1 Gases solid liquid gas Condensed phases (s, l) High density. Particles close to each other. Strong attraction forces between particles. Motion of particles limited. Gas phase (g) Low density. Particles far from each other. Weak attraction forces between particles. Particles move very fast. Volume of gas phase is volume of container. V/

1 Gases solid liquidgas Condensed phases (s, l) High density. Particles close to each other. Strong attraction forces between particles. Motion.
Dec 26, 2015
Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

1
Gases
solid liquid gas
Condensed phases (s, l)High density.Particles close to each other.Strong attraction forces between particles.Motion of particles limited.
Gas phase (g)Low density.Particles far from each other.Weak attraction forces between particles.Particles move very fast.Volume of gas phase is volume of container.
V/

2
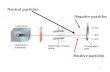
a. Particles move fast in all directions (randomly).b. Particles collide with each other.c. Particles collide with the walls. V/

3
Ideal gas
Hypothetical model1) Gas particles have zero volume.
Just a point (radius =0).
2) No attraction/repulsion between gas particles.Potential energy is zero.
Space between particles
V
Particle volume
b
Volume of gas= Space between particles + volume of particles
Vg = V + bVg ≈ V V/

4
Ideal Gas Law
TRnVp
Can be derived theoretically under above assumptions. Can be arrived to experimentally at relatively low pressures and high temperatures (Boyle, Charles, Guy Lussac, Avogadro, Amonton).
Gas volumeGas pressureAmount of gas
in molesGeneral gas
constant
temperature
Temperature (Kelvin, K): A measure of the kinetic energy of the gas particle
T α Ekinetic
T α ½ m v2 T α v2
v α √T
R8.314 J mol-1K-1
p in Pa, V in m3
0.0821 atm. L. mol-1 K-1
p in atm., V in LV/

5
Gas pressure Particles collide with the wall. Force exerted on wall. Pressure = Force / A Pa = N / m2
1 bar = 100 000 Pa 1 atm. = 101 325 Pa = 760 mmHg 1 mmHg = 1 torr
p = ρ g h
height
density
V/

6
Boyle’s Law
constant temperature
constant temperature, constant amount of gasconstant T and n
Vp
V
constp
VpVpconstVp
constTnTRnVp
1.
.
.,
2211
T3
T1
p
V
T3 > T
2 > T
1
T2
V/

7
Charles’ Law
TVTconstV
T
V
T
Vconst
p
Rn
T
V
constpnTRnVp
.
.
.,
2
2
1
1
constant pressure, constant amount of gasconstant p and n
A
gm
A
Fpp opposite
oppositegas
m
m
V
T
p1p2
p3
p1 < p2 < p3
V/

8
Amonton’s Law
TPTconstP
T
P
T
Pconst
V
Rn
T
P
constVnTRnVp
.
.
.,
2
2
1
1
constant volume, constant amount of gasconstant V and n
P
T
V1
V2
V3
V1 < V2 < V3
V/

9
Combined Gas Law
2
22
1
11.
.
T
Vp
T
Vpn
T
VpconstRn
T
Vp
constnTRnVp
constant amount of gasconstant n
Problem: What is the volume of a gas at STP if its volume at room temperature and at 300 mmHg was 250 cm3?
STP: Standard Temperature and Pressure T=0ºC=273 K p=1 atm.=760 torr
Room temperature25ºC=298 K
T / K = t /ºC + 273
298 K300 mmHg
250 cm3
T1 , p1 , V1
273 K760 mmHg
? cm3
T2 , p2 , V2
32
3
2
22
2
1
11
4.90
760
273
298
250300
cmV
mmHg
K
K
cmmmHgV
Vp
T
T
Vp
V/

10
Avogadro’s Law
nVnconstV
n
V
n
Vconst
p
TR
n
V
constpTTRnVp
.
.
.,
2
2
1
1
constant temperature and pressureconstant T and p
At constant temperature and pressure, the gas volume is proportional to its amount (number of moles).
Molar volume: volume of 1 mole of gas
Latm
KKmolLatmmolV
nKTatmpSTPAt
p
TRnV
4.22.1
273..0821.01
1273.1:11
At 1 atm. and 25ºC: molar volume is 24 L V/

11
Mwt
TRdp
V
mdTR
MwtV
mp
TRMwt
mVp
Mwt
mnTRnVp
Latm
KKmolLatm
molg
gV
p
TR
Mwt
mV
3.21.88.0
293..0821.0
32
25 11
1
What volume will 25 g of O2 occupy at 20ºC and 0.88 atm.?
Calculate the density of oxygen at STP?
LgKKmolLatm
molgatmd
TR
Mwtpd
Mwt
TRdp
/308.1298..0821.0
32.111
1
V/

12
Dalton’s Law of Partial PressureThe pressure of a gas mixture is the sum of the partial pressures of all gases in the mixture.
...321 pppppi
itotal
The partial pressure of a gas in a mixture is the pressure of that gas if it were alone.
V
TRnp AA
V
TRnp CC
V
TRnp BB
V/

13
V
TRnp
V
TRnnnp
V
TRn
V
TRn
V
TRnp
pppp
totaltotal
CBAtotal
CBAtotal
CBAtotal
Example: 200 mL of N2 at 25ºC and a pressure of 250 torr are mixed with 350 mL of O2 at 25ºC and a pressure of 300 torr so that the resulting volume is 350 mL. What would be the final pressure of the mixture?
mol
KKmolLatm
Latm
KKmolLatm
mLtorr
TR
Vpn
molKKmolLatm
Latm
KKmolLatm
mLtorr
TR
Vpn
O
OOO
N
NNN
00565.0298..0821.0
1000/350.760/300
298..0821.0
350300
00269.0298..0821.0
1000/200.760/250
298..0821.0
200250
1111
1111
2
22
2
2
22
2
torrtorratmatmLV
TRnp totaltotal 517760.680.0.680.0
300.0
2980821.000565.000269.0
V/

14
N2 O2
5 bar1 bar
Calculate the partial pressures of oxygen and nitrogen after opening the stopcock. Calculate the total pressure!
const. temperature!!!
V/

15
2 KClO3(s) → 2 KCl(s) + 3 O2(g)
Pressure of water vapor
A student collects 245 mL of O2 at 25ºC and 758 mmHg. If the vapor pressure of water at 25ºC equals 23.76 mmHg, calculate the partial pressure of oxygen and the volume of dry oxygen at STP!
mmHgp
p
ppp
ppp
O
O
OHtotalO
OHOtotal
734
76.23758
2
2
22
22
V/

16
A student collects 245 mL of O2 at 25ºC and 758 mmHg. If the vapor pressure of water at 25ºC equals 23.76 mmHg, calculate the partial pressure of oxygen and the volume of dry oxygen at STP!
298 K734 mmHg
245 cm3
T1 , p1 , V1
273 K760 mmHg
? cm3
T2 , p2 , V2
STP
2
2
1
112
2
22
1
11
p
T
T
VpV
T
Vp
T
Vp
32
3
2
217
760
273
298
245734
cmV
mmHg
K
K
cmmmHgV
V/

17
Graham’s Law of Effusion
• Escape of gas molecules through a tiny hole into an evacuated space.
• Diffusion:spread of 1 substance throughout a space of another substance.
• Effusion rate of a gas is inversely proportional to the square root of its density (molar mass).
1
2
1
2
1
2
2
1
t
t
d
d
M
M
r
r
dr
Mr
1
1
V/

18
• Compare the effusion rates of helium and molecular oxygen at the same temperature and pressure.
827.2/0003.4
/00.32
2
molg
molg
r
r
O
He
He effuses 2.827 times faster than oxygen.
• A sample of O2 gas (2.0 mmol) effused through a pinhole in 5.0 sec. It will take __??__ seconds for the same amount of H2 gas to effuse under the same conditions.
• A sample of HI gas (MW = 128) effuses at 0.0962 cm/sec. A sample of butylamine gas effuses at 0.126 cm/sec. What is the molecular weight of butylamine?
Laws of effusion apply also for Diffusion.
V/18

19
The glass tube shown above has cotton plugs inserted at either end. The plug on the left is moistened with a few drops of aqueous ammonia, from which NH3 gas slowly escapes. The plug on the right is similarly moistened with a strong solution of hydrochloric acid, from which gaseous HCl escapes. The gases diffuse in opposite directions within the tube; at the point where they meet, they combine to form solid ammonium chloride, which appears first as a white fog and then begins to coat the inside of the tube.
NH3(g) + HCl(g) → NH4Cl(s)
a) In what part of the tube (left, right, center) will the NH4Cl first be observed?b) If the distance between the two ends of the tube is 100 cm, how many cm from the left end of the tube will the NH4Cl first form?
.tanconstistdr
t
x
time
cedisr
1
2
2
1
M
M
x
x
x1 x2
V/
Related Documents