By Chris Paine h-ps://bioknowledgy.weebly.com/ 2.1 Molecules to metabolism EssenAal idea: Living organisms control their composiAon by a complex web of chemical reacAons. The background is just a small part of the IUBMB-Sigma-Nicholson Metabolic Pathways Chart aims to show all the metabolic pathways found in eukaryote cells. The chart in it's enArety shows how complex the chemicals reacAons needed to support life in a single cell unit.

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

ByChrisPaine
h-ps://bioknowledgy.weebly.com/
2.1Moleculestometabolism
EssenAalidea:LivingorganismscontroltheircomposiAonbyacomplexwebofchemicalreacAons.
ThebackgroundisjustasmallpartoftheIUBMB-Sigma-NicholsonMetabolicPathwaysChartaimstoshowallthemetabolicpathwaysfoundineukaryotecells.Thechartinit'senAretyshowshowcomplexthechemicalsreacAonsneededtosupportlifeinasinglecellunit.

2.1.U1 Molecular biology explains living processes in terms of the chemical substances involved.
ThestructureofDNAwasdiscoveredin1953,sincethenmolecularBiologyhastransformedourunderstandingoflivingprocesses
TherelaAonshipbetweengenesandthepolypepAdestheygenerateisattheheartofthisscience.Thecentralideacanbesimplifiedto“DNAmakesRNAmakesprotein”.TheinformaAoninthisflowcannotbereversedandtheproteingeneratedcannotchangetheRNAorDNA.
h-ps://en.wikipedia.org/wiki/File:DNA_chemical_structure.svg

2.1.U1 Molecular biology explains living processes in terms of the chemical substances involved.
TheapproachofamolecularBiologistisareducAonistone–theyidenAfythestepsinametabolicpathwayandbreakdowneachoneintoit’scomponentparts.ThisapproachhasbeenaveryproducAveone.OurunderstandingofrespiraAon(2.8)andphotosynthesis(2.9)aregoodexamplesofthesuccessofthisapproach.
Organicmolecules,especiallyproteins,areverycomplexandvaried.Henceorganiccompoundshaveahugelyvariedroleswithin(andoutsideof)cells.ThereisalotaboutorganicmoleculesincellswesAllhavenotdiscoveredorunderstood.
SomescienAststhinkthatthereducAonistapproachaloneisulAmatelylimited.Moleculescanhavedualroles(e.g.Melaninisthepigmentthatcoloursbothskinandeyes)andalsomayinteractwitheachotherinwaysthatareducAonistapproachoverlooks.
h-ps://en.wikipedia.org/wiki/File:DNA_chemical_structure.svg

2.1.U2 Carbon atoms can form four covalent bonds allowing a diversity of stable compounds to exist.
Despiteonlybeingthe15thmostabundantelementontheplanetcarbonformsthebackboneofeverysingleorganicmolecule.
Covalentbondsarethestrongesttypeofbondbetweenatoms.Stablemoleculescanbeformed.
Becauseofthestabilityofcovalentbondslargemoleculeswithmanybondscanbeformed.TiAnisthelargestknownproteinanditcontains539,000atoms(chemicalformulaC169723H270464N45688O52243S912.).TheimagebelowshowasmallpartoftheTiAnmolecule.
h-p://www.ks.uiuc.edu/images/ofmonth/2002-11/AAn.jpg
CarbonatomscontainfourelectronsintheiroutershellallowingthemtoformfourcovalentbondswithpotenAalfourotherdifferentatoms,e.g.methane(CH4).
TheresultoftheseproperAesisanalmostinfinitenumberofdifferentpossiblemoleculesinvolvingcarbon.
h-p://commons.wikimedia.org/wiki/File:Carbon-atom.jpg
h-p://upload.wikimedia.org/wikipedia/commons/3/3e/Methane-2D-dot-cross.png

ClassifyingCarbonCompounds(notinSkeletonnotes)-refpg65textbook
NameofGroup FullStructure SimplifiedNota:on
hydroxyl
amine
carboxyl
methyl

2.1.U3 Life is based on carbon compounds including carbohydrates, lipids, proteins and nucleic acids.
Carbohydrates• Containcarbon,hydrogenandoxygen• OrganiccompoundsconsisAngofoneormoresimplesugars• Monomersfollowthegeneralbasicformulaof(CH2O)x• Monomersarecommonlyringshapedmolecules
Note:ExcepAonstothisthebasicformulaandtheinclusionofotherelements(e.g.N)canoccur
Glucose–ahexose (6carbon)monomer
h-p://www.doctortee.com/dsu/Ahickjian/cse-img/biology/chemistry/polysaccharides.jpg
pentose(5carbon)monomers

2.1.U3 Life is based on carbon compounds including carbohydrates, lipids, proteins and nucleic acids.
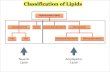
Lipids • Lipidsareagroupoforganicmoleculesthatareinsolubleinwaterbutsolubleinnon-
polarorganicsolvents• Commonlipidsincludetriglycerides(fats–solidatroomtemperatureandoils–liquid
atroomtemperature),phospholipidsandsteroidsh-p://hyperphysics.phy-astr.gsu.edu/hbase/organic/imgorg/lipid.gif

2.1.U3 Life is based on carbon compounds including carbohydrates, lipids, proteins and nucleic acids.
Leucine–anaminoacid
Proteins • Containcarbon,hydrogen,oxygenandnitrogen(addiAonallysulphuriscommoncomponent,butitisnotpresent
inallproteins) • Proteinsarelargeorganiccompoundsmadeofaminoacidsarrangedintooneormorelinearchains

2.1.U3 Life is based on carbon compounds including carbohydrates, lipids, proteins and nucleic acids.
Nucleicacids • Containcarbon,hydrogen,
oxygen,nitrogenandphosphorus
• Chainsofsub-unitscallednucleoAdes
• NucleoAdesconsistofbase,sugarandphosphategroupscovalentlybondedtogether
• IfthesugarisribosethenthenucleicacidformedisRNAifthesugarisdeoxyribosethenDNAisformed
h-p://www.nature.com/scitable/topicpage/Chemical-Structure-of-RNA-348
phosphate
base
sugar

ArginineAlanine Leucine
h-p://commons.wikimedia.org/wiki/File:Alanine.pngh-p://commons.wikimedia.org/wiki/File:Arginine.pngh-p://commons.wikimedia.org/wiki/File:Leucine.png
Herearethreeofthetwenty-oneaminoacidsfoundineukaryotes.IdenAfywhatpartsoftheirstructuresareidenAcal.
2.1.S2 Identification of biochemicals such as sugars, lipids or amino acids from molecular diagrams.

ArginineAlanine Leucine
Yeah,thatbit…
2.1.S2 Identification of biochemicals such as sugars, lipids or amino acids from molecular diagrams.

h-p://commons.wikimedia.org/wiki/File:AminoAcidball.svg
DrawnslightlydifferentlyyoucanseethebitthatisalwaysthesameandtheRGroup. TheRgroupislikexinanequaAon.Itisavariablethatstandsinforabunchofdifferentsidechains
2.1.S2 Identification of biochemicals such as sugars, lipids or amino acids from molecular diagrams.

Theaminegroup(NH2)
Thecarboxylgroup(COOH) n.b.thisisanacidicgroup
Lookoutforthisstructure
2.1.S2 Identification of biochemicals such as sugars, lipids or amino acids from molecular diagrams.

Hmmm…anaminegroupandanacidgroup… Whatshallwe callthisclass
ofmolecule?
h-p://commons.wikimedia.org/wiki/File:Filos_tercer_logo.JPG

Theamineandacidgroupscouldbeatoppositeends,theRcouldbeontop,bo-omorsidedependingonorientaAon.
2.1.S2 Identification of biochemicals such as sugars, lipids or amino acids from molecular diagrams.

Oritcouldberepresenteddifferently:
2.1.S2 Identification of biochemicals such as sugars, lipids or amino acids from molecular diagrams.

h-p://commons.wikimedia.org/wiki/File:Amino_Acids.svg
Don’tfreakout,youdon’tneedtoknowthemall,justthegeneralformula

H3C (CH2)n CO
OH
Generalstructuralformulaforafa-y*acid
*Iprefer“bigboned”
Carboxylicgroup
Chain(orring)ofcarbonandhydrogenatoms
h-p://www.eufic.org/arAcle/pt/nutricao/gorduras/expid/23/
2.1.S2 Identification of biochemicals such as sugars, lipids or amino acids from molecular diagrams.

2.1.S2 Identification of biochemicals such as sugars, lipids or amino acids from molecular diagrams.

2.1.S2 Identification of biochemicals such as sugars, lipids or amino acids from molecular diagrams.

2.1.S2 Identification of biochemicals such as sugars, lipids or amino acids from molecular diagrams.

2.1.S2 Identification of biochemicals such as sugars, lipids or amino acids from molecular diagrams.

2.1.S1 Drawing molecular diagrams of glucose, ribose, a saturated fatty acid and a generalized amino acid.
Trydrawingbyhand(oroneMolecules)thefollowingmolecules:• Glucose• Ribose• Ageneralisedsaturatedfa-yacid• Ageneralisedaminoacid• Anexampleaminoacide.g.Alanine(the
simplest)n.b.youalsoneedtotestyourself15mins,1dayandoneweeklatertomakesureyouremember
eMoleculesonlinedrawingtool
http://www.emolecules.com/

2.1.U4 Metabolism is the web of all the enzyme-catalysed reactions in a cell or organism.
RevisittheessenAalideaofthistopic.ExploretheIUBMB-Sigma-NicholsonMetabolicPathwaysChartandrealisethatmostcellsusethemajorityofthepathwaysandthateverypathiscontrolledbyadifferentenzyme.ThemetabolismasaconceptisthesumofallthepathwaysusedinaparAcularcell.

2.1.U5 Anabolism is the synthesis of complex molecules from simpler molecules including the formation of macromolecules from monomers by condensation reactions. 2.1.U5 Catabolism is the breakdown of complex molecules into simpler molecules including the hydrolysis of macromolecules into monomers.

2.1.U5 Anabolism is the synthesis of complex molecules from simpler molecules including the formation of macromolecules from monomers by condensation reactions. 2.1.U5 Catabolism is the breakdown of complex molecules into simpler molecules including the hydrolysis of macromolecules into monomers.

2.1.U5 Anabolism is the synthesis of complex molecules from simpler molecules including the formation of macromolecules from monomers by condensation reactions.
Maltosesynthasecondensestwomoleculesofglucoseintomaltoseformingaglycosidicbond
AribosomecondensestwoaminoacidsintoadipepAdeformingapepAdebond
Examplesofanabolismbycondensa:on
h-p://commons.wikimedia.org/wiki/File:PepAdformaAonball.svg
Thebondsformedaretypesofcovalentbonds.
Bondingmonomerstogethercreatesapolymer(mono=one,poly=many)

2.1.U5 Catabolism is the breakdown of complex molecules into simpler molecules including the hydrolysis of macromolecules into monomers.
LactasehydrolysesLactoseintoGlucoseandGalactosebreakingtheglycosidicbond
h-p://commons.wikimedia.org/wiki/File:Amino_acid4.png
h-p://commons.wikimedia.org/wiki/File:Lactose_hydrolysis.svg
AproteasehydrolysesadipepAdeintotwoaminoacidsbreakingthepepAdebond
Examplesofcatabolismbyhydrolysis

Youmightseetheterm“dehydra:onreac:on”menAonedinterchangeablywithcondensaAonreacAon.
TechnicallyadehydraAonreacAoniswhenthewater
moleculehascomefromoneofthereactants.
WhereasforacondensaAonreacAon,partofeachwatermoleculehascomefromeachreactant.
Inthecaseofthepreviousslide,OHfromoneglucose
andHfromtheother.
h-p://www.flickr.com/photos/zachd1_618/5738829330/

2.1.A1 Urea as an example of a compound that is produced by living organisms but can also be artificially synthesized.
Source:h-p://www.biog1445.org/demo/08/nitrogenouswastes.html
Vitalismnowadayshasnocreditasatheory,butabovestatementisseenbymanyfromahistoricalperspecAvetobeuntrue.ForanoutlineonvitalismreadthisarAclebyWilliamBetchel.TheapplicaAonstatementaboveimpliesthatthecentraltenetVitalismis‘onlyorganismscansynthesiseorganiccompounds’.Thisisnotaccurate,inessensevitalismproposesthatanunknowablefactorisessenAalinexplaininglife.VitalismonthispremiseisbothunscienAficandandunfalsifiable.
NatureofScience:Falsifica:onoftheories—thear:ficialsynthesisofureahelpedtofalsifyvitalism.(1.9)Wöhleraccidentallysynthesisedureain1828,whilsta-empAngtoprepareammoniumcyanate.Inale-ertoacolleaguehesays“Icannolonger,sotospeak,holdmychemicalwaterandmusttellyouthatIcanmakeureawithoutneedingakidney,whetherofmanordog".Thisissupposedtounderminevitalismasorganicchemicalswerepreviouslythoughttobesynthesisedonlybyorganisms.
Source:h-p://en.wikipedia.org/wiki/File:Friedrich_woehler.jpg

Bibliography/Acknowledgments
JasondeNys
Related Documents