2010;70:9423-9433. Published OnlineFirst November 9, 2010. Cancer Res Mathieu Darsigny, Jean-Philippe Babeu, Ernest G. Seidman, et al. Species and Protects against the Production of Reactive Oxygen Promotes Gut Neoplasia in Mice α Hepatocyte Nuclear Factor-4 Updated version 10.1158/0008-5472.CAN-10-1697 doi: Access the most recent version of this article at: Material Supplementary http://cancerres.aacrjournals.org/content/suppl/2010/11/05/0008-5472.CAN-10-1697.DC1.html Access the most recent supplemental material at: Cited Articles http://cancerres.aacrjournals.org/content/70/22/9423.full.html#ref-list-1 This article cites by 43 articles, 12 of which you can access for free at: Citing articles http://cancerres.aacrjournals.org/content/70/22/9423.full.html#related-urls This article has been cited by 4 HighWire-hosted articles. Access the articles at: E-mail alerts related to this article or journal. Sign up to receive free email-alerts Subscriptions Reprints and . [email protected] Department at To order reprints of this article or to subscribe to the journal, contact the AACR Publications Permissions . [email protected] Department at To request permission to re-use all or part of this article, contact the AACR Publications Research. on September 16, 2014. © 2010 American Association for Cancer cancerres.aacrjournals.org Downloaded from Published OnlineFirst November 9, 2010; DOI: 10.1158/0008-5472.CAN-10-1697 Research. on September 16, 2014. © 2010 American Association for Cancer cancerres.aacrjournals.org Downloaded from Published OnlineFirst November 9, 2010; DOI: 10.1158/0008-5472.CAN-10-1697

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript
2010;70:9423-9433. Published OnlineFirst November 9, 2010.Cancer Res Mathieu Darsigny, Jean-Philippe Babeu, Ernest G. Seidman, et al. Speciesand Protects against the Production of Reactive Oxygen
Promotes Gut Neoplasia in MiceαHepatocyte Nuclear Factor-4
Updated version
10.1158/0008-5472.CAN-10-1697doi:
Access the most recent version of this article at:
Material
Supplementary
http://cancerres.aacrjournals.org/content/suppl/2010/11/05/0008-5472.CAN-10-1697.DC1.html
Access the most recent supplemental material at:
Cited Articles
http://cancerres.aacrjournals.org/content/70/22/9423.full.html#ref-list-1
This article cites by 43 articles, 12 of which you can access for free at:
Citing articles
http://cancerres.aacrjournals.org/content/70/22/9423.full.html#related-urls
This article has been cited by 4 HighWire-hosted articles. Access the articles at:
E-mail alerts related to this article or journal.Sign up to receive free email-alerts
Subscriptions
Reprints and
To order reprints of this article or to subscribe to the journal, contact the AACR Publications
Permissions
To request permission to re-use all or part of this article, contact the AACR Publications
Research. on September 16, 2014. © 2010 American Association for Cancercancerres.aacrjournals.org Downloaded from
Published OnlineFirst November 9, 2010; DOI: 10.1158/0008-5472.CAN-10-1697
Research. on September 16, 2014. © 2010 American Association for Cancercancerres.aacrjournals.org Downloaded from
Published OnlineFirst November 9, 2010; DOI: 10.1158/0008-5472.CAN-10-1697
Tum
HepMicRea
MathiEmile
Abst
Intro
Hepreceptsmalldenceendogconceas a nof targhave bdiseasthe Hsevera
AuthorDigestiFacultéSherbroUniverSte-Jus
Note:Resear
Correset de BSanté,Phone:usherbr
doi: 10
©2010
www.a
D
Published OnlineFirst November 9, 2010; DOI: 10.1158/0008-5472.CAN-10-1697
Canceresearch
or and Stem Cell Biology
atocyte Nuclear Factor-4α Promotes Gut Neoplasia ine and Protects against the Production of
R
ctive Oxygen Species
eu Darsigny1, Jean-Philippe Babeu1, Ernest G. Seidman2, Fernand-Pierre Gendron1,
Levy3, Julie Carrier1, Nathalie Perreault1, and François Boudreau1ractHep
phogelium inshowmice.Analysnovelof reationallof ROSsignifineutra
NF4A genl forms o
s' Affiliatiove Epitheliude Médeoke, Sherbsity Healttine, Univer
Supplemench Online (h
ponding Auiologie Ce3001 12e819-820-68ooke.ca.
.1158/0008-
American A
acrjourna
ownloade
atocyte nuclear factor-4α (Hnf4α) is a transcription factor that controls epithelial cell polarity and mor-nesis. Hnf4α conditional deletion during postnatal development has minor effects on intestinal epithe-tegrity but promotes activation of the Wnt/β-catenin pathway without causing tumorigenesis. Here, wethat Hnf4α does not act as a tumor-suppressor gene but is crucial in promoting gut tumorigenesis inPolyp multiplicity in ApcMin mice lacking Hnf4α is suppressed compared with littermate ApcMin controls.is of microarray gene expression profiles from mice lacking Hnf4α in the intestinal epithelium identifiesfunctions of this transcription factor in targeting oxidoreductase-related genes involved in the regulationctive oxygen species (ROS) levels. This role is supported with the demonstration that HNF4α is func-y involved in the protection against spontaneous and 5-fluorouracil chemotherapy-induced productionin colorectal cancer cell lines. Analysis of a colorectal cancer patient cohort establishes that HNF4α is
cantly upregulated compared with adjacent normal epithelial resections. Several genes involved in ROSlization are also induced in correlation with HNF4A expression. Altogether, the findings point to the
nuclear receptor HNF4α as a potential therapeutic target to eradicate aberrant epithelial cell resistance toROS production during intestinal tumorigenesis. Cancer Res; 70(22); 9423–33. ©2010 AACR.
Polymassoc(3, 4)identisuscepOne
specifthe usimpormousembrycells o
duction
atocyte nuclear factor-4α (HNF4α) is an orphan nuclearor expressed in the liver, kidney, pancreas, stomach,intestine, and colon (1). Recent experimental evi-has led to the identification of linoleic acid as anenous and reversible HNF4α ligand, reinforcing thept that this nuclear receptor could be consideredutritional and pharmaceutical target (2). The strategyeting HNF4α has gained interest because many linkseen established between HNF4A integrity and humanes. Indeed, a number of mutations identified within
e are considered to represent the causes off maturity-onset diabetes of the young (3).
with icreatidepenwork (liver ctheliutrollinHnf4αand is(10, 12to beduringis depmentdid nlong-t
ns: 1Canadian Institute of Health Research Team onm, Département d'Anatomie et Biologie Cellulaire,cine et des Sciences de la Santé, Université derooke, Québec, Canada; 2Research Institute, McGillh Center; and 3Department of Nutrit ion, CHUsité de Montréal, Montréal, Québec, Canada
tary data for this article are available at Cancerttp://cancerres.aacrjournals.org/).
thor: François Boudreau, Département d'Anatomiellulaire, Faculté de Médecine et des Sciences de laAvenue Nord, Sherbrooke, QC, Canada, J1H 5N4.76; Fax: 819-564-5324; E-mail: francois.boudreau@
5472.CAN-10-1697
ssociation for Cancer Research.
ls.org
Research. on September 16, 2cancerres.aacrjournals.org d from
orphisms within the HNF4A promoter region areiated with an increased risk of type 2 diabetes. Moreover, genome-wide association studies havefied the HNF4A region as a novel ulcerative colitistibility locus (5).powerful way of defining the functional relevance of a
ic transcriptional regulator in a given tissue is throughe of conditional gene deletion. This was particularlytant for Hnf4α for which classic gene deletion in thee resulted in severe gastrulation defects with earlyonic lethality (6). Conditional deletion of Hnf4α in βf the mouse pancreas resulted in hyperinsulinemiampaired glucose tolerance (7). Expansion of adult pan-c β-cell mass in response to metabolic demand isdent on a specific Hnf4α-dependent transcription net-8). Conditional genetic removal of Hnf4α in the mouseonfirmed its crucial role in hepatic functions and epi-m integrity (9, 10). The specific role of Hnf4α in con-g intestinal epithelial functions has emerged recently.can polarize intestinal epithelial cells in culture (11)potent for the epithelialization of different cell types, 13). Maturation of colonic epithelial cells was showndependent on Hnf4α when conditionally removedearly embryonic colonic development (14). This effectendent on the early phases of embryonic develop-because colonic epithelial deletion at later stages
ot severely impair epithelial functions but led toerm chronic inflammatory defects resembling human9423
014. © 2010 American Association for Cancer
inflamdeletioon theThe ubinatitionalto thepolypoInte
the Wtaininregulamaint(19). Alead tin theactivaprolife(20). Rstemcell–spneoplaWit
hypothof tumpressefied Hgenesexpresprodusuppoing inttion a
Mate
Cell cHT-
were g10% feL-alan25 mmtomycbeforecontaCATGcontroCACCprotei2',7'-diDCF-D2.5 μmAxioveAxioCthe saof HCwere g
at 16,mentof 12.5by usicount
MiceApc
lect R12.4KJacksoHnf4αgotes)on a pmined
ImmuImm
chemiAnti–P2 fro(Neom(Cell S
RNA eRNA
PCR (PrimeFor m430 2.and th
Darsigny et al.
Cance9424
D
Published OnlineFirst November 9, 2010; DOI: 10.1158/0008-5472.CAN-10-1697
matory bowel diseases (15). Hnf4α conditionaln in the small intestine led to minor consequencesoverall integrity of the epithelium structure (13, 16).
se of an inducible tamoxifen-dependent Cre recom-on system led to similar conclusions with the addi-observation that loss of Hnf4α was contributingactivation of the Wnt/β-catenin without causingsis (17).stinal cancer is initiated from mutations that activatent pathway (18). This pathway is responsible in main-g the nuclear-activated status of the transcriptionaltory β-catenin/Tcf-4 complex and contributes to theenance of the crypt stem cell/progenitor phenotypedenomatous polyposis coli (APC) gene mutations
o the upregulation of the Wnt signaling that resultsstabilization of the β-catenin/Tcf-4 complex and
tion of gene targets important in the control of cellration and the initiation of intestinal tumorigenesisecent evidence has described intestinal cancer as acell disease, with the demonstration that the stemecific loss of Apc was the major source of intestinalsia growth (21).h the use of the ApcMin mouse model, we tested theesis that Hnf4α could act as an intestinal suppressorors. Surprisingly, we found that loss of Hnf4α sup-d polyposis in this specific genetic context. We identi-NF4α as a positive regulator of oxidoreductase-relatedand established a functional link between HNF4αsion and restriction of reactive oxygen species (ROS)ction in human colon cancer cells. These observationsrt a novel and unsuspected role for HNF4α in facilitat-
estinal cancer initiation as well as intracellular protec- data abioinfat Gen
EpithmalonHnf
sacrifiby thDickinmalonprevioa 10%supernthiobacoolinextracat 37°Cmethaperfor
TumoSam
haveresect
gainst cancer-related ROS production.
rials and Methods
ulture and treatments29 [American Type Culture Collection (ATCC)] cellsrown in McCoy's growth medium supplemented withtal bovine serum (Wisent), 4.5 g/L D-glucose, 2 mmol/Lyl-L-glutamine (GlutaMAX, Life Technologies),ol/L HEPES, 50 U/mL penicillin, and 50 μg/mL strep-in. Cells were infected at 80% confluence, as described(22), with the pLKO.1-puro lentiviral vector (Sigma)
ining a shRNA against all HNF4α isoforms (5′-CCA-TACTCCTGCAGATT-3′) and a nontarget shRNAl vector used as control (5′-CAACAAGATGAAGAG-AA-3′). Five days postinfection, cells were used forn and RNA extracts, or the 5-(and-6)-chloromethyl-chlorodihydrofluorescein diacetate, acetyl ester (CM-A) ROS probe (Molecular Probes) was added atol/L for 30 minutes at 37°C and visualized on a Zeissrt 200M inverted microscope equipped with a Zeissam HR camera. HCT116 (ATCC) cells were grown inme growing medium as HT-29 cells. Stable populations
T116 cells expressing pBabe Hnf4α1 or empty vectorenerated as described elsewhere (13). Cells were seededTissuesent, a
r Res; 70(22) November 15, 2010
Research. on September 16, 2cancerres.aacrjournals.org ownloaded from
000 cells/cm2 and cultured for 2 days before treat-with 5-fluorouracil (5-FU; Sigma) at a concentrationμmol/L. ROS generation was measured 18 hours later
ng a CM-DCF-DA probe. Positive cells per field wereed using ImageJ 1.41 software (NIH).
and adenoma countsMin mice on a C57BL/6J background were used to col-NA and tissue samples as described elsewhere (13).bVilCre transgenic mice (23) and Hnf4αtm1Gonz (Then Laboratory; ref. 9) were used to produce ApcMin;+/+(control), ApcMin; 12.4KbVilCre/Hnfαfx/+ (heterozy-, and ApcMin;12.4KbVilCre/Hnf4αfx/fx (Hnf4αΔIEC) miceure C57BL/6J background. Polyp counts were deter-as described previously (22).
nodetectionsunoblotting, immufluorescence, and immunohisto-stry were conducted as described elsewhere (13, 15).β-actin and anti-HNF4α that recognizes both P1 andm Santa Cruz were used for immublotting. Anti-Ki67arkers), anti-HNF4α (R&D), and anti–active-caspase-3ignalling) were used in immunofluorescence.
xtraction, microarray, and quantitative PCRwas extracted and quantitative reverse transcriptase-
qRT-PCR) was conducted as previously described (13).r sequences are available in Supplementary Materials.icroarray analysis, Affymetrix GeneChip Mouse Genome0 arrays were used to compare three Hnf4αΔIEC miceree appropriate controls. FlexArray 1.3 was used fornalysis (http://qc.gqinnovationcenter.com/services/ormatics/flexarray/index.aspx?l=e). Data are availablee Expression Omnibus (accession number GSE20968).
elial cell isolation anddialdehyde measurement4αmutant and control mice were fasted overnight andced. Epithelial cells from the jejunumwere then isolatede MatriSperse (Collaborative Biomedical, Bectonson) technique as described previously (24). Freedialdehyde (MDA) levels were determined as reportedusly (25). Briefly, proteins were first precipitated withNaWO4 as described elsewhere (26). Protein-freeatant was then reacted with an isovolume of 0.5%rbituric acid solution at 95°C for 30 minutes. Afterg to room temperature, the pink chromogene wasted with n-butanol and dried over a stream of nitrogen. The dry extract was then resuspended in the KH2PO4/nol (70:30) mobile phase before MDA detection by high-mance liquid chromatography.
r collectionples of colon cancer and paired normal colon tissuesbeen obtained from 41 patients undergoing surgicalion. Patients did not receive neoadjuvant therapy.
s were obtained after patient's written informed con-ccording to the protocol approved by the InstitutionalCancer Research
014. © 2010 American Association for Cancer
HumaUnivernitrogby theparaff(13). Pprotei
ChromquantT84
C57BLper asitationmanuwith 4
was evFinal Dand elwas dtics) osequen
StatisGro
cept fwas uused idistribducted
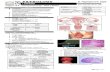
FigureHnf4α ((white afrom small intestine and colon epithelium of C57BL/6J-APC animals. Hnf4α expression is reduced in the polyp (P) and absent from Ki67 cells inopposit whead
Hnf4α Promotes Gut Neoplasia
www.a
D
Published OnlineFirst November 9, 2010; DOI: 10.1158/0008-5472.CAN-10-1697
n Subject Review Board of the Centre Hospitaliersitaire de Sherbrooke. All tissues were frozen in liquiden within 15 minutes from resection as recommendedCanadian Tumor Repository Network. Tissues were
in embedded and immunostained as described beforeaired tissues were used for total RNA isolation (13) orn isolation (27).
atin immunoprecipitation anditative PCRand cells obtained by jejunal mucosa scrapings of/6J mice were used at a concentration of 2 × 106 cellssay. Cells were sonicated and chromatin immunoprecip-(ChIP) was performed with EZ-Chip as described by the
ion to margin tissue (M). Some cells retained Hnf4α expression (white arro
facturer (Millipore). HNF4α was immunoprecipitatedμg of anti-HNF4α from Santa Cruz, and background
cant (expres
acrjournals.org
Research. on September 16, 2cancerres.aacrjournals.org ownloaded from
aluated with 4 μg of normal goat IgG from Santa Cruz.NA was purified with High Pure PCR Cleanup (Roche)
uted in 150 μL of elution buffer. Quantitative PCR (qPCR)one with a LightCycler apparatus V2.0 (Roche Diagnos-n 2 μL of precipitated DNA in each reaction. Primerces are available in Supplementary Materials.
tical analysisups were compared with unpaired Student's t test ex-or tumor collection for which paired Student's t testsed for comparisons. Wilcoxon signed-rank test wasn ChIP experiments to accommodate the non-Gaussianution within all replicates. Correlation tests were con-with Spearman test. P < 0.05 was considered signifi-
s). Scale bars, 100 μm.
1. Hnf4α distribution in the mouse intestinal epithelium. A, immunofluorescence of small intestine and colon tissues using an antibody againstred) and the Ki67 proliferative marker (green). Ki67+ proliferative cells are positive for Hnf4α (turquoise arrowheads). Some Ki67− cells expressedrrowheads) or did not express (gray arrowheads) Hnf4α. B, representative immunofluorescence against Hnf4α (red) and Ki67 (green) in polyps
Min +
GraphPad Prism 5, GraphPad Software) and data weresed as mean ± SEM.
Cancer Res; 70(22) November 15, 2010 9425
014. © 2010 American Association for Cancer
Resu
Hnf4αnovelHnf
T-cellity, anliferatactiveduringTo exHnf4αitoredcrypt-charaWnt/βrevealand dcells (were aand ccencedetectitheliagene tless elnormaInte
to pospolypstion ofoundgrowthsion cmice s
Figuremarginnormalisegmenpresent
Darsigny et al.
Cance9426
D
Published OnlineFirst November 9, 2010; DOI: 10.1158/0008-5472.CAN-10-1697
lts
does not act as a tumor-suppressor gene but is amodifier of intestinal adenoma formation4α physically interacts with Tcf-4, reduces β-catenin/factor/lymphoid enhancer factor transcriptional activ-d is involved in the control of intestinal epithelial pro-ion (13, 17). The Wnt/β-catenin pathway is mostlyin crypt stem cell/progenitors, and Hnf4α is inducedintestinal epithelial cell differentiation in culture (11).plore the apparent opposite relationship betweenexpression and sustained β-catenin activity, we mon-the expression level of Hnf4α along the intestinal
villus axis as well as in intestinal ApcMin polyps wellcterized by harboring sustained activation of the-catenin pathway (28). Immunofluorescence detectioned nuclear expression of Hnf4α in transit amplifyingifferentiated cells as well as in crypt stem/progenitorFig. 1A). In addition, proliferative Ki67-labeled cellslways positive for Hnf4α in both the small intestineolon (Fig. 1B, left). On the contrary, immunofluores-experiments showed that Hnf4α protein was poorlyed in the majority of the Ki67-positive ApcMin polyp ep-l cells of the jejunum and colon (Fig. 1B, right). Hnf4αranscript expression was confirmed to be significantlyevated in ApcMin polyps when compared with adjacentl intestinal biopsies (Fig. 2).stinal polyps originate from stem cells (21). This led ustulate that low level of Hnf4α expression within ApcMin
could either be functionally related to polyp forma-r only be reflective of the nature of the disorganizeder stem cells that contribute to polyp formation and. To genetically test whether reducing Hnf4α expres-
ould enhance intestinal tumorigenesis, we generatedP2 tradition97.6%smallHnf4αand Aand thalong
t compared with three margin biopsies (M) per segment]. Data areed as mean ± SEM. *, P < 0.05; **, P < 0.01.
FigureC57BL(M, n =age inpolyp size (C) in the whole intestine of individual mice from A. Data arepresen
r Res; 70(22) November 15, 2010
Research. on September 16, 2cancerres.aacrjournals.org ownloaded from
nscripts into a C57BL/6J ApcMin background. The con-al deletion of Hnf4α fourth and fifth exons led to areduction (P < 0.001) of P1 and P2 transcripts in theintestine (Supplementary Fig. S1; ref. 13). ApcMin;ΔIEC (mutants), ApcMin; Hnf4αΔIEC/+ (heterozygotes),pcMin; Hnf4α (control) littermate mice were sacrificed,
ted as mean ± SEM. *, P < 0.05; **, P < 0.01.
e number of polyps was assessed in each individual
pecifically lacking intestinal epithelial Hnf4α P1 and
2. qPCR of Hnf4α mRNA in enriched polyp tissue versus intestinalof C57BL/6J-APCMin animals. Relative expression of Hnf4αzed with the housekeeping gene Tbp [n = 6 polyps (P) per
3. Reduced polyp multiplicity in Hnf4αΔIEC mice on a/6J-APCMin background. A, polyp macrocount of mutants6), heterozygotes (H, n = 6), and controls (C, n = 7) at 4 months ofeach different intestinal segment. Total polyp macrocount (B) and
the rostrocaudal axis of the gut. A major effect on
Cancer Research
014. © 2010 American Association for Cancer
intestimiceThe mloss wpolyps(4.2 ±versusallelesicantgrounnotedbackgmainicantlyHnf4αled tonal eptinalaffecti
CondepitheexpreThe
us to eregula
clarifyated wlial cA stattranscnum o(Hnf4αTo gaclassifAnnotmaticsoxidorlar lippredicthe je74 genpotenSpecif2-foldfurtheysis coscriptadult(Fig. 4
Figuregenes ioxidoreA, exprmodulamicroarat least(see SuThese goxidoremonitoRNA extissue (Gapdhused toB, ChIP(n = 6)jejunalData wand areenrichmwith anrelativewith nopresent*, P < 0***, P <
Hnf4α Promotes Gut Neoplasia
www.a
D
Published OnlineFirst November 9, 2010; DOI: 10.1158/0008-5472.CAN-10-1697
nal tumor initiation was observed in ApcMin; Hnf4αΔIEC
compared with ApcMin; Hnf4α control mice (Fig. 3A).ultiplicity of intestinal polyps with Hnf4α epithelialas dramatically reduced in the jejunum (7.8 ± 2.9versus 26.1 ± 4.7 polyps in controls; P = 0.0084), ileum1.4 versus 14.9 ± 2.4; P = 0.0034), and colon (0.16 ± 0.161.6 ± 0.3; P = 0.0024). Overall, the loss of both Hnf4αin the intestinal epithelium led to a drastic and signif-68.1% reduction of the polyp load under ApcMin back-d (P = 0.0017), whereas no significant tendency waswhen one single Hnf4α allele was lost under the sameround (Fig. 3B). The average diameter size of the re-ng polyps of ApcMin; Hnf4αΔIEC mice was not signifi-different when compared with the polyps of ApcMin;control mice (Fig. 3C). Overall, these observationsthe unexpected conclusion that abrogation of intesti-ithelial Hnf4α expression limited the initiation of intes-neoplasia under the ApcMin background withoutng polyp growth.
itional loss of Hnf4α in the mouse small intestinallium is associated with an oxidative stress genession signatureHnf4α-promoting effect during gut tumorigenesis led
xplore a novel physiologic role for this transcriptionaltor in controlling jejunum epitconfir
0.001.
acrjournals.org
Research. on September 16, 2cancerres.aacrjournals.org ownloaded from
the nature of overall gene expression changes associ-ith the loss of Hnf4α gene integrity in intestinal epithe-ells, a gene expression profiling was performed.istical analysis (P ≤ 0.05) predicted 285 unique mouseripts being significantly modulated between the jeju-f control and conditionally deleted Hnf4α adult miceΔIEC; differential ratio ≥2.0; Supplementary Table S1).in insight into how these modifications could beied as biological meaning, we used the Database foration, Visualization, and Integrated Discovery Bioinfor-Resources tool (29, 30). Specific systems, such as theeductase component as well as the immune and cellu-id metabolic processes, were identified as the mostted significant biological pathways to be affected injunum of Hnf4αΔIEC mice. The proximal promoter ofes from the oxidoreductase system was screened fortial Hnf4α binding sites (Supplementary Table S3).ic gene targets predicted to be modulated by at leastwith at least one putative Hnf4α-binding site werer selected (Supplementary Table S3). A qRT-PCR anal-nfirmed that these selected 18 individual gene tran-s were significantly reduced in the small intestine ofHnf4αΔIEC mice when compared with control miceA). The promoter region for most of these genes was
med to be occupied by Hnf4α as revealed by ChIP-helial functions. To qPCR analysis (Fig. 4B). Consequently, the lipid peroxidation
4. Hnf4α regulatesnvolved in the control ofductase activity.ession of 18 genested by >2-fold on theray and predicted to haveone Hnf4α binding sitepplementary Table S3).enes involved inductive functions werered by qPCR on totaltracted from adult jejunumn = 3 for each group).housekeeping gene wasnormalize expression data.analysis of six samples
of cells obtained fromtissue of two adult mice.ere obtained by qPCRexpressed as the foldent of the DNA precipitatedantibody against Hnf4αto the DNA precipitatedrmal goat-IgG. Data areed as mean ± SEM..05; **, P < 0.01;
Cancer Res; 70(22) November 15, 2010 9427
014. © 2010 American Association for Cancer
end prsignififrom HP ≤ 0.0higherintesticencethe lothe exa modoxidat
HNF4coloreTo
role iROS, icoloreefficietion oleft). RDCF-Dtotal n(P < 0sion cshRNAfirmedscripts(Supp
integrled totion oty conHCT1and thof ROevalua5-FU,CRC,treatmcientoverexreduc5-FU t
HNF4expreThe
CRC sat thepared(Fig. 7signifiin a suparedTo
pressiin CRgets tHnf4αin the(2.60-f(1.54-ftranscCRC pnificaCYP2Bin aveductapancyidentinificanpromHNF4er regremaivationlevelsROS-nROS p
Discu
Hnftarget
Figurecaspaseach grcrypt-villus units. Data are presented as mean ± SEM. ***, P < 0.001.DAPI, 4
Darsigny et al.
Cance9428
D
Published OnlineFirst November 9, 2010; DOI: 10.1158/0008-5472.CAN-10-1697
oduct MDA, a typical marker for oxidative stress, wascantly elevated in intestinal epithelial cells harvestednf4αΔIEC mice compared with control mice (1.50-fold;5, n = 9). This observation coincided with a significantindex of intestinal epithelial cell apoptosis in the smallne of Hnf4αΔIEC mice as monitored by immunofluores-detection of cleaved caspase-3 (Fig. 5A and B). Thus,ss of intestinal epithelial Hnf4α led to a reduction inpression of oxidoreduction-related genes, along withest but significant increase of intestinal epithelialive stress and apoptosis.
α protects against the accumulation of ROS inctal cancer cellsfurther establish whether HNF4α could have a directn protecting against intracellular accumulation ofts expression was modulated within two independentctal cancer (CRC) cell lines. HNF4α expression wasntly neutralized in HT-29 cells with the stable integra-f a specific HNF4α-targeted shRNA lentivirus (Fig. 6A,OS-positive cells were visualized with the use of a CM-A fluorescent labeling probe (Fig. 6A, middle) and theumber of ROS-labeled cells was increased by 2.37-fold.001) in HT-29 cells made deficient for HNF4α expres-ompared with genome-integrated control nontargetlentivirus (Fig. 6A, right). A qRT-PCR analysis con-the reduction of oxidoreductase-related gene tran-
′,6-diamidino-2-phenylindole.
in HT-29 cells harboring reduced HNF4α expressionlementary Fig. S2). On the contrary, stable retroviral
variouover, H
r Res; 70(22) November 15, 2010
Research. on September 16, 2cancerres.aacrjournals.org ownloaded from
ation of Hnf4α in the HCT116 cell line (Fig. 6B, left)a significant and spontaneous 51.1% (P < 0.001) reduc-f the number of ROS-positive cells compared with emp-trol HCT116 cells (Fig. 6B, middle and right). Because16 cells do not harbor mutation within the TP53 geneus retain the capacity to produce a significant amountS following chemotherapeutic treatments (31), we nextted whether ROS deleterious production in response toan agent used in the clinic as an adjuvant therapy forcould be altered in the presence of Hnf4α. Althoughent of control HCT116 cells with 5-FU led to an effi-production of intracellular ROS, HCT116 cell lines thatpressed Hnf4α displayed a significant 55.3% (P < 0.001)tion in the number of ROS-producing cells followingreatment (Fig. 6C).
α and oxidoreductase-related genes are overlyssed in CRCexpression of HNF4α was next assessed in humanamples. HNF4A was increased by 2.19-fold (P = 0.002)gene transcript levels in tumor samples when com-with paired margin resections in 31 patients of 40A). A similar analysis at the protein level indicated acant 2.81-fold induction (P < 0.0001) of HNF4α proteinbset of human cancer samples when individually com-with their paired margin resection (Fig. 7B–D).further validate the relationship between HNF4A ex-on and oxidoreductase-related gene expression statusC, we next monitored expression of specific gene tar-hat were validated to be decreased in the intestine ofΔIEC mice. This analysis revealed significant increasesexpression of CYP2B6 (4.38-fold; P = 0.048), CYP2D6old; P = 0.0182), GSTA4 (1.66-fold; P = 0.024), GSTK1old; P = 0.002), and NQO1 (3.80-fold; P = 0.0003) generipts when comparing the ratio of tumor/margin fromatient samples (Fig. 8A). These modulations were sig-ntly correlated with the induced level of HNF4A for6 and GSTK1 (Fig. 8A). Specific primer sets spanningrage 300 bp within the proximal promoter of oxidore-se-related genes were used to test for HNF4α occu-by ChIP-qPCR on cultured CRC cells. This analysis
fied the CYP2B6 and GSTK1 gene promoters to be sig-tly targeted by HNF4α in the proximal region of theoter transcriptional start site (Fig. 8B). However,α could be interacting outside of the proximal promot-ion for each human oxidoreductase-related genes thatned negative by this assay. Taken together, these obser-s support a strong relationship between increasedof HNF4α , high expression of HNF4α-targetedeutralizing genes, and increased protection againstroduction in CRC.
ssion
4α plays major roles in epithelial cell polarization bying specific genes encoding cell junction molecules in
5. Hnf4αΔIEC mice display a higher apoptotic index. A, cleavede-3 immunofluorescence in Hnf4αΔIEC and control mice (n = 3, foroup). B, quantification of cleaved caspase-3–positive cells per
s tissues, including the intestine (10, 11, 15, 17, 32). More-nf4α acts as a morphogen with its capacity to trigger
Cancer Research
014. © 2010 American Association for Cancer
microdenceveloptranscbryonthat aadult tthe livsuppoThe react wit(33) sugene. Otional
ROStial roferentof ROtuentslular lfrom Rtases,chronfyingcanceROS-d
FigureagainstA, immin HT-2pLKO.1nontarg(left). ImROS-poand shNmonitothe CM(middleand qucells peB, immHCT11with pB(left). ImROS-poor Hnf4incubatROS prmagnifiROS-po(right, ncells inpopulatposttre5-FU aCM-DC20× oriquantifiper higvehiclewith 12Data armean ±and areindepen
Hnf4α Promotes Gut Neoplasia
www.a
D
Published OnlineFirst November 9, 2010; DOI: 10.1158/0008-5472.CAN-10-1697
villus formation in nonepithelial cell lines (12, 13). Evi-for a role of Hnf4α in governing the pattern of tissue de-ment came from early conditional deletion of thisriptional regulator duringmouse liver (10) and colon em-ic development (14). Hnf4α gene deletion approachesimed to investigate the specific role of this regulator onissuemaintenance have revealed lesser consequences oner maintenance (9) and intestinal tissue integrity (13, 15),rting the nature of Hnf4α as an embryonic morphogen.cent findings that theWnt/β-catenin pathway can inter-hHnf4α in intestinal epithelial cells (17) and hepatocytesggested that Hnf4α could act as a tumor-suppressor
ur findings suggest that neutralizing HNF4α transcrip-activity during intestinal cancer ispressu
dent experiments.
acrjournals.org
Research. on September 16, 2cancerres.aacrjournals.org ownloaded from
are chemically reactive molecules that display essen-les in biological functions such as cell growth, dif-iation, and inflammation (34). Excessive productionS can lead to oxidative damage to intracellular consti-, including lipids, protein, and DNA (35). ROS intracel-evels are controlled with their balanced eliminationOS-scavenging systems such as the superoxide dismu-glutathione peroxidase, and catalase (34). Althoughic increases in ROS may contribute to cancer by ampli-genomic instability, it is well described that manyr cells develop paradoxical enhancement of theiretoxifying capacities to become adapted to exogenous
re and oxidative stress and then more resistant tobeneficial. chemotherapy-induced cell death (36). On the other hand,
6. HNF4α protectsthe production of ROS.unoblot of HNF4α9 cells infected with-puro shHNF4α or shet (NT) shRNA controlmunofluorescence ofsitive cells in shHNF4α-T-infected cells as
red by incubation with-DCF-DA ROS probe, 20× original magnification)antification of ROS-positiver high field (right, n = 3).unoblot of HNF4α in6 cells after infectionabe Hnf4α1 retrovirusesmunofluorescence ofsitive cells in empty vectorα1 populations of cells afterion with the CM-DCF-DAobe (middle, 20× originalcation) and quantification ofsitive cells per high field= 3). C, ROS-positiveempty vector or Hnf4α1ion of cells at 18 hoursatment with 12.5 μmol/Lfter the incubation ofF-DA ROS probe (middle,ginal magnification) andcation of ROS-positive cellsh field less 2% DMSOat 18 hours posttreatment.5 μmol/L 5-FU (right, n = 3).e presented asSEM. ***, P < 0.001representative of three
Cancer Res; 70(22) November 15, 2010 9429
014. © 2010 American Association for Cancer
profoustem cmentfurtheminatadvannificanto norThe
relatedupregugastritumorof HNcell prdepenthis rtheseour finHow
in therelatioactivitin theare thgrowthsion.Hnf4αprolifevation
Hnf4αtranscepithewas a(15) acryptmanifmechainitiatficultof LOGeneas thebe chproduexpresproduthe exwith tlowerpleteexertsROS tcell sufor th(44). Afoundbetwe
Figuregene wmatchematchesample
Darsigny et al.
Cance9430
D
Published OnlineFirst November 9, 2010; DOI: 10.1158/0008-5472.CAN-10-1697
nd suppression of ROS can be deleterious for normalell maintenance, resulting in the paradoxical enhance-of genomic alterations (37). Strategies designed tor induce oxidative stress by decreasing but not eli-ing oxidant-detoxifying activities present conceptualtages in targeting specific malignant cells with a sig-t misbalance of their antioxidant system as opposedmal cells.finding that HNF4α expression can be positively cor-with cancer is not unique. HNFα was reported to belated in human hepatocellular carcinoma (38), humanc intestinal metaplasia (39), and ovarian mucinuss (40). Recent observations implied that a reductionF4α activity by ligand antagonists could attenuateoliferation and promote apoptotic cell death in two in-dent CRC cell lines (41). Given the functional role ofegulator in cellular polarization and differentiation,results were unexpected but are in agreement withdings.does Hnf4α contribute to the tumor multiplicityApcMin context? Our observations have supported anship between increased levels of Wnt/β-cateniny and reduced levels of Hnf4α in pathologic crypt cellsmouse intestine. Proliferating progenitor/stem cellse origin entities required for polyp initiation and(21) and display a detectable level of Hnf4α expres-
Thus, it remains plausible that further reduction ofin epithelial progenitor cells could not influence the
rative events that lead to polyp growth. The obser-that polyp size was not influenced by the loss oflethalpressi
d samples in Western blot (n = 41) expressed as fold change in expression. D, is. Data are presented as mean ± SEM. **, P < 0.01; ***, P < 0.001.
r Res; 70(22) November 15, 2010
Research. on September 16, 2cancerres.aacrjournals.org ownloaded from
in ApcMin mice supports this idea. Loss of Hnf4αriptional activity resulted in spontaneous intestinallial apoptosis in the small intestine, a feature thatlso recently observed in the mouse colonic epitheliumnd cultured CRC cell lines (41). A decrease in epithelialcell survival following loss of heterozygosity (LOH)estation could represent an early cell autonomousnism leading to premature clearance of nascent polypion in absence of Hnf4α. This possibility remains dif-to show in vivo given the unpredictable occurrenceH in a given pool of ApcMin intestinal epithelial cells.clustering analysis identified oxidoreductase activitiesmost significant biological functions predicted to
allenged within Hnf4αΔIEC mice. We found that ROSction in CRC cell lines was dependent on HNF4αsion. ApcMin polyp formation is associated with higherction of oxidative stress (42) as well as changes inpression of genes involved in ROS (43) correlatinghe observation that polyp epithelial cells are expressingHnf4α levels. However, we rationalize that a com-loss of Hnf4α in a biological situation that constantlyoxidative stress pressure would promote the reach of ahreshold that could become deleterious for epithelialrvival. The recent identification of a candidate genee modifier of Min 2 locus supports this rationaletp5a1, a gene required for oxidative metabolism, wasto be mutated in this locus, providing a novel linken dysfunction of mitochondrial respiratory chain,
overproduction of intracellular ROS, and polyp sup-on in ApcMin mice (44). On the other hand, increased7. HNF4α is upregulated in CRC. A, qPCR of HNF4A gene transcripts in tumor versus margin tissues of 40 patients with CRC. B2M housekeepingas used to normalize expression data. B, five representative immunoblots against HNF4α within 41 patients with CRC. Densitometry ofd samples expressed as fold change in expression is indicated below for Western blot (WB) and paired qPCR expression. C, densitometry of
mmunofluorescence against HNF4α in the corresponding tissue
Cancer Research
014. © 2010 American Association for Cancer
level osubseqtant aovercoIn c
facilitabe a trsion. Talso cFinallexpresductasthe detions
the prdesigned to reduce antioxidant activities and promote theefficie
Disclosure of Potential Conflicts of Interest
No p
Ackn
We
FigureoxidoreA, qPCas direcmice (ncorrelatmRNAaccordtest areHNF4αwere obexpressof the Dantibodthe DNgoat-Igand nopresent*, P < 0***, P < 0.001.
Hnf4α Promotes Gut Neoplasia
www.a
D
Published OnlineFirst November 9, 2010; DOI: 10.1158/0008-5472.CAN-10-1697
f HNFα expression in human CRC would represent auent step for tumor epithelial cells to become resis-gainst sustained ROS intracellular production andme cell death.onclusion, our results indicate that Hnf4α is crucial tote tumorigenesis in ApcMin mice. Hnf4α was found toanscriptional regulator of oxidoreductase gene expres-he modulation of HNF4α expression in CRC cell linesonfirmed its crucial role in regulating ROS production.y, a significant parallel was found between elevatedsion of HNF4α and high expression of some oxidore-e genes in CRC biopsies. With the growing interest in
velopment of antagonists for blocking HNF4α func-as a therapeutic approach, our observations highlightQuébecand Virhuman
acrjournals.org
Research. on September 16, 2cancerres.aacrjournals.org ownloaded from
omising exploration of such strategies in a program
ncy of CRC chemotherapies.
otential conflicts of interest were disclosed.
owledgments
thank the Microarray Platform Lab at McGill University, Génome
8. Genes involved inductase activity in CRC.R of the 11 genes identifiedt targets of Hnf4α in= 20–27 patients). Theion factor with HNF4Aexpression and P valueing to Spearman correlationindicated below. B, ChIP ofin T84 cells (n = 4). Datatained by qPCR and areed as the fold enrichmentNA precipitated with any against HNF4α relative toA precipitated with normalG used as the backgroundrmalized to input. Data areed as mean ± SEM..05; **, P < 0.01;
Innovation Centre, for generation of the cDNA arrays; Evelyne Royginie Dozias for technical assistance; and Gérald Bernatchez forcolorectal tumor processing.
Cancer Res; 70(22) November 15, 2010 9431
014. © 2010 American Association for Cancer
Grant
CanaCanadiaand J. CQuébecof the FClinique
Chair inof a J.Arecipien
Theof pageaccorda
Refe1. Du
factispriU S
2. Yubo
3. Gudia
4. Lotes
5. Baofthe
6. ChgenonDe
7. Gureg200
8. GuresGe
9. HacytnanBio
10. Pacongen
11. Lustoclial
12. Chnumic
13. BafacplaPh
14. GaHeof
15. Dafaction4:e
16. Ahintdis
17. Ca4αfun629
18. Cle200
Darsigny et al.
Cance9432
D
Published OnlineFirst November 9, 2010; DOI: 10.1158/0008-5472.CAN-10-1697
Support
dian Institute of Health Research (MOP-89770; F. Boudreau) and an Institute of Health Research team grant (CTP-82942). F. Boudreauarrier are scholars from the “Fonds de la Recherche en Santé du
”. F. Boudreau, N. Perreault, F.P. Gendron, and J. Carrier are membersonds de la Recherche en Santé du Québec–funded “Centre de RechercheÉtienne Lebel”. E.G. Seidman is a recipient of a Canadian ResearchReceOnlineF
4–308.vers H. Wnt/β-catenin signaling in development and disease. Cell6;127:469–80.
19. Pinint
20. Sade69
21. Baof-
22. BoacFA
23. MaGuin(du33
24. Bonuprgetio31
25. BrMoenBio
26. Lenehig27
27. Gareqan
28. KoClneMs
29. DeAn20
30. HuanNa
31. Hwdece
32. Spsioen
33. ConaGa
34. TraRORe
35. PeHodis
r Res; 70(22) November 15, 2010
Research. on September 16, 2cancerres.aacrjournals.org ownloaded from
Immune-Mediated Gastrointestinal Disorders. E. Levy is the recipient. deSève Endowed Chair in Nutrition. M. Darsigny and J.P. Babeu arets of Natural Sciences and Engineering Research Council fellowships.costs of publication of this article were defrayed in part by the paymentcharges. This article must therefore be hereby marked advertisement innce with 18 U.S.C. Section 1734 solely to indicate this fact.
ived 05/11/2010; revised 08/27/2010; accepted 09/10/2010; publishedirst 11/09/2010.
rencesncan SA, Manova K, Chen WS, et al. Expression of transcriptiontor HNF-4 in the extraembryonic endoderm, gut, and nephrogenicsue of the developing mouse embryo: HNF-4 is a marker formary endoderm in the implanting blastocyst. Proc Natl Acad SciA 1994;91:7598–602.
an X, Ta TC, Lin M, et al. Identification of an endogenous ligandund to a native orphan nuclear receptor. PloS One 2009;4:e5609.pta RK, Kaestner KH. HNF-4α: from MODY to late-onset type 2betes. Trends Mol Med 2004;10:521–4.ve-Gregory L, Permutt MA. HNF4A genetic variants: role in diabe-. Curr Opin Clin Nutr Metab Care 2007;10:397–402.rrett JC, Lee JC, Lees CW, et al. Genome-wide association studyulcerative colitis identifies three new susceptibility loci, includingHNF4A region. Nat Genet 2009;41:1330–4.en WS, Manova K, Weinstein DC, et al. Disruption of the HNF-4e, expressed in visceral endoderm, leads to cell death in embry-
ic ectoderm and impaired gastrulation of mouse embryos. Genesv 1994;8:2466–77.pta RK, Vatamaniuk MZ, Lee CS, et al. The MODY1 gene HNF-4αulates selected genes involved in insulin secretion. J Clin Invest5;115:1006–15.pta RK, Gao N, Gorski RK, et al. Expansion of adult β-cell mass inponse to increased metabolic demand is dependent on HNF-4α.nes Dev 2007;21:756–69.yhurst GP, Lee YH, Lambert G, Ward JM, Gonzalez FJ. Hepato-e nuclear factor 4α (nuclear receptor 2A1) is essential for mainte-ce of hepatic gene expression and lipid homeostasis. Mol Celll 2001;21:1393–403.rviz F, Matullo C, Garrison WD, et al. Hepatocyte nuclear factor 4αtrols the development of a hepatic epithelium and liver morpho-esis. Nat Genet 2003;34:292–6.sier CR, Babeu JP, Auclair BA, Perreault N, Boudreau F. Hepa-yte nuclear factor-4α promotes differentiation of intestinal epithe-cells in a coculture system. Am J Physiol 2008;294:G418–28.iba H, Sakai N, Murata M, et al. The nuclear receptor hepatocyteclear factor 4α acts as a morphogen to induce the formation ofrovilli. J Cell Biol 2006;175:971–80.beu JP, Darsigny M, Lussier CR, Boudreau F. Hepatocyte nucleartor 4α contributes to an intestinal epithelial phenotype in vitro andys a partial role in mouse intestinal epithelium differentiation. Am Jysiol 2009;297:G124–34.rrisonWD,BattleMA,YangC, Kaestner KH,Sladek FM,DuncanSA.patocyte nuclear factor 4α is essential for embryonic developmentthe mouse colon. Gastroenterology 2006;130:1207–20.rsigny M, Babeu JP, Dupuis AA, et al. Loss of hepatocyte-nuclear-tor-4α affects colonic ion transport and causes chronic inflamma-resembling inflammatory bowel disease in mice. PloS One 2009;
7609.n SH, Shah YM, Inoue J, et al. Hepatocyte nuclear factor 4α in theestinal epithelial cells protects against inflammatory bowelease. Inflamm Bowel Dis 2008;14:908–20.ttin AL, Le Beyec J, Barreau F, et al. Hepatocyte nuclear factor, a key factor for homeostasis, cell architecture, and barrierction of the adult intestinal epithelium. Mol Cell Biol 2009;29:
to D, Clevers H. Wnt control of stem cells and differentiation in theestinal epithelium. Exp Cell Res 2005;306:357–63.ncho E, Batlle E, Clevers H. Signaling pathways in intestinalvelopment and cancer. Annu Rev Cell Dev Biol 2004;20:5–723.rker N, Ridgway RA, van Es JH, et al. Crypt stem cells as the cells-origin of intestinal cancer. Nature 2009;457:608–11.udreau F, Lussier CR, Mongrain S, et al. Loss of cathepsin Ltivity promotes claudin-1 overexpression and intestinal neoplasia.SEB J 2007;21:3853–65.dison BB, Dunbar L, Qiao XT, Braunstein K, Braunstein E,mucio DL. Cis elements of the villin gene control expressionrestricted domains of the vertical (crypt) and horizontalodenum, cecum) axes of the intestine. J Biol Chem 2002;277:275–83.udreau F, Rings EH, van Wering HM, et al. Hepatocyteclear factor-1 α, GATA-4, and caudal related homeodomainotein Cdx2 interact functionally to modulate intestinalne transcription. Implication for the developmental regulan of the sucrase-isomaltase gene. J Biol Chem 2002;277:909–17.unet S, Thibault L, Lepage G, Seidman EG, Dube N, Levy E.dulation of endoplasmic reticulum-bound cholesterol regulatoryzymes by iron/ascorbate-mediated lipid peroxidation. Free Radicl Med 2000;28:46–54.page G, Munoz G, Champagne J, Roy CC. Preparative stepscessary for the accurate measurement of malondialdehyde byh-performance liquid chromatography. Anal Biochem 1991;197:7–83.rneau H, Paquin MC, Carrier JC, Rivard N. E2F4 expression isuired for cell cycle progression of normal intestinal crypt cellsd colorectal cancer cells. J Cell Physiol 2009;221:350–8.ngkanuntn R, Bubb VJ, Sansom OJ, Wyllie AH, Harrison DJ,arke AR. Dysregulated expression of β-catenin marks earlyoplastic change in Apc mutant mice, but not all lesions arising inh2 deficient mice. Oncogene 1999;18:7219–25.nnis G, Jr., Sherman BT, Hosack DA, et al. DAVID: Database fornotation, Visualization, and Integrated Discovery. Genome Biol03;4:P3.ang da W, Sherman BT, Lempicki RA. Systematic and integrativealysis of large gene lists using DAVID bioinformatics resources.t Protoc 2009;4:44–57.ang PM, Bunz F, Yu J, et al. Ferredoxin reductase affects p53-pendent, 5-fluorouracil-induced apoptosis in colorectal cancerlls. Nat Med 2001;7:1111–7.ath GF, Weiss MC. Hepatocyte nuclear factor 4 provokes expres-n of epithelial marker genes, acting as a morphogen in dediffer-tiated hepatoma cells. J Cell Biol 1998;140:935–46.lletti M, Cicchini C, Conigliaro A, et al. Convergence of Wnt sig-ling on the HNF4α-driven transcription in controlling liver zonation.stroenterology 2009;137:660–72.chootham D, Alexandre J, Huang P. Targeting cancer cells byS-mediated mechanisms: a radical therapeutic approach? Natv 2009;8:579–91.
rry G, Raina AK, Nunomura A, Wataya T, Sayre LM, Smith MA.w important is oxidative damage? Lessons from Alzheimer'sease. Free Radic Biol Med 2000;28:831–4.Cancer Research
014. © 2010 American Association for Cancer
36. Scsw
37. Li Tqui
38. Xurolcar
39. KonuccorPa
40. Sufac
41. Screc20
42. Mainpa
43. Leco20
44. Ba
Hnf4α Promotes Gut Neoplasia
www.a
D
Published OnlineFirst November 9, 2010; DOI: 10.1158/0008-5472.CAN-10-1697
humacker PT. Reactive oxygen species in cancer cells: live by theord, die by the sword. Cancer Cell 2006;10:175–6.S, Marban E. Physiological levels of reactive oxygen species are re-red tomaintaingenomic stability in stemcells. StemCells 28:1178–85.L, Hui L, Wang S, et al. Expression profiling suggested a regulatorye of liver-enriched transcription factors in human hepatocellularcinoma. Cancer Res 2001;61:3176–81.jima K, Kishimoto T, Nagai Y, et al. The expression of hepatocytelear factor-4α, a developmental regulator of visceral endoderm,relates with the intestinal phenotype of gastric adenocarcinomas.
thology 2006;38:548–54.gaiM,UmezuH,YamamotoT, et al. Expressionof hepatocytenucleartor 4α in primary ovarianmucinous tumors. Pathol Int 2008;58:681–6.(Msu20
acrjournals.org
Research. on September 16, 2cancerres.aacrjournals.org ownloaded from
hwartz B, Algamas-Dimantov A, Hertz R, et al. Inhibition of colo-tal cancer by targeting hepatocyte nuclear factor-4α. Int J Cancer09;124:1081–9.bley JG, Pacher P, Bai P, et al. Suppression of intestinal polyposisApcmin/+ mice by targeting the nitric oxide or poly(ADP-ribose)thways. Mutat Res 2004;548:107–16.clerc D, Deng L, Trasler J, Rozen R. ApcMin/+ mouse model oflon cancer: gene expression profiling in tumors. J Cell Biochem04;93:1242–54.ran AA, Silverman KA, Zeskand J, et al. The modifier of Min 2om2) locus: embryonic lethality of a mutation in the Atp5a1 gene
ggests a novel mechanism of polyp suppression. Genome Res07;17:566–76.Cancer Res; 70(22) November 15, 2010 9433
014. © 2010 American Association for Cancer
Related Documents