UROPATHOLOGY REPORT Batch#: 24724 1 of 2 STPL IS;RL;MMR; (212) 123-4568 (212) 123-4567 Suite: 8A NEW YORK, NY 10016 (718) 555-2541 ABC SURGERY CENTER Acct # 352-549-2 PHYSICIAN: JOHN U. SMITH DOB: Age: 01/02/19XX 58 PATIENT: JACK JONES FLUSHING, NY 11365 Male 1018 Acct: Sex: Accession: Received: Obtained: 09/18/20XX 09/18/20XX 6:07 pm 000000-20XX Final Report Date Preliminary Date Specimen N/A 09/19/XX A N/A 09/19/XX B 09/19/XX 09/22/XX C 09/19/XX 09/22/XX D 09/19/XX 09/22/XX E 09/19/XX 09/22/XX F N/A 09/19/XX G 09/19/XX 09/22/XX H N/A 09/19/XX I 09/19/XX 09/22/XX J 09/19/XX 09/22/XX K 09/19/XX 09/22/XX L 12548 MAIN ST NEW YORK, NY 10011 456 SAMPLE BLVD. DR. JANE U. DOE 28 South Terminal Drive Tel#: www.acupath.com Fax#: 1-888-ACUPATH 1-516-326-3452 Plainview, NY 11803 1234 FIRST AVE 6 Rte A Copy was sent to: Submitted: 12 vials-A,B,C,D,E,F,G,H,I,J,K,L Comments: 790.93 C, D, J, L: ADENOCARCINOMA OF THE PROSTATE. There is a gland forming neoplasm in prostatic stroma composed of neoplastic epithelial cells with enlarged nuclei and prominent nucleoli. E, F, K: ADENOCARCINOMA OF THE PROSTATE WITH FOCAL PERINEURAL INVASION. Sections reveal a gland forming neoplasm adjacent to a nerve. H: SMALL FOCUS OF ADENOCARCINOMA OF THE PROSTATE. There is a gland forming neoplasm in prostatic stroma composed of neoplastic epithelial cells with enlarged nuclei and prominent nucleoli. A: BENIGN PROSTATIC HYPERPLASIA. There is hyperplasia of glands and surrounding fibrous stroma. B, G: BENIGN PROSTATIC HYPERPLASIA WITH ATROPHIC FEATURES. There is focal hyperplasia of glands and surrounding fibrous stroma. Other areas reveal dilated glands with atrophic epithelium. I: FIBROMUSCULAR STROMA ONLY. This specimen consists of fibromuscular stroma only. No glandular elements are seen. DIAGNOSIS & MICROSCOPIC DESCRIPTION GLEASONS SCORE & % OF BIOPSY REPLACED BY CARCINOMA A B C 6 (3+3) 10% D 6 (3+3) 5% E 7 (3+4) 60% F 7 (3+4) 50% G H 6 (3+3) 2% I J 7 (3+4) 50% K 7 (3+4) 60% L 6 (3+3) 90% RLA(I) BENIGN GS:6(3+3) RLM(H) RLB(G) BENIGN 2% RB(A) RM(B) RA(C) GS:6(3+3) BENIGN BENIGN 10% LB(D) LM(E) LA(F) GS:7(3+4) GS:7(3+4) GS:6(3+3) 5% 60% 50% LLB(J) LLM(K) LLA(L) GS:7(3+4) GS:7(3+4) GS:6(3+3) 50% 60% 90% Color Key Photomicrograph of J Adenocarcinoma Benign Infectious Inflammatory Suspicious Pin Other No Diag Atypical John Brown, M.D. Continued on next page Olga Falkowski, M.D., Medical Director JACK JONES 09/18/20XX JACK JONES RLB(G) RLM(H) RLA(I) BENIGN GS:6(3+3) BENIGN RB(A) RM(B) RA(C) BENIGN BENIGN GS:6(3+3) LB(D) LM(E) LA(F) GS:6(3+3) GS:7(3+4) GS:7(3+4) LLB(J) LLM(K) LLA(L) GS:7(3+4) GS:7(3+4) GS:6(3+3) 6(3+3) 6(3+3) 7(3+4) 7(3+4) 6(3+3) 7(3+4) A B C D E F G H I J JACK JONES 09/18/20XX 6(3+3) 6(3+3) 7(3+4) 7(3+4) 6(3+3) 7(3+4) JACK JONES A B C D E F G H I J Note: Prostate diagrams and labels indicate disease summaries only. See diagnosis above for details K L K L 7(3+4) 6(3+3) 7(3+4) 6(3+3) John Brown

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

UROPATHOLOGY REPORT Batch#: 24724 1 of 2STPLIS;RL;MMR;
(212) 123-4568 (212) 123-4567
Suite: 8A NEW YORK, NY 10016 (718) 555-2541
ABC SURGERY CENTER
Acct # 352-549-2
PHYSICIAN: JOHN U. SMITH
DOB:
Age:
01/02/19XX
58
PATIENT: JACK JONES
FLUSHING, NY 11365
Male 1018 Acct:
Sex:
Accession:
Received: Obtained: 09/18/20XX
09/18/20XX 6:07 pm
000000-20XX
Final Report Date Preliminary Date Specimen
N/A
09/19/XX
A
N/A
09/19/XX
B
09/19/XX
09/22/XX
C
09/19/XX
09/22/XX
D
09/19/XX
09/22/XX
E
09/19/XX
09/22/XX
F
N/A
09/19/XX
G
09/19/XX
09/22/XX
H
N/A
09/19/XX
I
09/19/XX
09/22/XX
J
09/19/XX
09/22/XX
K
09/19/XX
09/22/XX
L
12548 MAIN ST
NEW YORK, NY 10011 456 SAMPLE BLVD. DR. JANE U. DOE
28 South Terminal Drive
Tel#:
www.acupath.com Fax#:
1-888-ACUPATH 1-516-326-3452
Plainview, NY 11803
1234 FIRST AVE
6 Rte
A Copy was sent to: Submitted: 12 vials-A,B,C,D,E,F,G,H,I,J,K,L
Comments: 790.93
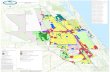
C, D, J, L: ADENOCARCINOMA OF THE PROSTATE. There is a gland forming neoplasm in prostatic stroma composed of neoplastic epithelial cells with enlarged nuclei and prominent nucleoli. E, F, K: ADENOCARCINOMA OF THE PROSTATE WITH FOCAL PERINEURAL INVASION. Sections reveal a gland forming neoplasm adjacent to a nerve. H: SMALL FOCUS OF ADENOCARCINOMA OF THE PROSTATE. There is a gland forming neoplasm in prostatic stroma composed of neoplastic epithelial cells with enlarged nuclei and prominent nucleoli. A: BENIGN PROSTATIC HYPERPLASIA. There is hyperplasia of glands and surrounding fibrous stroma. B, G: BENIGN PROSTATIC HYPERPLASIA WITH ATROPHIC FEATURES. There is focal hyperplasia of glands and surrounding fibrous stroma. Other areas reveal dilated glands with atrophic epithelium. I: FIBROMUSCULAR STROMA ONLY. This specimen consists of fibromuscular stroma only. No glandular elements are seen.
DIAGNOSIS & MICROSCOPIC DESCRIPTION
GLEASONS SCORE & % OF BIOPSY REPLACED BY CARCINOMA
A B C 6 (3+3)
10%
D 6 (3+3)
5%
E 7 (3+4)
60%
F7 (3+4)
50%
G H6 (3+3)
2%
I J 7 (3+4)
50%
K7 (3+4)
60%
L6 (3+3)
90%
RLA(I)
BENIGN
GS:6(3+3)
RLM(H)
RLB(G)
BENIGN
2%
RB(A)
RM(B)
RA(C)
GS:6(3+3)
BENIGN
BENIGN
10%
LB(D)
LM(E)
LA(F)
GS:7(3+4)
GS:7(3+4)
GS:6(3+3)
5%
60%
50%
LLB(J)
LLM(K)
LLA(L)
GS:7(3+4)
GS:7(3+4)
GS:6(3+3)
50%
60%
90%
Color Key
Photomicrograph of J
Adenocarcinoma Benign Infectious Inflammatory Suspicious Pin Other No Diag Atypical
John Brown, M.D.
Continued on next page
Olga Falkowski, M.D., Medical Director
JACK JONES 09/18/20XX JACK JONES
RLB(G)
RLM(H)
RLA(I)
BENIGN
GS:6(3+3)
BENIGN
RB(A)
RM(B)
RA(C)
BENIGN
BENIGN
GS:6(3+3)
LB(D)
LM(E)
LA(F)
GS:6(3+3)
GS:7(3+4)
GS:7(3+4)
LLB(J)
LLM(K)
LLA(L)
GS:7(3+4)
GS:7(3+4)
GS:6(3+3)
6(3+3) 6(3+3) 7(3+4) 7(3+4)
6(3+3) 7(3+4)
A B C D E F
G H I J
JACK JONES 09/18/20XX
6(3+3) 6(3+3) 7(3+4) 7(3+4)
6(3+3) 7(3+4)
JACK JONES A B C D E F
G H I J
Note: Prostate diagrams and labels indicate disease summaries only. See diagnosis above for details
K L
K L
7(3+4) 6(3+3)
7(3+4) 6(3+3)
John Brown

2 of 2JACK JONES PATIENT: 000000-20XX Accession:
www.acupath.com 1-516-326-3452 Fax#: 1-888-ACUPATH Tel#:
Plainview,NY 11803 28 South Terminal Drive
Final Date
Preliminary Date
Specimen
N/A
A
09/19/XX
N/A
B
09/19/XX
09/19/XX
C
09/22/XX
09/19/XX
D
09/22/XX
09/19/XX
E
09/22/XX
09/19/XX
F
09/22/XX
N/A
G
09/19/XX
09/19/XX
H
09/22/XX
N/A
I
09/19/XX
09/19/XX
J
09/22/XX
09/19/XX
K
09/22/XX
09/19/XX
L
09/22/XX
NOTESC: Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals weak staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 reveals no evidence of perineural invasion. D: Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals weak staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 reveals no evidence of perineural invasion. E: Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals moderate staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 is positive, supporting the diagnosis of perineural invasion.
F: Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals moderate staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 is positive, supporting the diagnosis of perineural invasion.
H: Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals weak staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 reveals no evidence of perineural invasion. J: Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals moderate staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 reveals no evidence of perineural invasion. K: Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals moderate staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 is positive, supporting the diagnosis of perineural invasion.
L: Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals weak staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 reveals no evidence of perineural invasion.
This test was developed and its performance characteristics determined by Acupath Laboratories, Inc. It has not been cleared or approved by the U.S. Food and Drug Administration. The FDA has determined that such clearance or approval is not necessary. This test is used for clinical purposes. It should not be regarded as investigational or for research. This laboratory is certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA) as qualified to perform high complexity clinical laboratory testing.
Fixative Color Shape Cassette Piece Measurement(mm)
GROSSING INFORMATION
Site
10% Formalin white Cylindrical 1 07x01x01 A Right Base 2
10% Formalin white Cylindrical 1 09x01x01 B Right Mid 1
10% Formalin white Cylindrical 1 09x01x01 C Right Apex 1
10% Formalin white Cylindrical 1 10x01x01 D Left Base 1
10% Formalin white Cylindrical 1 10x01x01 E Left Mid 1
10% Formalin white Cylindrical 1 08x01x01 F Left Apex 1
10% Formalin white Cylindrical 1 04x01x01 G Right Lateral Base 1
10% Formalin white Cylindrical 1 05x01x01 H Right Lateral Mid 1
10% Formalin white Cylindrical 1 04x01x01 I Right Lateral Apex 1
10% Formalin white Cylindrical 1 07x01x01 J Left Lateral Base 1
10% Formalin white Cylindrical 1 09x01x01 K Left Lateral Mid 1
10% Formalin white Cylindrical 1 10x01x01 L Left Lateral Apex 1

(212) 123-4567 Dr. JOHN U. SMITH Doctor:
01/02/19XX Date of Birth:
09/18/20XX Date Obtained:
JACK S JONES Patient:
PATIENT FACT SHEET TM
PROSTATE ADENOCARCINOMA Definition: The most common form of cancer in men. It is a growth that originates in the peripheral cells of the prostate and is malignant, meaning that it can spread to other organs of the body. The prostate is a gland that surrounds the male urethra and secretes a fluid during ejaculation. ______POSSIBLE RISK FACTORS_________________________________________________ -Age -Prostate cancer is very rarely found in men under 40 years old. -Men over 65 are 40 times more likely to have prostate cancer than are men under 65. -Prostate cancer is the most common cause of death from cancer in men over 75. - Prostate Specific Antigen (PSA) levels PSA of up to 4.0 is considered normal. PSA of 4.1 to 10.0 is associated with a 15%-20% risk of prostate adenocarcinoma. PSA of over 10.0 is associated with a 50% risk of prostate adenocarcinoma. -Race -Prostate cancer is most common in black men over age 60. -Prostate cancer is least common in men of Asian (Oriental) decent. -Heredity Patients with a family history of prostate cancer are more likely to develop it than are patients without a family history. ______STAGES OF PROSTATE CANCER_____________________________________________ Stage A. The carcinoma is confined to the prostate, and is not palpable (detectable) by Digital Rectal Examination (DRE). Patients usually do not complain about symptoms, and it can only be detected by elevated PSA levels or a biopsy. Stage B. The cancer is confined to the prostate, but is palpable by DRE. There are usually no symptoms. Stage C. In this stage, the carcinoma has spread to regions outside the prostate. Patients usually experience some pain. Stage D. The carcinoma has spread into the spinal column, causing back pain in addition to the pains found in stage C. ______DIAGNOSIC TESTS_______________________________________________________ -Digital Rectal Examination (DRE) Your doctor inserts a gloved, lubricated finger into the rectum and feels for any abnormalities in the prostate. -Prostate Specific Antigen (PSA) Test PSA is a chemical that is produced only by the prostate. A high PSA level in your blood suggests that your prostate is enlarged. ______METHOD OF DIAGNOSIS___________________________________________________ Clinicians can occasionally make the diagnosis by careful observation. However, the removal of a small sample from the affected area for analysis at a reputable pathology laboratory was required for absolute confirmation of the diagnosis, as other conditions can mimic it clinically. Your sample was studied by a specialized pathologist at Acupath Laboratories, Inc. before being conclusively diagnosed as Prostate Cancer. ______METHODS OF TREATMENT__________________________________________________ Prostate adenocarcinoma usually requires surgery. However, other non-surgical methods are appropriate in some cases. Your doctor will determine which treatment is best suited for your specific case. ______COMMENTS______________________________________________________________
For Site(s): C, D, H, J, L
Provided as a service by Acupath Laboratories in cooperation with your doctor. © Acupath Laboratories, Inc. "For the absolute highest standard in pathology services."

(212) 123-4567 Dr. JOHN U. SMITH Doctor:
01/02/19XX Date of Birth:
09/18/20XX Date Obtained:
JACK S JONES Patient:
PATIENT FACT SHEET TM
BENIGN PROSTATIC HYPERPLASIA (BPH) Definition: A non-cancerous enlargement of the prostate that often begins around age 45 and can continue indefinitely. The prostate is a gland that surrounds the male urethra and secretes a fluid during ejaculation. As the prostate grows, it begins to pinch the urethra, causing the symptoms associated with BPH. There are many causes of an enlarged prostate, and it is important that BPH be distinguished from prostate cancer. _____FACTS________________________________________________ -BHP may be the result of the hormone dihydrotestosterone (DHT), which triggers the enlargement of the prostate. -BHP affects 50% of men age 60 or older, and 90% of men age 85 or older. -Most cases of an enlarged prostate are BPH, not cancer. _____SYMPTOMS_____________________________________________ -Frequent urination -Weak or interrupted urinary stream -Feeling that bladder will not empty completely -Feeling of delay or hesitation when you begin to urinate -Feeling that you cannot postpone urination -Pain in lower back, pelvis, and/or upper thigh -Burning pain during urination sometimes indicates that an infection is present _____DIAGNOSIC TESTS________________________________________ -Digital Rectal Examination (DRE) Your doctor inserts a gloved, lubricated finger into your rectum and feels for an enlargement of the prostate. _____METHOD OF DIAGNOSIS_____________________________________ Clinicians can occasionally make the diagnosis by careful observation. However, the removal of small samples from the affected area for analysis at a reputable pathology laboratory is required for absolute confirmation of the diagnosis, as other more serious conditions can mimic it clinically. Your sample is studied by a specialized pathologist at Acupath Laboratories, Inc. before being conclusively diagnosed as a Benign Prostatic Hyperplasia. _____METHODS OF TREATMENT____________________________________ There are various surgical and non-surgical methods of treating BPH, including several technologically advanced methods. Each method is applicable for certain specific cases, and your doctor will determine which method is best suited for you. _____COMMENTS_____________________________________________
For Site(s): A, B, G
Provided as a service by Acupath Laboratories in cooperation with your doctor. © Acupath Laboratories, Inc. "For the absolute highest standard in pathology services."

DR. JANE U. DOE 456 SAMPLE BLVD. NEW YORK, NY 10011
UROPATHOLOGY REPORT Batch#: 24724 1 of 2STPLIS;RL;MMR;
(212) 123-4568 (212) 123-4567
Suite: 8A NEW YORK, NY 10016 (718) 555-2541
ABC SURGERY CENTER
Acct # 352-549-2
PHYSICIAN: JOHN U. SMITH
DOB:
Age:
01/02/19XX
58
PATIENT: JACK JONES
FLUSHING, NY 11365
Male 1018 Acct:
Sex:
Accession:
Received: Obtained: 09/18/20XX
09/18/20XX 6:07 pm
000000-20XX
Final Report Date Preliminary Date Specimen
N/A
09/19/XX
A
N/A
09/19/XX
B
09/19/XX
09/22/XX
C
09/19/XX
09/22/XX
D
09/19/XX
09/22/XX
E
09/19/XX
09/22/XX
F
N/A
09/19/XX
G
09/19/XX
09/22/XX
H
N/A
09/19/XX
I
09/19/XX
09/22/XX
J
09/19/XX
09/22/XX
K
09/19/XX
09/22/XX
L
12548 MAIN ST
NEW YORK, NY 10011 456 SAMPLE BLVD. DR. JANE U. DOE
28 South Terminal Drive
Tel#:
www.acupath.com Fax#:
1-888-ACUPATH 1-516-326-3452
Plainview, NY 11803
1234 FIRST AVE
6 Rte
Duplicate Report For: Submitted: 12 vials-A,B,C,D,E,F,G,H,I,J,K,L
Comments: 790.93
C, D, J, L: ADENOCARCINOMA OF THE PROSTATE. There is a gland forming neoplasm in prostatic stroma composed of neoplastic epithelial cells with enlarged nuclei and prominent nucleoli. E, F, K: ADENOCARCINOMA OF THE PROSTATE WITH FOCAL PERINEURAL INVASION. Sections reveal a gland forming neoplasm adjacent to a nerve. H: SMALL FOCUS OF ADENOCARCINOMA OF THE PROSTATE. There is a gland forming neoplasm in prostatic stroma composed of neoplastic epithelial cells with enlarged nuclei and prominent nucleoli. A: BENIGN PROSTATIC HYPERPLASIA. There is hyperplasia of glands and surrounding fibrous stroma. B, G: BENIGN PROSTATIC HYPERPLASIA WITH ATROPHIC FEATURES. There is focal hyperplasia of glands and surrounding fibrous stroma. Other areas reveal dilated glands with atrophic epithelium. I: FIBROMUSCULAR STROMA ONLY. This specimen consists of fibromuscular stroma only. No glandular elements are seen.
DIAGNOSIS & MICROSCOPIC DESCRIPTION
GLEASONS SCORE & % OF BIOPSY REPLACED BY CARCINOMA
A B C 6 (3+3)
10%
D 6 (3+3)
5%
E 7 (3+4)
60%
F7 (3+4)
50%
G H6 (3+3)
2%
I J 7 (3+4)
50%
K7 (3+4)
60%
L6 (3+3)
90%
RLA(I)
BENIGN
GS:6(3+3)
RLM(H)
RLB(G)
BENIGN
2%
RB(A)
RM(B)
RA(C)
GS:6(3+3)
BENIGN
BENIGN
10%
LB(D)
LM(E)
LA(F)
GS:7(3+4)
GS:7(3+4)
GS:6(3+3)
5%
60%
50%
LLB(J)
LLM(K)
LLA(L)
GS:7(3+4)
GS:7(3+4)
GS:6(3+3)
50%
60%
90%
Color Key
Photomicrograph of J
Adenocarcinoma Benign Infectious Inflammatory Suspicious Pin Other No Diag Atypical
John Brown, M.D.
Continued on next page
Olga Falkowski, M.D., Medical Director
JACK JONES 09/18/20XX JACK JONES
RLB(G)
RLM(H)
RLA(I)
BENIGN
GS:6(3+3)
BENIGN
RB(A)
RM(B)
RA(C)
BENIGN
BENIGN
GS:6(3+3)
LB(D)
LM(E)
LA(F)
GS:6(3+3)
GS:7(3+4)
GS:7(3+4)
LLB(J)
LLM(K)
LLA(L)
GS:7(3+4)
GS:7(3+4)
GS:6(3+3)
6(3+3) 6(3+3) 7(3+4) 7(3+4)
6(3+3) 7(3+4)
A B C D E F
G H I J
JACK JONES 09/18/20XX
6(3+3) 6(3+3) 7(3+4) 7(3+4)
6(3+3) 7(3+4)
JACK JONES A B C D E F
G H I J
Note: Prostate diagrams and labels indicate disease summaries only. See diagnosis above for details
K L
K L
7(3+4) 6(3+3)
7(3+4) 6(3+3)
John Brown

2 of 2JACK JONES PATIENT: 000000-20XX Accession:
www.acupath.com 1-516-326-3452 Fax#: 1-888-ACUPATH Tel#:
Plainview,NY 11803 28 South Terminal Drive
Final Date
Preliminary Date
Specimen
N/A
A
09/19/XX
N/A
B
09/19/XX
09/19/XX
C
09/22/XX
09/19/XX
D
09/22/XX
09/19/XX
E
09/22/XX
09/19/XX
F
09/22/XX
N/A
G
09/19/XX
09/19/XX
H
09/22/XX
N/A
I
09/19/XX
09/19/XX
J
09/22/XX
09/19/XX
K
09/22/XX
09/19/XX
L
09/22/XX
NOTESC: Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals weak staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 reveals no evidence of perineural invasion. D: Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals weak staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 reveals no evidence of perineural invasion. E: Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals moderate staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 is positive, supporting the diagnosis of perineural invasion.
F: Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals moderate staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 is positive, supporting the diagnosis of perineural invasion.
H: Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals weak staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 reveals no evidence of perineural invasion. J: Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals moderate staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 reveals no evidence of perineural invasion. K: Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals moderate staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 is positive, supporting the diagnosis of perineural invasion.
L: Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals weak staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 reveals no evidence of perineural invasion.
This test was developed and its performance characteristics determined by Acupath Laboratories, Inc. It has not been cleared or approved by the U.S. Food and Drug Administration. The FDA has determined that such clearance or approval is not necessary. This test is used for clinical purposes. It should not be regarded as investigational or for research. This laboratory is certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA) as qualified to perform high complexity clinical laboratory testing.
Fixative Color Shape Cassette Piece Measurement(mm)
GROSSING INFORMATION
Site
10% Formalin white Cylindrical 1 07x01x01 A Right Base 2
10% Formalin white Cylindrical 1 09x01x01 B Right Mid 1
10% Formalin white Cylindrical 1 09x01x01 C Right Apex 1
10% Formalin white Cylindrical 1 10x01x01 D Left Base 1
10% Formalin white Cylindrical 1 10x01x01 E Left Mid 1
10% Formalin white Cylindrical 1 08x01x01 F Left Apex 1
10% Formalin white Cylindrical 1 04x01x01 G Right Lateral Base 1
10% Formalin white Cylindrical 1 05x01x01 H Right Lateral Mid 1
10% Formalin white Cylindrical 1 04x01x01 I Right Lateral Apex 1
10% Formalin white Cylindrical 1 07x01x01 J Left Lateral Base 1
10% Formalin white Cylindrical 1 09x01x01 K Left Lateral Mid 1
10% Formalin white Cylindrical 1 10x01x01 L Left Lateral Apex 1

ABC SURGERY CENTER JOHN U. SMITH, M.D.
1234 First Avenue New York, NY 10016
212-123-4567
September 22, 20XX
Dear Dr. U. Doe:
Re: Jack S. Jones456 Sample Blvd. New York, NY 10011
Dr. Jane U. Doe
I had the pleasure of treating your patient Jack S Jones, a 58 year old male, on September 18, 20XX. The specimens were sent for pathologic interpretation to Acupath Laboratories, Inc. The histopathologic interpretation of the specimen from the (A) Right Base revealed benign prostatic hyperplasia. The histopathologic interpretation of the specimen from the (B) Right Mid revealed benign prostatic hyperplasia with atrophic features. The histopathologic interpretation of the specimen from the (C) Right Apex revealed adenocarcinoma of the prostate. Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals weak staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 reveals no evidence of perineural invasion. The histopathologic interpretation of the specimen from the (D) Left Base revealed adenocarcinoma of the prostate. Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals weak staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 reveals no evidence of perineural invasion. The histopathologic interpretation of the specimen from the (E) Left Mid revealed adenocarcinoma of the prostate with focal perineural invasion. Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals moderate staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 is positive, supporting the diagnosis of perineural invasion. The histopathologic interpretation of the specimen from the (F) Left Apex revealed adenocarcinoma of the prostate with focal perineural invasion. Keratin 903 is absent from glandular basal cells, supporting the diagnosis of carcinoma. IHC stain for P63 reveals no evidence of basal myoepithelial cells favoring a diagnosis of adenocarcinoma. IHC stain for P504S is positive, supporting the diagnosis of adenocarcinoma. IHC stain for CPP32 reveals moderate staining consistent with the Gleason's grade. IHC stain for Ki67 is negative. IHC stain for S100 is positive, supporting the diagnosis of perineural invasion. See report for more.
I would like to personally thank you for your kind referral. If you have any additional questions, please do not hesitate to contact me at 212-123-4567.
Sincerely,
JOHN U. SMITH, M.D.

CYTOPATHOLOGY REPORT 1 of 1*RL;MMR;Batch#: 28369
Accession:
Received: Obtained: 04/02/20XX
04/02/20XX 10:24 pm
Final Report Date
Specimen
04/03/XX
A
DR. JANE U. DOE 456 SAMPLE BLVD. NEW YORK, NY 10011
12548 MAIN ST
Age:
FLUSHING, NY 11365 Acct#:
PATIENT: JACK JONES
1018 59 DOB:
(718) 555-2541 01/02/19XX
Male Sex:
ABC SURGERY CENTER PHYSICIAN: JOHN U. SMITH
NEW YORK, NY 10016 1234 FIRST AVE, Suite: 8A
6 (212) 123-4568352-549-2 Acct # Rte (212) 123-4567
28 South Terminal Drive
Tel#:
www.acupath.com Fax#:
1-888-ACUPATH Plainview, NY 11803
1-516-326-3452
Duplicate Report was faxed to: 000000-20XX
Client Comments:
DIAGNOSIS
A Urine Cytology
DIAGNOSIS: POSITIVE FOR MALIGNANT CELLS, CONSISTENT WITH HIGH GRADE UROTHELIAL CARCINOMA
Microscopic Description: Thin prep slide reveals single urothelial cells with high N/C ratio and hyperchromatic nucleus.
ADEQUACY: SATISFACTORY FOR EVALUATION. Rec'd 10ml of fixed clear urine in Thin Prep vial. Prepared 1 Thin Prep slide.
JOHN BROWN, M.D.
Photomicrograph of A
Color KeyCancer BenignSuspiciousOther AtypicalNon-Diagnostic
Diagram not indicated
Olga Falkowski, M.D., Medical Director
A: (Urine Cytology) POSITIVE FOR MALIGNANT CELLS, CONSISTE...See Report
A: (Urine Cytology) POSITIVE FOR MALIGNANT CELLS, CONSISTE...See Report
000000-20XX
JACK JONES
000000-20XX
(718) 555-2541 01/02/19XX 04/02/20XX
JACK JONES 000000-20XX
(718) 555-2541 01/02/19XX 04/02/20XX04/02/20XX
JACK JONES
JACK JONES
Submitted: 1 vial-A
Submitted: 1 vial-A DOB:
DOB:
Page 1 of 1
John Brown

FISH ANALYSIS REPORT Page 1 of 1 RL;MMR;
(212) 123-4568(212) 123-4567
Suite: 8A NEW YORK, NY 10016
(516) 123-4567
ABC SURGERY CENTER
Account # 352-549-2
PHYSICIAN: JOHN U. SMITH
DOB:Age: 06/05/19XX 80
BETSY JONES
ANYWHERE, NY 11554
Female 1012 Acct:Sex:
ACCESSION:
Received: Obtained:
6 Rte
1234 ANYWHERE
28 South Terminal Drive
Tel#:
www.acupath.com Fax#:
1-888-ACUPATH 1-516-326-3452
Plainview, NY 11803
1234 FIRST AVE
PRACTICE: PATIENT:
Phone #:
NEW YORK, NY 10011 456 SAMPLE BLVD. DR. JANE U. DOE A Copy was sent to:
12/13/20XX
12/12/20XX 000000-20XX
Indications:
Date Reported FISH:
Urine Source of Tissue: Bladder Cancer
12/14/20XX 12:48 PM
FISH ANALYSIS DATA
Assay # 1 Probe Region: Chromosomes 3, 7, 17 and 9p21 25 No. of Cells Scored: Process and limitations of use: This test was developed and its performance characteristics determined by Acupath Laboratories. It has not been cleared or approved by the U.S. Food and Drug Administration.
TEST RESULT: Abnormal- Positive Aneuploidy of chromosomes 3, 7, 17 and/or 9p21 locus. A positive control had all 25 cells with an abnormal hybridization pattern.
INTERPRETATION: The Vysis UroVysion Bladder Cancer FISH assay was performed on the analyzable cells from the patient’s voided urine. A minimum of twenty-five morphologically abnormal cells were analyzed. Of these cells, 25 of 25 (100%) were determined to have an abnormal profile. A review of the entire specimen slide revealed ten cells which were aneuploid for chromosomes 3, 7, 17 and/or the 9p21 locus. Aneuploidy of these chromosomes is associated with urothelial carcinoma progression. In addition, sixty other cells were observed which were tetraploidy/ near tetraploidy for chromosomes 3,7,17, and/or the 9p21 locus. This is greater than the 10 cells needed to consider the specimen abnormal by tertaploidy/ near tetraploidy (Bubendorf et al., Am J Clin Pathol 2001; 116:79-86). These results should be correlated with other clinical and laboratory tests.
FISH
Electronically signed by: John Brown, MD, Director of Genetics Olga Falkowski, M.D., Medical Director

PROSTATE FISH ANALYSIS REPORT Page 1 of 2 IS;RL; STPL
(212) 555-1112 (212) 555-1111
Suite: 100 ANYWHERE, NY 10000
(212) 555-1234
ABC UROLOGY
Account # 0000-000-1
PHYSICIAN: JOHN SMITH
DOB: Age: 08/17/19XX 69
JOHN DOE
ANYWHERE, NY 10000
Male 000001 Acct: Sex:
ACCESSION:
Received: Obtained:
2 Rte
123 STATE STREET
28 South Terminal Drive Tel#: www.acupath.com Fax#:
1-888-ACUPATH 1-516-326-3452
Plainview, NY 11803
123 ANY STREET
PRACTICE: PATIENT:
Phone #:
08/06/20XX 08/01/20XX
000001-20XX
Source of Tissue: Prostate Biopsy Indications: Adenocarcinoma; PTEN FISH: ERG FISH
Date Reported FISH: 08/09/20XX 3:16 PM
ABNORMAL: LOSS OF HMGN1 AND DSCAM, RESULTING IN ERG REARRANGEMENTS IN 33% OF THE CELLS:
HETEROZYGOUS LOSS OF THE PTEN, FAS AND WAPAL LOCI IN 28% OF THE CELLS ANALYZED.
FINAL DIAGNOSIS:
Assay # FISH ANALYSIS DATA
Probe Region Chromosome or Locus Normal Reference Range
No. of Cells Scored
Normal
Percentage 1 100 DSCAM (21q22.2) 67% 90-100% ERG:TMPRSS2 Panel 1 100 ERG (21q22.2) 67% 90-100% ERG:TMPRSS2 Panel 1 100 HMGN1 (21q22.2) 67% 90-100% ERG:TMPRSS2 Panel 1 100 TMPRSS2 (21q22.2) 67% 90-100% ERG:TMPRSS2 Panel 2 100 CEP10 (10p11.1-q11.1) 100% Hetero. > 79%, Homo. > 69% PTEN-del-TECT Panel 2 100 FAS (10q23.31) 72% Hetero. > 79%, Homo. > 69% PTEN-del-TECT Panel 2 100 PTEN (10q23.31) 72% Hetero. > 79%, Homo. > 69% PTEN-del-TECT Panel 2 100 WAPAL (10q23.31) 72% Hetero. > 79%, Homo. > 69% PTEN-del-TECT Panel
FISH
nuc ish(ERGx2,HMGN1x1,DSCAMx1,TMPRSS2x1~2)[33/100],(CEP10x2,WAPALx1,PTENx1,FASx1)[28/100] ISCN RESULT:
Interphase fluorescence in situ hybridization (FISH) was completed on the submitted formalin fixed paraffin embedded lower prostate biopsy (part I), using a panel of four locus specific identifier probes for the following loci: ERG (centromeric), HMGN1, DSCAM, and TMPRSS2 (telomeric), all located in the long arm of chromosome 21 (21q22.2). One hundred non overlapping cells were identified, of which 33/100 (33%) showed a heterozygous loss of both the HMGN1 and DSCAM, and TMPRSS loci – resulting in the ERG locus rearranging with an unknown fusion partner. This is greater than the 10% of the tissue needed with an ERG rearrangement to consider the specimen abnormal. These are ABNORMAL results. Patients with ERG rearrangements and with loss of PTEN are associated with a poor prognosis (Reid et al. British Journal of Cancer (2010) 102, 678-684). In addition, FISH was also completed using a panel of three locus specific identifier probes and one chromosome enumeration probe for the following loci: CEP10 (10p11.1-q11.1), telomeric FAS (10q23.31), PTEN (10q23.31), and centromeric WAPAL (10q23.31). A minimum of one hundred non overlapping cells were identified. Of these, 28/100 cells showed a heterozygous loss of the PTEN locus as well as a heterozygous loss of the WAPAL and FAS loci - showing the deletion is more likely the whole arm. This is greater than the 20% of the tissue needing to have the heterozygous loss of the PTEN locus to consider the specimen abnormal. The heterozygous PTEN deletion is associated with a much earlier onset of biochemical recurrence (Yoshimoto M, et al. Br J Cancer (2007) p678-685).
INTERPRETATION:
Process and limitations of use:
Electronically signed by: John Brown, M.D. Olga Falkowski, M.D., Medical Director

PROSTATE FISH ANALYSIS REPORT Page 2 of 2 STPL
(212) 555-1112 (212) 555-1111
Suite: 100 ANYWHERE, NY 10000
(212) 555-1234
ABC UROLOGY
Account # 0000-000-1
PHYSICIAN: JOHN SMITH
DOB: Age: 08/17/19XX 69
JOHN DOE
ANYWHERE, NY 10000
Male 000001 Acct: Sex:
ACCESSION:
Received: Obtained:
2 Rte
123 STATE STREET
28 South Terminal Drive Tel#: www.acupath.com Fax#:
1-888-ACUPATH 1-516-326-3452
Plainview, NY 11803
123 ANY STREET
PRACTICE: PATIENT:
Phone #:
08/06/20XX 08/01/20XX
000001-20XX
Source of Tissue: Prostate Biopsy Indications: Adenocarcinoma; PTEN FISH: ERG FISH
Date Reported FISH: 08/09/20XX 3:16 PM
1. The PTEN FISH assay is not an FDA-approved FISH test. It is designed to aid in the detection of chromosomal abnormalities related to patients with Adenocarcinoma of the prostate. This FISH assay is intended for use as an adjunct to traditional diagnostic procedures and should not be used as the sole basis for the diagnosis of new cancers or for the surveillance of tumor recurrence. 2. A positive result by the PTEN FISH assay requires that a total of 100 non overlapping interphase cells be evaluated and that these cells show at least two copies of the CEP10 locus, homozygous loss of PTEN in >30% of the tissue, or heterozygous loss of PTEN in >20% of the tissue. Loss of FAS or WAPAL are included to determine the size of the deletion, and to prevent truncation artifact reporting. 3. This PTEN FISH assay was developed and its performance characteristics determined by the Acupath Cytogenetics Laboratory. Although it has not been cleared or approved by the U.S. Food and Drug Administration, the FDA has determined that such clearance or approval is not necessary. Pursuant to the requirements of CLIA ’88, however, this laboratory has established and verified the test’s accuracy and precision, and is therefore used for clinical purposes. 4. The ERG:TMPRSS2 FISH assay is not an FDA-approved FISH test. It is designed to aid in the detection of chromosomal abnormalities related to patients with Adenocarcinoma of the prostate. This FISH assay is intended for use as an adjunct to traditional diagnostic procedures and should not be used as the sole basis for the diagnosis of new cancers or for the surveillance of tumor recurrence. 5. A positive result by ERG:TMPRSS2 FISH assay requires that a total of 100 non overlapping interphase cells be evaluated and that these cells show at least two copies of at least three of the four loci tested to prevent truncation artifact reporting. Specific cut-off values have been established at Acupath Laboratories, Inc. 6. The ERG:TMPRSS2 FISH assay was developed and its performance characteristics determined by the Acupath Cytogenetics Laboratory. Although it has not been cleared or approved by the U.S. Food and Drug Administration, the FDA has determined that such clearance or approval is not necessary. Pursuant to the requirements of CLIA ’88, however, this laboratory has established and verified the test’s accuracy and precision, and is therefore used for clinical purposes.
Electronically signed by: John Brown, M.D. Olga Falkowski, M.D., Medical Director

MOLECULAR ISH REPORT Page 1 of 1 IS;RL;MMR;
(718) 555-2541 DOB:Age: 01/02/19XX 58
JACK JONES
FLUSHING, NY 11365
Male 1018 Acct:Sex:
ACCESSION:
Received: Obtained: 11/03/20XX
12548 MAIN ST
28 South Terminal Drive
Tel#:
www.acupath.com Fax#:
1-888-ACUPATH 1-516-326-3452
Plainview, NY 11803
PATIENT:
Phone #:
NEW YORK, NY 10011 456 SAMPLE BLVD DR. JANE DOE A Copy was sent to:
000000-20XX
11/05/20XX 12:00 am
Indications: R/O Condyloma Source of Tissue: Penis
Date Reported:
PRACTICE: ABC SURGERY CENTER PHYSICIAN: JOHN U. SMITH
1234 FIRST AVE Suite: 8A NEW YORK, NY 10016
Account # 352-549-2 Rte 6
(212) 123-4567(212) 123-4568
11/13/20XX 2:45PM
TEST RESULT
ISH/Probe Marker For Results
HPV WS Wide Screen HPV Positive (BLOCK B) HPV 6/11 HPV Types 6/11 Positive (BLOCK B) HPV 16/18 HPV Types 16/18 Negative (BLOCK B) HPV 31/33 HPV Types 31/33 Negative (BLOCK B)
INTERPRETATION: Positive for one or more of the following strains of HPV: 6/11/16/18/31/33/35/45/51/52. The HPV Wide Spectrum is used to detect HPV DNA by In Situ Hybridization (ISH) in biopsies; it does not discriminate between the HPV subtype. The HPV Wide Spectrum detects subtypes 6, 11, 16, 18, 30, 31, 33, 35, 45, 51, and 52. The absence of hybridization with HPV subtypes 6, 11, 16, 18, 31, and 33 may indicate other subtypes that were not analyzed in this study. Positive for Low-Risk HPV strains 6/11. Negative for High-Risk HPV strains 16/18. Negative for High-Risk HPV strains 31/33.
This analysis is an adjunct to the evaluation of the referring physician and does not represent a final diagnosis.
Photomicrographs:
HPV WideScreen-Block B HPV 6/11-Block B HPV 16/18 HPV 31/33
Electronically signed by: John Brown, M.D.
This test was developed and its performance characteristics determined by Acupath Laboratories, Inc. It has not been cleared or approved by the U.S. Food and Drug Administration. The FDA has determined that such clearance or approval is not necessary. This test is used for clinical purposes. Pursuant to the requirements of CLIA ’88, this laboratory has established the test’s accuracy and precision. This test has been approved by New York State as a laboratory specific assay. This test cannot be used as sole evidence for or against cancer and has to be interpreted in the context of all available clinical and pathological information.

(212) 123-4567 Dr. JOHN U. SMITH Doctor:
01/02/19XX Date of Birth:
11/03/20XX Date Obtained:
JACK S JONES Patient:
PATIENT FACT SHEET TM
CONDYLOMA ACUMINATA Definition: Commonly known as genital warts, Condyloma Acuminata is a sexually transmitted disease caused by the Human Papilloma Virus (HPV). It is characterized by wart-like growths on the penis, vagina, vulva, and/or rectum. In rarer cases HPV may cause warts on the throat, tongue, and mouth. _____FACTS/RISK FACTORS________________________________________ Over 50 different types of HPV have been identified. Some lead to rough raised warts, while others to soft flat ones. Several types have been associated with abnormal pap smears in women, which may indicate an increased risk for cervical cancer. Unsafe sexual practices such as multiple/unknown sexual partners and lack of condom use increase the risk of contracting HPV. _____SYMPTOMS__________________________________________________ Although many patients display no symptoms at all, infection by HPV may cause: - warts/lesions in the genital and/or anal areas - cauliflower-like growths around the genitals and/or anus - dampness - itching _____METHOD OF DIAGNOSIS_______________________________________ Many clinicians can diagnose Condyloma by careful observation. However, the removal of a small sample from the affected area for analysis at a reputable pathology laboratory is required for absolute confirmation of the diagnosis, as other conditions can mimic it clinically. Your sample is studied by a specialized pathologist at Acupath Laboratories, Inc. before being conclusively diagnosed as Condyloma Acuminata. _____METHODS OF TREATMENT______________________________________ Methods of treatment include both topical medications and minor surgical procedures. Your doctor will decide which type of therapy is best suited for you. _____COMMENTS__________________________________________________
For Site(s): B
Provided as a service by Acupath Laboratories in cooperation with your doctor. © Acupath Laboratories, Inc. "For the absolute highest standard in pathology services."
Related Documents