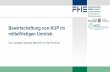
1 Synthesis reactions 2 r more elements or compounds are joined together to make 1 compound Usually do Nottake place in solution but are solids and gases General form A B AB I Decompositionreactions opposite of synthesis one compound is broken into 2 parts element compound Usually do not take place in solution Cage but are solids G and gases g General form a AB A t B III Single replacement reactions one element replaces a similar element in a compound Take place in aqueous solution aq mutual metal General form A t BX AX t B w halogen halogen Y t BX By t x Catton metal

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript
1 Synthesis reactions 2 r more elements or compounds are
joinedtogethertomake 1 compoundUsuallydoNottakeplacein solutionbutare solidsandgasesGeneralform
A B ABI Decompositionreactions opposite of synthesisone compound is brokeninto 2parts element compoundUsuallydonottakeplace in solutionCagebut aresolids G and gases gGeneral forma
AB A t B
III Singlereplacementreactionsone elementreplaces a similarelement in a compoundTake place in aqueoussolution aqmutual metalGeneralform A t BX AX t B
whalogen halogen
Y t BX By t x
Cattonmetal
N Double replacement reactions Ionic reactionsIons of 2 Separate compoundschangeplacesAlmostalwaystakeplacein aqueoussolutionCaqBothreactants mustbe solublecap Oneproductisusuallysoluble and theothermaybe a solid precipitate or maybegas 01 maybewaterGeneralform
AX t BY AY t BXCage Carl Cage s
lg
w Combustion reactions reactantsare a hydrocarbon
and oxygen
Complete combustion
hydrocarbons burnin air toyield carbondioxide andwaterI
CHucg t 02cg 8 CO2cg t He0cg
Incomplete combustioni
hydrocarbons combust in air to yield carbon dioxide
water carbon assoot and carbonmonoxide
CHutOz Coz HzO t Ces t C0cgG Cg
Types of reactionsand reaction prediction 1There are 5 maintypes of reactions Each typehasseveral8 or categories
1 Synthesis reactions 2 r more elements or compounds are
joinedtogethertomake 1 compoundUsuallydoNottakeplacein solutionbutare solidsandgasesGeneralform
A B D ABSubtypes1 Reaction of elementwithoxygentoform oxide
a2mg s t Omg 3 2mgOcs2 Reaction of 2nonmetals toform amolecular compound
A Hzcgy t 02cg 8 H2OCgb Sg s t 02cg 3 502cg 503cg
C Ces t Hzg 3 CxHy3 Reactionofmetalwith nonmetal otherthanoxygen
to form a salta halogen t alkalimetal alkalineearthmetal transitionmetal
b Macs t Clegg 0 NaCl4 Reaction ofmetallicoxidewithwateryieldmetalhydroxidesA Calles Azole CaLoH z s base
5 Reaction of nonmetallicoxidewithwaterproduceoxyacidsacid
a 503cg t Azole D H2504Cad6 Reaction of metaloxidewithnonmetal.oxideproduces saltsA Caoes t 503 Cas04Csg
g
Il Decompositionreactions opposite of synthesis 2One compound is brokeninto 2parts element compoundUsuallydonottakeplace in solutionCaqlbut aresolids G and gases gGeneral forma
AB A t Bsubtype zeumentst Binaryampoundswhenhealed decomposeintotheirelements
a HgOcs Is Agee 02cgA heat
2 Somebinarycompoundsdecomposewhenan electricalcurrentis passedthroughthem rn electricalcurrent
rna2h20ee 2Hugs t 02cg
3 Metal CarbonatesWhenhealed breakdown intometaloxide and carbondioxide
a Cacoz CaOes t Cong4 Metalhydroxideswhenhealed breakdowninto metaloxides
and watera CaOHzg
D Calles t H2O g
5Metal chlorates whentreated decomposeintometalchlorideand oxygengasa closes
DKCles t 02cg
6 Manyoxyacids decompose into anonmetallicoxide t water
Hz slags coz t Helleg
HI Singlereplacementreactions 3one elementreplaces a similarelement in a compoundTake place in aqueoussolution aqGeneralform A t BX AX t B
w
subtypesEET In
1 Replacementof ametalbyamoreactivemetalAlesi t FeNO3 z D fees t Al NO3 3CageylaysFeG t Al Nos z age NoRXM
2 Replacement of hydrogeninwaterby ametalMacs t lettuce 3 NaOHCaq t Hzgmoreactivemetals reactwithwaterbaseless activemetals reactwith steam
3 Replacement of hydrogen in acid by ametalMgoss t HClcap MgCk t Hzeg
aq4 Replacementof halogen by amoreactivehalogen
Clzeg t kBread 2 KClcan BEGatwarmtemp
N Double replacement reactions Ionic reactions 4Ions of 2 Separate compoundschangeplacesAlmostalwaystakeplacein aqueoussolutionCaqBothreactants mustbe solublecap Oneproductisusuallysoluble and theothermaybe a solid precipitate or maybegas 01 maybewaterGeneralform
AX t BY AY t BXsubtypes
a formationof a precipitate pptPb Nosz t z KI 2KNos t Pbiz
lag Caq aq s
b formationof a gasFes 2HClcap Feckeapt HfsCg
1 HI tFesc formationofwater usually acid base neutralization
reaction0
Hclap T NaoHeagy HOHayt Madcap
It coz2
ZHUcaq t Cacozes CaAzt HzC0zaq carbonicacid6
Hzcoz I Had 1CozyCaa cel
V Combustion reactions
hydrocarbonscombinewith oxygen to give coz Azocore Coe CO C Oz
complete combustion
Csltgcg t 02cg cozy t HzOcg
Mgc t HCl Hz t NgCkMogg tHFHIqgytmgiagtzclc.gg
oxidation state electrons an atomhas gained or lostoxidation lossof electrons ex Mg Mgt2tze LEOreduction gainofelectrons Cl t e a I says
GERReactionornot
My t Cudz tngt't za
MI t Ca tat Cues t MgdzSilver bluesoln brown colorless
ppt shn
NO RANnanny ti gM brown t cosigyss
Ppt ppt
Mg will replacedudu will Not replacerag
Mgis more activethan Ca
Related Documents