or a _double helix____.DNA is made up of sugars and phosphates (the side of the ladder) and pairs of nitrogen bases (the steps of the ladder). The bases in DNA are called adenine, thymine, guanine, and cytosine. __A_ always bonds with _T__ and _C__ always bonds with _G__.Any change in the base sequence of DNA is known as a _mutation____. Mutations can lead to a change in the protein that is coded for by the affected gene. This change can lead to certain disorders like sickle-cell anemia.
-
Genetics
-
Mitosis & MeiosisMITOSISProduces _identical_ genetic copies 1 cell copies its DNA and organelles and then splits into_2_ cellsOccurs for the purposes of growth and repair or asexual reproductionHappens in body cellsConsists of Prophase, Metaphase, Anaphase, Telophse
MEIOSISProduces _gametes___ or sex cells (sperm and egg) for sexual reproduction1 cell copies its DNA and splits into __4_ cells during two cell divisionsEach of the resulting gametes is __different__ from the starting cell and from each otherThe end cells have the amount of DNA as the starting cellHappens in cells of the testes and ovaries onlyConsists of Prophase I, Metaphase I, Anaphase I, Telophase I, Prophase II,
-
Natural SelectionNatural SelectionIdea first stated by __Charles Darwin_______Survival of the __fittest______Organisms that are best ___suited_____to their environment are more likely to live long enough to produce offspring and pass their traits on to the next generation.
-
EvolutionChange in groups of organisms over a long period of timeEvidence for evolutionary changesFossils (The deeper the fossil, the _older__ it is)Comparative anatomy and the study of _homologous___ structures (Example: human arm, dolphin fin, bat wing, dog foreleg)Comparative _biochemistry__ (The fewer the differences in DNA, the closer the organisms are related)Comparative __embryology__ (Example: all vertebrates have gill slits, tail, and notochord in early development)__direct___ evidence (Example: bacteria can quickly become resistant to antibiotics)
-
LifeCharacteristics of Living ThingsReproduceGrow DevelopNeed food and require energyMade of cell(s)Respond to environmentAs a group, evolve over time Homeostasis is an organisms ability to maintain a _stable internal environment__
-
Photosynthesis and Cell RespirationRespirationProcess of using energy from sugar (glucose) to produce __energy__C6H12O6 +6O2 6CO2 + 6H2O + 38ATPOccurs in ___mitochondria______Occurs in all living organismsATP provides energy to do work in the cellWhen ATP is used, it is converted to __ADP__, releasing energyPhotosynthesisProcess of using energy from the sun to produce __sugar__(glucose)6CO2 + 6H2O + Light Energy C6H12O6 +6O2Occurs in _chloroplasts____of plants and some algae (autotrophs)How are photosynthesis and respiration related?
-
ProteinsProtein SynthesisTranscription_copying___ information from DNA to mRNAmRNA then transported from DNA in the nucleus to a _ribosome__TranslationInformation in mRNA used to construct specific sequence of amino acidsInformation is translated from language of nucleotides to the language of amino acids_tRNA___carries amino acids to ribosomes where they are linked together
-
Cell TransportActive Transport- requires ATP (energy); particles move from a __lower__ to _higher__ concentrationEndocytosis into cellExocytosis- out of cellPassive Transport- does not require ATP (energy); particles move from a _higher_ to _lower__ concentration; works to result in dynamic equilibriumDiffusion- movement of particles down a concentration gradientOsmosis- movement of ___water___ through a semi-permeable membraneFacilitated diffusion- movement of particles down a concentration gradient through the use of a transport protein
-
States of MatterMatter is anything that has __mass____ and takes up __space___.Matter is composed of units known as _elements__. Elements are substances that are made up of only one kind of atom and that cannot be broken down by ordinary means such as heating, cooling, or crushing.A substance made up of two or more elements chemically combined, such as H2O, is known as a __compound___. The states of matter are the characteristic forms of all substances:__solid__-matter that has a definite shape and volume__liquid___-matter that has a definite volume, but takes the shape of its container_gas__-matter that has no definite shape and no definite volume; it fills its container
-
ChemistryA __mixture__ is a combination of two or more kinds of matter that can be separated; there are 2 kinds of mixtures:Homogeneous- a mixture that is the same throughoutHeterogeneous-a mixture with an unequal composition throughoutA homogeneous mixture of two or more substances is known as a ___solution__. It has two parts: 1. the _solvent__, which is the substance that is doing the dissolving, and 2. the __solute__, the substance that is being dissolvedPhysical properties are characteristics of a substance that can be observed with the senses. Examples include: hardness, colorChemical properties are characteristics of a substance that describe how the substance reacts with other substances. Examples: oxidation
-
AtomsThe _atom_ is the smallest unit of an element; all matter is composed of atoms
-
AtomsAn atom that has gained or lost an electron is known as a __ion__. These have positive or negative charges. __isotopes___ are atoms of the same element with different numbers of neutrons.The Periodic Table Atomic number identifies the element and tells the number of _protons___ in an element.Atomic mass is the average mass of one atom of that element in atomic mass units.Mass number is the number of protons plus neutrons in the nucleus.
-
Chemistry Matching
-
BondingMost atoms need 8 valence (outer) electrons to become stable. This is known as the __octet__ rule.Ionic bonds are chemical bonds that result from the _transfer__ of an electron.Ionic compounds contain an ionic bond; these compounds are electrically charged
Covalent bonds are chemical bonds that results from the __sharing__ of an electron.Covalent compounds contain a covalent bond and are also known as _molecules__.
-
Physical and Chemical ChangesA physical change occurs when there is a change in the __physical___ property of a substance. This type of change does not change the original substance into something else. Includes:DissolvingMeltingFreezingExamplesA chemical change occurs when there is a _rearrangement__ of atoms that makes or breaks chemical bonds. This forms a different substance. Includes:CombustionFermentationElectrolysisRusting/ oxidationTarnishingExamples
-
Chemical ReactionsA chemical reaction is the process by which the atoms of one or more substances are __changed__ to form different substances.Reactants yield product(s).A__catalyst__ is a substance that can speed up the rate of a chemical reaction by lowering the activation energy but is not itself consumed in the reaction.A chemical equation is a statement using chemical formulas to describe the identities and relative amounts of the reactants and products involved in the chemical reaction.Remember the Law of Conservation of Matter: matter is neither __created__ nor __destroyed___. Chemical equations should be balanced.
-
Chemical ReactionChemical reactions always involve a change in energy. But remember that energy is neither created nor destroyed. Chemical reactions in which energy is absorbed are _endergonic____. Energy is required for the reaction to occur.Chemical reactions in which energy is released are __exergonic___. The energy that is released was originally stored in the chemical bonds of the reactants. Often the heat give off causes the product(s) to feel hot.
-
SolutionsSolutions are ____homogenous___ mixtures of two or more substances.Solutions are composed of two parts:The __solvent____ does the dissolving.The ___solute___ is dissolved.Solubility is the amount of solute that will __dissolve__ in a given amount of solvent at a given temperature
-
SolutionsThere are three types of solutions:In an _unsaturated__ solution, more solute can be added at a certain temperature and still dissolve.In a _saturated__ solution, the maximum amount of solute at a given temperature is able to dissolve (if more solute is added, it will settle out).In a _supersaturated__ solution, more solute has been added than the solvent can typically hold at a certain temperature because the solution was heated up and then allowed to cool slowly. A supersaturated solution contains more solute than it would if the dissolved solute were in equilibrium with the undissolved solute.
-
Acids and BasesHigh concentrations of H+ ions indicate an __acidic__ solution.High concentrations of OH- ions indicated a _basic_ (or alkaline) solution.Equal concentrations of H+ and OH- ions in the same solution indicates a _neutral__ solution.pH is the measure of hydronium ion concentration in a solution.
-
Acids and BasesAcid rain is rain that is more acidic than normal rain due to the addition of _nitric and sulfuric acid___. Acid rain has a pH less than _5.5_. The precursors of acid rain formation can result from both natural sources (such as volcanoes and decaying vegetation) and man-made sources (primarily emissions of sulfur dioxide and nitrogen oxide resulting from the burning of fossil fuels)sourbitterBelow 7Above 7
Related Documents