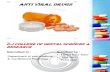
No Penggolonga n Antiretrovi ral & Contoh Nama Obat Farmakokinetik Farmakodinamik & Struktur Kimia Spektrum Antiretrov iral & Indikasi Efek Samping & Kontraindikasi Interaksi Obat 1. NRTI (Nucleoside Reverse Transcripta se Inhibitor) Zidovudin A: Rapidly absorbed. T max is 0.5 to 1.5 h. Oral bioavailability is 64% D: It is extensively distributed. Binding to plasma protein is low, less than 38%. Apparent Vd is 1.6 L/kg M: Hepatic metabolism. Major metabolites are D- glucopyranuronosy lthymidine (GZDV) and 3′-amino-3′- deoxythymidine E: Primarily eliminated by hepatic metabolism. Half- life is 0.5 to 3 Gugus AZT 5’-monofosfat bergabung pada ujung 3’ rantai DNA virus & menghambat enzim reverse transcriptase (RT) Spektrum: HIV type 1 & 2 Indikasi: infeksi HIV dalam kombinasi dengan anti-HIV lainnya (lamivudin , abakavir), mencegah penularan HIV dari ibu ke janin Efek samping: anemia, neutropenia, sakit kepala, mual Kontraindikasi: Potentially life- threatening hypersensitivit y to any component. Adriamycin, amphotericin B, dapsone, flucytosine, interferon, pentamidine, vinblastine, vincristine May increase risk of toxicity, including nephrotoxicity, cytotoxicity, or hematologic toxicity. Atovaquone, fluconazole, methadone, probenecid, valproic acid May increase serum concentration and potential toxicity of zidovudine. Doxorubicin May antagonize the effect of zidovudine. Avoid use. Experimental nucleoside analogs May affect RBC/WBC counts or function and may increase potential for hematologic toxicity. Ganciclovir Life-threatening hematologic toxicity may occur. Nelfinavir, ribavirin, rifamycin, ritonavir, stavudine May decrease zidovudine serum concentrations, reducing the therapeutic effect.

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript
NoPenggolongan Antiretroviral & Contoh Nama ObatFarmakokinetikFarmakodinamik & Struktur KimiaSpektrum Antiretroviral & IndikasiEfek Samping & KontraindikasiInteraksi Obat
1.2.3.4.5.6.7.NRTI (Nucleoside Reverse Transcriptase Inhibitor)
Zidovudin
Didanosin
Zalsitabin
Stavudin
Lamivudin
Emtrisitabin
AbakavirA: Rapidly absorbed. T max is 0.5 to 1.5 h. Oral bioavailability is 64%D: It is extensively distributed. Binding to plasma protein is low, less than 38%. Apparent Vd is 1.6 L/kgM: Hepatic metabolism. Major metabolites are D-glucopyranuronosylthymidine (GZDV) and 3-amino-3-deoxythymidineE: Primarily eliminated by hepatic metabolism. Half-life is 0.5 to 3 h. GZDV (74%) and zidovudine (14%) are recovered in the urine.A: T max is 0.25 to 1.5 h (buffered formulation), 2 h (delayed-release). Bioavailability is approximately 42% (buffered formulation). Food decreases the C max and AUC by approximately 55% when didanosine tablets were administered up to 2 h after a meal. C max and AUC of the delayed-release capsules decreased by approximately 46% and 19%, respectively, in the presence of food. Should be taken on an empty stomach.D: Less than 5% protein bound. Vd is approximatelyM: Hepatic metabolismE: Half-life is approximately 1.5 h (buffered formulation). Approximately 18% recovered in the urine (buffered formulation). Renal Cl is approximately 458 mL/min (buffered formulation) in patients with CrCl of at least 90 mL/min.A: Oral bioavailability is about 80%. Absorption rate of a 1.5 mg oral dose was reduced with food. C max is 25.2 ng/mL. T max is 0.8 h. AUC is 72 ngh/mLD: Vd is 0.534 L/kg. Less than 4% protein bound. Drug interaction involved in binding site is unlikely.
M: Major metabolite is dideoxyuridineE: Renal elimination is the primary route of eliminationA: Following oral administration, stavudine is rapidly absorbed, with peak plasma concentrations occurring within 1 hour after dosingD: Binding of stavudine to serum proteins was negligible over the concentration range of 0.01 to 11.4g/mL. Stavudine distributes equally between red blood cells and plasmaM: Metabolism plays a limited role in the clearance of stavudine. Unchanged stavudine was the major drug-related component circulating in plasma after an 80-mg dose of 14C-stavudine, while metabolites constituted minor components of the circulating radioactivity. Minor metabolites include oxidized stavudine, glucuronide conjugates of stavudine and its oxidized metabolite, and an N-acetylcysteine conjugate of the ribose after glycosidic cleavage, suggesting that thymine is also a metabolite of stavudineE: Following an 80-mg dose of 14C-stavudine to healthy subjects, approximately 95% and 3% of the total radioactivity was recovered in urine and feces, respectively. Radioactivity due to parent drug in urine and feces was 73.7% and 62.0%, respectively. The mean terminal elimination half-life is approximately 2.3 hours following single oral doses. Mean renal clearance of the parent compound is approximately 272 mL/min, accounting for approximately 67% of the apparent oral clearance
A: C max is approximately 1.28mcg/mL (single dose of 100 mg). T max is 0.5 to 2h. Absolute bioavailability is approximately 87%D: Less than 36% protein bound. Vd is approximately 1.3 L/kgM: Metabolism of lamivudine is a minor route of elimination. The metabolite is trans-sulfoxide metaboliteE: The majority is eliminated unchanged in the urine. Mean half-life is 5 to 7 h. Cl is approximately 398.5 mL/minA: Rapidly and extensively absorbed after oral administration. Absolute bioavailability is about 93% (capsules) and 75% (solution). Postdose T max about 1 to 2 h. Steady-state C max is about 1.8 mcg/mL. AUC is about 10 mcg/mLh. Mean steady-state trough plasma concentration at 24 h postdose is 0.09 mcg/mLD:-M:-E: Plasma t is approximately 10 h. Eliminated in the urine (86% with 13% as putative metabolites) and feces (14%).A: Rapidly and extensively absorbed. Bioavailability is 83% (tablets). C max is approximately 3 mcg/mL and AUC 0-12 is approximately 6.02 mcgh/mLD: Vd after IV administration is approximately 0.86 L/kg. Plasma protein binding is approximately 50%M: Metabolized to inactive metabolites by alcohol dehydrogenaseE: 1.2% is excreted in the urine as abacavir; 81% as inactive metabolites; 16% is excreted in the feces. The half-life is approximately 1.54 h and Cl is approximately 0.8 L/h/kg (after IV administration).
Gugus AZT 5-monofosfat bergabung pada ujung 3 rantai DNA virus & menghambat enzim reverse transcriptase (RT)
Bekerja pada HIV RT dengan cara menghentikan pembentukan rantai DNA virus
Bekerja pada HIV RT dengan cara menghentikan pembentukan rantai DNA virus
Bekerja pada HIV RT dengan cara menghentikan pembentukan rantai DNA virus
Bekerja pada HIV RT & HBV RT dengan cara menghentikan pembentukan rantai DNA virus
Merupakan derivate 5-flourinated lamivudin, diubah ke bentuk trifosfat oleh enzim selular. Bekerja pada HIV RT & HBV RT dengan cara menghentikan pembentukan rantai DNA virus
Bekerja pada HIV RT dengan cara menghentikan pembentukan rantai DNA virus
Spektrum: HIV type 1 & 2
Indikasi: infeksi HIV dalam kombinasi dengan anti-HIV lainnya (lamivudin, abakavir), mencegah penularan HIV dari ibu ke janinSpektrum: HIV type 1 & 2
Indikasi: infeksi HIV, terutama infeksi HIV tingkat lanjut, dalam kombinasi dengan anti HIV lainnya
Spektrum: HIV type 1 & 2
Indikasi: infeksi HIV, terutama pasien dewasa infeksi HIV tingkat lanjut yang tidak responsive terhadap zidovudin, dalam kombinasi dengan anti HIV lainnya (bukan didanosin)
Spektrum: HIV type 1 & 2
Indikasi: infeksi HIV, terutama infeksi HIV tingkat lanjut, dalam kombinasi dengan anti HIV lainnya
Spektrum: HIV type 1 & 2, HBV
Indikasi: infeksi HIV & HBV, infeksi HIV dalam kombinasi dengan anti HIV lainnya (zidovudin & abakavir)Spektrum: HIV type 1 & 2, HBV
Indikasi: infeksi HIV & HBVSpektrum: HIV type 1 & 2
Indikasi: infeksi HIV, dalam kombinasi dengan anti HIV lainnya (zidovudin, lamivudin)Efek samping: anemia, neutropenia, sakit kepala, mual
Kontraindikasi: Potentially life-threatening hypersensitivity to any component.Efek samping: diare, pancreatitis, neuropati perifer
Kontraindikasi:
Potentially life-threatening hypersensitivity to any component.Efek samping: neuropati perifer, stomatitis, ruam, pankreatitis
Kontraindikasi:
Potentially life-threatening hypersensitivity to any component.Efek samping: neuropati perifer, asidosis laktat, sakit kepala, ruam, mualKontraindikasi: Stavudine is contraindicated in patients with clinically significant hypersensitivity to stavudine or to any of the components contained in the formulationEfek samping: asidosis laktat, hepatomegali dengan steatosis, sakit kepala, mual
Kontraindikasi:
Potentially life-threatening hypersensitivity to any component.Efek samping: nyeri abdomen, diare, malaise, sakit kepala, lipodistrofi, mual, rhinitis, pruritus, ruam. Jarang: reaksi alergi, asidosis laktat, mimpi buruk, parestesia, pneumonia, steatosis hati
Kontraindikasi:
Potentially life-threatening hypersensitivity to any component.Efek samping: mual, muntah, diare, reaksi hipersensitif (demam, malaise, ruam), gangguan gastrointestinal
Kontraindikasi: Moderate or severe hepatic function impairment; hypersensitivity to any component of the product.Adriamycin, amphotericin B, dapsone, flucytosine, interferon, pentamidine, vinblastine, vincristine
May increase risk of toxicity, including nephrotoxicity, cytotoxicity, or hematologic toxicity.
Atovaquone, fluconazole, methadone, probenecid, valproic acid
May increase serum concentration and potential toxicity of zidovudine.
Doxorubicin
May antagonize the effect of zidovudine. Avoid use.
Experimental nucleoside analogs
May affect RBC/WBC counts or function and may increase potential for hematologic toxicity.
Ganciclovir
Life-threatening hematologic toxicity may occur.
Nelfinavir, ribavirin, rifamycin, ritonavir, stavudine
May decrease zidovudine serum concentrations, reducing the therapeutic effect.
Phenytoin
Phenytoin levels have been reported to increase, decrease, or not change with coadministration. Zidovudine Cl is decreased.Allopurinol
Because allopurinol may cause increased didanosine plasma levels, do not coadminister.
Antacids
Aluminum- or magnesium-containing antacids may potentiate adverse reactions associated with the antacid component of didanosine pediatric powder.
Antiretroviral agents
Antiretroviral agents have caused fatal lactic acidosis in women when coadministered with didanosine.
Delavirdine, indinavir
Administer 1 h prior to didanosine to avoid decreasing plasma levels of delavirdine or indinavir.
Drugs that cause peripheral neuropathy or pancreatitis
Increased risk of these toxicities.
Food
Reduces absorption of didanosine by as much as 50%.
Fluoroquinolones, tetracyclines
Do not administer within 2 h of didanosine.
Ganciclovir, valganciclovir
Didanosine plasma concentrations may be elevated, increasing the risk of toxicity.
Itraconazole, ketoconazole, dapsone, and other drugs whose absorption can be affected by gastric acidity
Administer at least 2 h before didanosine.
Methadone
May decrease didanosine plasma levels.
Ribavirin
Risk of didanosine toxicity may be increased. Avoid coadministration.
Tenofovir disoproxil fumarate
Didanosine plasma concentrations may be increased, necessitating a dose reduction in didanosine.
Aminoglycosides, amphotericin, foscarnet
May increase risk of peripheral neuropathy and other zalcitabine toxicities caused by decreased clearance of zalcitabine.
Chloramphenicol, cisplatin, dapsone, disulfiram, ethionamide, glutethimide, gold, hydralazine, iodoquinol, isoniazid, metronidazole, nitrofurantoin, phenytoin, ribavirin, vincristine
May increase risk of peripheral neuropathy.Drugs associated with pancreatitis (eg, pentamidine)
Fatal pancreatitis has occurred, possibly related to zalcitabine and IV pentamidine given concurrently.
Zidovudine: Zidovudine competitively inhibits the intracellular phosphorylation of stavudine. Therefore, use of zidovudine in combination with stavudine should be avoided.
Doxorubicin: In vitro data indicate that the phosphorylation of stavudine is inhibited at relevant concentrations by doxorubicin.
Ribavirin: In vitro data indicate ribavirin reduces phosphorylation of lamivudine, stavudine, and zidovudine. However, no pharmacokinetic (eg, plasma concentrations or intracellular triphosphorylated active metabolite concentrations) or pharmacodynamic (eg, loss of HIV/HCV virologic suppression) interaction was observed when ribavirin and lamivudine (n=18), stavudine (n=10), or zidovudine (n=6) were coadministered as part of a multi-drug regimen to HIV/HCV co-infected patients
Stavudine does not inhibit the major cytochrome P450 isoforms CYP1A2, CYP2C9, CYP2C19, CYP2D6, and CYP3A4; therefore, it is unlikely that clinically significant drug interactions will occur with drugs metabolized through these pathways.
Because stavudine is not protein-bound, it is not expected to affect the pharmacokinetics of protein-bound drugs
Drugs with active renal secretion via the organic cationic transport system (eg, trimethoprim)
Lamivudine Cl may be reduced, increasing plasma concentrations.
Interferon- and ribavirin-based regimens
Risk of hepatic decompensation may be increased.
Zalcitabine
Lamivudine and zalcitabine may inhibit the intracellular phosphorylation of one another. Use in combination is not recommended
Do not coadminister with Atripla or Truvada , or drugs containing lamivudineEthanol
Increases exposure to abacavir by decreasing the elimination and prolonging the half-life.
Methadone
Plasma levels of methadone may be decreased in some patients, reducing the therapeutic effect.
8.NtRTI (Nucleotide Reverse Transcriptase Inhibitor)
Tenofovir disoproksil fumaratA: Tenofovir C max is approximately 0.3 mcg/mL and AUC is approximately 2.29 ngh/mL. T max is approximately 1 h, and bioavailability is approximately 25%. Administration following a high-fat meal increases the oral bioavailability, with an increase in AUC of approximately 40% and a C max increase of approximately 14%.
D: Vd of tenofovir is approximately 1.3 L/kg. Binding to plasma or serum proteins is less than 0.7% and 7.2%, respectively M: Tenofovir is not a CYP-450 substrateE: Elimination of tenofovir is by glomerular filtration and tubular secretion. Approximately 70% to 80% is recovered in urine as unchanged drug.Bekerja pada HIV RT & HBV RT dengan cara menghentikan pembentukan rantai DNA virus
Spektrum: HIV type 1 & 2 serta berbagai retrovirus lainnya & HBV
Indikasi: infeksi HIV dalam kombinasi dengan efavirens, tidak boleh dikombinasi dengan lamivudin & abakavirEfek samping: mual, muntah, flatulens, diare
Kontraindikasi: None well documented.Atazanavir, indinavir
May increase tenofovir plasma levels. Tenofovir may decrease plasma levels of atazanavir and indinavir, decreasing the therapeutic effect. Do not administer atazanavir without ritonavir in patients receiving tenofovir.
Didanosine
Plasma concentrations of didanosine may be increased, increasing the risk of adverse reactions. Reduce the didanosine dose to 250 mg when administered with tenofovir in adults weighing more than 60 kg. Monitor patients closely and administer under fasting conditions.
Drugs that reduce renal function or compete for active tubular secretion (eg, acyclovir, adefovir, cidofovir, ganciclovir)
May increase serum levels of tenofovir and/or increase the levels of other renally eliminated drugs. Avoid coadministration with adefovir.
Lopinavir/Ritonavir
May increase tenofovir plasma levels.
Nephrotoxic agents
Risk of nephrotoxicity may be increased; avoid tenofovir in patients who are currently receiving or have recently received nephrotoxic agents.
NSAIDs (eg, ibuprofen)
May increase the pharmacologic and toxic effects of tenofovir. Coadminister with caution.
Tacrolimus
Coadministration increased C max of tenofovir by 13%.
9.10.11.NNRTI (Non-Nucleoside Reverse Transcriptase Inhibitor)
Nevirapin
Delavirdin
EfavirenzA: Nevirapine oral absorption is more than 90%, bioavailability is 93% (tablets) and 91% (oral solution), T max is 4 h, and C max is approximately 2 mcg/mLD: Nevirapine crosses the placenta and is found in breast milk. Protein binding is approximately 60% and is highly lipophilic and widely distributed. Nevirapine Vd is 1.21L/kg (IV), and CSF approximates 45% of concentration in plasma.M: Extensively metabolized by CYP isozymes. In vitro studies indicated that metabolism is primarily by CYP2B6 and CYP3A4E: Nevirapine is eliminated in urine (81.3%) and feces (10.1%). Less than 3% of the parent compound is excreted in urine. Autoinduction results in a decrease of the half-life from 45 h (single dose) to approximately 25 to 30 h (multiple dosesA: Rapidly absorbed. T max approximately 1 h. C max approximately 35 mcM. AUC approximately 180 mcMh.
D: Approximately 98% protein bound, primarily albuminM: Primarily metabolized by CYP3A and possibly CYP2D6 to several inactive metabolites.E: Approximately 44% excreted in feces and approximately 51% in urine (less than 5% as unchanged drug). t is approximately 5.8 h.
A: C max is 1.6 to 9.1 mcM. T max is 3to 5h. Food significantly increases the AUC and C max . Should be taken on an empty stomachD: Approximately 99.5% to 99.75% is protein bound, predominantly to albuminM: Metabolized in the liver by CYP-450 (primarily CYP3A4 and CYP2B6) to inactive metabolitesE: The half-life is 52 to 76 h (single dose) and 40to 55 h (multiple doses). Approximately 14% to 34% is excreted in the urine (less than 1% as unchanged drug), and 16% to 61% is excreted in the feces.Bekerja pada siklus alosterik tempat ikatan non-substrat HIV-1 RT
Bekerja pada siklus alosterik tempat ikatan non-substrat HIV-1 RT
Bekerja pada siklus alosterik tempat ikatan non-substrat HIV-1 RT
Spektrum: HIV type 1 Indikasi: infeksi HIV-1 dalam kombinasi dengan anti-HIV lainnya, terutama NRTISpektrum: HIV type 1 Indikasi: infeksi HIV-1 dalam kombinasi dengan anti-HIV lainnya, terutama NRTI
Spektrum: HIV type 1 Indikasi: infeksi HIV-1 dalam kombinasi dengan anti-HIV lainnya, terutama NRTI & NtRTIEfek samping: ruam, demam, fatigue, sakit kepala, somnolens, mual, peningkatan enzim hati
Kontraindikasi: Moderate or severe (Child-Pugh class B or C) hepatic impairment.
Efek samping: ruam, peningkatan tes fungsi hati, neutropenia
Kontraindikasi: Potentially life-threatening hypersensitivity to any component.Efek samping: sakit kepala, pusing, mimpi buruk, sulit konsentrasi, ruam
Kontraindikasi: Concomitant use with astemizole, bepridil, cisapride, ergot derivatives, midazolam, pimozide, St. John's wort, triazolam, or voriconazole (in standard doses); hypersensitivity to any component of the product.
Amiodarone, carbamazepine, cisapride, clonazepam, cyclophosphamide, cyclosporine, diltiazem, disopyramide, ergotamine, ethosuximide, fentanyl, itraconazole, lidocaine systemic, nifedipine, sirolimus, tacrolimus, verapamil
Plasma levels may be decreased by nevirapine.
Clarithromycin
Clarithromycin concentrations may be reduced, while concentrations of the active metabolite of clarithromycin may be increased.
Contraceptives, hormonal
Lower hormone levels and potential contraceptive failure.
Efavirenz, methadone
Concentrations of these agents may be decreased by nevirapine.
Fluconazole
Nevirapine concentrations may be increased.
HMG-CoA reductase inhibitors (eg, atorvastatin, simvastatin)
Risk of severe myopathy or rhabdomyolysis may be increased.
Ketoconazole
Coadministration resulted in significant reduction in ketoconazole plasma concentrations. Do not coadminister ketoconazole and nevirapine.
Protease inhibitors (eg, indinavir)
Lower protease inhibitor plasma levels.
Rifabutin
Rifabutin concentrations may be increased.
Rifampin, rifabutin, rosiglitazone
Lower nevirapine plasma levels.
St. John's wort
May reduce nevirapine concentrations, resulting in loss of virologic response and possible resistance to nevirapine and the class of NNRTIs.
Warfarin
Plasma concentrations of warfarin may be altered, resulting in potential increases in coagulation time.
Antacids
Antacids reduce absorption of delavirdine. Separate doses by at least 1h.
Anticonvulsants (eg, carbamazepine, phenobarbital, phenytoin)
Induce hepatic metabolism of delavirdine resulting in decreased plasma concentrations.
Benzodiazepines (eg, alprazolam, midazolam, triazolam)
Delavirdine may increase blood levels of these drugs, which may produce extreme sedation and respiratory depression.
Cisapride, dapsone, ergot derivatives, quinidine, rifabutin, warfarin
Delavirdine may elevate blood levels of these drugs, which may increase the risk of arrhythmias or other potentially serious adverse reactions.
Clarithromycin
Coadministration may increase blood levels of delavirdine or clarithromycin.
Didanosine
Separate administration of didanosine and delavirdine by at least 1 h; coadministration results in a 20% reduction in systemic exposure of both drugs.
Dihydropyridine calcium channel blockers (eg, nifedipine)
Delavirdine may elevate blood levels, which may increase toxicity.
Fluoxetine, ketoconazole
Increased delavirdine plasma concentrations.
H 2 antagonists (eg, cimetidine)
Concurrent use may reduce absorption of delavirdine. Chronic use of these drugs with delavirdine is not recommended.
Indinavir
Delavirdine inhibits metabolism of indinavir. Consider indinavir dosage reduction if coadministered with delavirdine.
Rifabutin, rifampin
Induce hepatic metabolism of delavirdine resulting in decreased plasma concentrations. These agents should not be coadministered with delavirdine.
Saquinavir
Delavirdine inhibits metabolism of saquinavir. Monitor hepatocellular enzymes frequently if coadministered.
Astemizole, bepridil, cisapride, ergot derivatives, midazolam, pimozide, triazolam
May elevate levels of these drugs, which may increase the risk of arrhythmias, hematologic abnormalities, or other potentially serious adverse reactions. Coadministration is contraindicated.
Atazanavir, calcium channel blockers (eg, diltiazem, felodipine, nicardipine, nifedipine, verapamil), clarithromycin, fosamprenavir, HMG-CoA reductase inhibitors (atorvastatin, pravastatin, simvastatin), indinavir, itraconazole, ketoconazole, lopinavir, methadone, rifabutin, saquinavir, sertraline
Efavirenz may decrease plasma concentrations of these agents, which could reduce their efficacy.
Carbamazepine, phenobarbital, phenytoin
Plasma concentrations of the anticonvulsant may decrease.
Carbamazepine, phenobarbital, phenytoin, rifampin
May decrease plasma levels of efavirenz, which may reduce antiviral activity.
Ethinyl estradiol, nelfinavir, ritonavir
Efavirenz may increase plasma concentrations, which could increase activity or toxicity of these agents.
Food
Efavirenz plasma concentrations may be elevated, increasing the risk of adverse reactions. Efavirenz should be taken on an empty stomach, preferably at bedtime.
Ritonavir
May increase efavirenz plasma levels, which could increase adverse reactions.
St. John's wort
May reduce efavirenz plasma concentrations, which may decrease the clinical efficacy. Coadministration is contraindicated.
Voriconazole
Contraindicated at standard doses. Voriconazole doses should be increased while efavirenz doses should be decreased.
Warfarin
Plasma concentrations may be increased or decreased
12.13.14.15.16.17.18.PI (Protease Inhibitor)
Sakuinavir
Ritonavir
Indinavir
Nelfinavir
Amprenavir
Lopinavir
AtazanavirA:
D:
M:
E:
A:
D:
M:
E:
A:
D:
M:
E:
A:
D:
M:
E:
A:
D:
M:
E:
A:
D:
M:
E:
A:
D:
M:
E:Bekerja pada tahap transisi, menghambat enzim HIV protease peptidomimetic
Bekerja pada tahap transisi, menghambat enzim HIV protease peptidomimetic
Bekerja pada tahap transisi, menghambat enzim HIV protease peptidomimetic
Bekerja pada tahap transisi, menghambat enzim HIV protease peptidomimetic
Bekerja pada tahap transisi, menghambat enzim HIV protease peptidomimetic
Bekerja pada tahap transisi, menghambat enzim HIV protease peptidomimetic
Bekerja pada tahap transisi, menghambat enzim HIV protease peptidomimetic
Spektrum: HIV type 1 & 2
Indikasi: infeksi HIV dalam kombinasi dengan anti-HIV lainnya (NRTI & beberapa PI seperti ritonavir)
Spektrum: HIV type 1 & 2
Indikasi: infeksi HIV dalam kombinasi dengan anti-HIV lainnya (NRTI & beberapa PI seperti sakuinavir)
Spektrum: HIV type 1 & 2
Indikasi: infeksi HIV dalam kombinasi dengan anti-HIV lainnya seperti NRTI
Spektrum: HIV type 1 & 2
Indikasi: infeksi HIV dalam kombinasi dengan anti-HIV lainnya seperti NRTI
Spektrum: HIV type 1 & 2
Indikasi: infeksi HIV dalam kombinasi dengan anti-HIV lainnya seperti NRTISpektrum: HIV type 1 & 2
Indikasi: infeksi HIV dalam kombinasi dengan anti-HIV lainnya seperti NRTI
Spektrum: HIV type 1 & 2
Indikasi: infeksi HIV dalam kombinasi dengan anti-HIV lainnya seperti NRTIEfek samping: diare, mual, nyeri abdomen
Kontraindikasi:
Efek samping: mual, muntah, diare
Kontraindikasi:
Efek samping: mual, hiperbilirubinemia, batu ginjal
Kontraindikasi:
Efek samping: diare, mual, muntah
Kontraindikasi:
Efek samping: mual, diare, ruam, parestesia perioral/ oral
Kontraindikasi:
Efek samping: mual, muntah, peningkatan kadar kolesterol & TG, peningkatan -GT
Kontraindikasi:
Efek samping: hiperbilirubinemia, mual, perubahan EKG (jarang)
Kontraindikasi:
19.Viral Entry Inhibitor
EnfuvirtidA:
D:
M:
E:Menghambat masuknya HIV-1 ke dalam sel dengan cara menghambat fusi virus ke membrane sel: berikatan dengan HR1 (first heptad-repeat) pada subunit gp41 envelope glikoprotein virus serta menghambat terjadinya perubahan konformasi yang dibutuhkan untuk fusi virus ke membrane sel
Spektrum: HIV type 1
Indikasi: infeksi HIV-1 dalam kombinasi dengan anti-HIV lainnya Efek samping: reaksi local (nyeri, eritema, pruritus, iritasi, nodul/ kista), eosinofilia, pneumonia bakterial
Kontraindikasi:
Related Documents