1 Cubes aka. Thermodynamics and Heat Transfer

1 Melting Ice Cubes aka. Thermodynamics and Heat Transfer.
Dec 23, 2015
Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript
3
Melting Ice Cubes: “Icebreaker”
Establish teams and have them create a company name
Run first experiment
First experiment objective: Determine how fast each different test
material melts an ice cube AND how the melting of the ice cube effects the test material’s temperature.
5
What is the room temperature? Measure and record the temperature of each material. Measure the weight of the material being tested.
– 5th grade: Convert the weight from pounds to kilograms– 6th grade: Calculate the mass of the material and compare
it to the actual measurement. – High school: Compare methods for calculating mass and
converting units. I.e., by hand, calculator, spreadsheet, draw 3-dimensionally on a CAD system and measure the properties, web (http://n93.cs.fiu.edu/measures/fulltable.asp), etc.
6
Calculate the area of the ice cube.– Discuss:
» What shape is the ice cube?
» What is the formula for this shape?
» What measurements will be needed?
» How can the necessary measurements be found?
8
Calculate Area
Discuss the best way to locate the following measurements:– Measure angle: α =– Measure the radius: r =
Calculate l
A rl c r h 12
[ ( )]
l r 0 0 1 7 4 5.
9
To Locate Center of Circle
Rotate ice cube, overlapping the curved portion of the ice cube, and trace it again.
Fold the circle in quarters to locate center or use a compass.
10
Area answers.– Give your results to your teacher.– Break into small groups and compare answers.– Come up with one answer per group.
Compare group answers. Using the initial readings, calculate the average. Compare the average to the group answers. The teacher will use this answer to calculate the
volume of the ice cube.
11
Place the ice cube, side down, on the material. Time from the placement to completely melted. Students discuss:
– Why is the ice cube melting?
– What is happening?
– How is it changing form?
– Where does the heat come from?
12
Record:– the finish time – temperature at the center of the puddle– outside edge of the plate
Share data with other groups.
13
Summary of First Experiment Where did the heat come from to melt each
ice cube (from the test material or from the surrounding air) ?
What makes one test material faster at melting the ice cube than another ?
Why did the ice cubes move ?
Level of answers will depend on grade level.
14
Thermodynamics
Greek words describe early forms of thermodynamics– Therme (heat)– Dynamics (power)
Today it covers a wider spectrum of energy and energy transformation– I.e., space shuttle to refrigeration
15
Thermodynamics Is the interaction between energy and matter and
it is everywhere– Hair dryers and heat guns, irons, furnace, air
conditioners, hot water tanks, etc.– Also must be considered when designing computers,
automobile engines, VCRs, CD players, dimmer switches, etc.
What happens if – a hair dryer gets too hot?– a computer gets too hot?
16
5th Grade
Calculate T (Delta Time - change in temperature of the material being tested.)– (Tfinal - Tinitial)
Compare student T results to calculated T, supplied by the teacher, in a line graph on graph paper or using a spreadsheet.– Discuss the results
17
5th Grade con’t.
Compare the amount of heat (Q) each material has available to the amount of heat required to melt the ice cube in a combination bar/line graph. (Data supplied by the teacher)– Which material(s) did not have enough heat available
to melt the ice cube?
– What can be done to increase the available heat?
Do you see any correlations between the two graphs?
18
Summary What test material was the best at
melting ice cubes ? Did the color seem to effect the
performance ? Why would an ice cube melt, even if
the test material did not have enough energy to do it ?
19
Thermodynamics Therefore, different materials are used to
the transfer heat» I.e., the material in the computer chip in the electric
radio alarm clock is used to help keep the chip from overheating.
21
Suggestions for Higher Grades Complete 5th grade level mathematics, graphs,
etc., only have the students calculate:– The volume and mass of the ice cube.– The amount of heat generated by each material. – How long a specific material will take to melt an ice
cube. Calculate the volume and mass of the material
being tested, and compared to actual measured weight.
Discuss heat transfer in more depth.
22
Suggestions for Higher Grades con’t.
Create an interactive animated computer program that demonstrates the experiment.– Example:– http://socrates.berkeley.edu:7009/simple_machines/
23
Additional Exercises Compare the same material with different masses. Compare different materials with the same mass. Conduct a web search of items that use heat sinks. Examples:
– Library of Thermodynamics Arizona State Univ. » http://www.asu.edu/lib/noble/physics/thermo.htm
– Heating system (heat pipe sinks) and fans» http://www.kita.or.kr/catalog/cheil/index.html
– Laptops » http://www.indek.com/heatpipe/hp_app.htm
– Computers» http://www.thermalloy.com/catalog/htm/dhs57.htm » http://www.web_tronics.com/webtronics/heatredmouns.html
– http://www.heatsink.com/– http://www.execpc.com/industrialelectronics/wakefld/wakepg19.html – http://www.marlow.com/d_heat.htm
– Dimmer» http://home.swbell.net/evansjim/MyHomeRepair/DimmerSwitch.htm
– http://www.thermalloy.com/catalog/htm/eprof41b.htm
26
THERMODYNAMICS The science of energy (or its
ability to cause changes), and The relationships among the
properties of matter. HEAT, Q, is the form of energy
which melted our ice cubes. In the SI system, we measure Q in Joules.
27
THERMODYNAMICS
Some important material properties: m is the mass of the material (kg) V is the volume (m3) is the density (kg/m3) C is the specific heat (J/kg-oC)
28
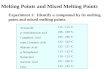
Some Material PropertiesSpecific Thermal
Density Heat Conductivity
Material (kg/m3) (J/kg-oC) (W/m-oC)
Steel 7,850 434 60Iron 7,870 447 73
Aluminum 2,770 875 177Copper 8,930 385 401Lead 11,340 129 35
Pyrex Glass 2,225 835 1.4Brick 1,920 835 0.72
Pine Wood 640 2,805 0.15Plywood 545 1,215 0.12
Ice (near 0oC) 920 2,040 1.88
Water (near 0oC) 1,000 4,230 0.57
Latent Heat of Fusion for Solid/Liquid Water: 333,700 J/kg
29
THERMODYNAMICS For a solid, Q = m C T This is the amount of heat corresponding
to a change in temperature If you don’t know the mass, calculate it
from: m = V T is the change in temperature,
(Tfinal - Tinitial)
30
How much heat does it take to melt one of our ice cubes ?
If the ice cube is at 0oC, “Latent Heat of Fusion” (amount of
energy needed to go from solid to liquid states.
For water, that is 333,700 Joules/kg. If our ice cube is 0.01 kg, the heat
required is 3,337 Joules.
31
Do we have enough energy in our test materials to do that ?
Example: A 0.5 kg. chunk of steel, starting at 22oC,
releases 3255 Joules of heat when it is cooled to 7oC.
Q = m C T = (0.5 kg)(434 Joules/kg-oC)(22-7 oC) = 3255 Joules
3337 Joules is needed, therefore, there isn’t enough heat to melt the ice cube
32
Conservation of EnergyBetter yet, we can solve for the final
temperature of the steel to melt the ice:
Q
m C
Jo u les
k gJo u les
k g C
C
T T T C C
o
o
fin a l in itia lo o
3 3 3 7
0 5 4 3 41 5 3 8
2 2 1 5 3 8 6 6 2
( . )( ).
. .
33
Conservation of Energy A 0.5 kg block of steel Cools from room temperature
(22oC) to 6.62oC Gives up enough heat to melt a
0.01 kg ice cube.
34
Heat Transfer– is the flow of energy which happens when
a difference in temperature exists. – can happen between two bodies or even
within a single body.
What was the difference in temperature between our ice cubes and our test materials ?
36
Fourier’s Law Q is the heat transfer rate k is a material property, thermal
conductivity A is the area which heat flows through T is the temperature difference x is the distance the heat must travel
Q k AT
x
37
How do you make the ice cubes melt faster ?
What do the terms in Fourier’s Law show us ?
Which variables can you control ?
Fourier’s Law Q k AT
x
38
Fourier’s Law Fourier’s Law tells us how fast
heat will flow. Do we know if there is enough
energy available in our test materials to melt our ice cube ?
Q k AT
x
39
Fourier’s Law
The rate of heat flow is: The steel block cools from 22oC to 6.62oC
in melting the ice which is 0oC. As that happens, the value of T decreases. Therefore, the rate of heat transfer to the ice
decreases. How can we increase the rate for a given
material ?
Q k AT
x
Related Documents