Cell Reports, Volume 18 Supplemental Information b-Hydroxybutyrate Deactivates Neutrophil NLRP3 Inflammasome to Relieve Gout Flares Emily L. Goldberg, Jennifer L. Asher, Ryan D. Molony, Albert C. Shaw, Caroline J. Zeiss, Chao Wang, Ludmilla A. Morozova-Roche, Raimund I. Herzog, Akiko Iwasaki, and Vishwa Deep Dixit

Welcome message from author
This document is posted to help you gain knowledge. Please leave a comment to let me know what you think about it! Share it to your friends and learn new things together.
Transcript

Cell Reports, Volume 18
Supplemental Information
b-Hydroxybutyrate Deactivates Neutrophil
NLRP3 Inflammasome to Relieve Gout Flares
Emily L. Goldberg, Jennifer L. Asher, Ryan D. Molony, Albert C. Shaw, Caroline J.Zeiss, Chao Wang, Ludmilla A. Morozova-Roche, Raimund I. Herzog, AkikoIwasaki, and Vishwa Deep Dixit

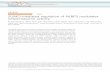
- - - + + + + + + + + +- - - - - - - - - - - -- + - - 5 25 50 100 - - - -- - + - - - - - 5 25 50 100
- - - - - - - - - +- + + + + + + + + +- 5 25 50 100 - - - - -- - - - - 5 25 50 100 -
LPSATP
S100A9 fibril (μg)S100A9 oligo (μg)
pro-IL-1β
p17
E
0 20 40 60 80 100Vector Length (nm)
Hei
ght (
nm)
0.00.51.01.52.02.5
0 500 1000 1500Time (min)
Nor
m. f
luor
esce
nce
0.0
0.5
1.0B
Vector Length (nm)
Hei
ght (
nm)
1
2
3
0 10 20 30 400
C DA
F
Figure S1. Role of S100A9 in NLRP3 inflammasome activation and KD during bacterial infection, related to Figure 1. (A) AFM height image of S100A9 amyloid oligomers; scale bars equal to 120 nm. (B, C) AFM cross-sectional analysis of representative amyloid (B) oligomer and (C) fibril. (D) Kinetics of S100A9 amyloid monitored by thioflavin T fluorescence intensity. (E) BMDM were primed with S100A9 in lieu of LPS. (F) LPS-primed BMDM were treated with S100A9 as a DAMP. (E, F) IL-1β secretion was measured in culture supernates by Western blot. (G-I) Mice were infected with 106 or 108 cfu S. aureus. (G) Blood BHB levels were monitored through the course of infection. 24 hours post-infection, (H) blood BHB and (I) total cells in BALF were measured. Data are pooled from 2 independent experiments. Statistical differences were calculated by 2-way ANOVA (G) and 1-way ANOVA (H, I) with Bonferroni’s correction for multiple comparisons.
GH I

NLRP3
ASC
β-Actin
A O A O A O
LPSLPS+ATP
IB: anti-IL-1β
p17
A O A O
LPSLPS+
C6
Figure S2. NLRP3 inflammasome activation is maintained during aging, related to Figure 2.(A) Neutrophil isolation purity from adult and old mice. (B) Human neutrophil isolation purity. Top panel shows staining in un-enriched PBMC fraction after Ficoll density gradient. (C) Representative FCM gating strategy to identify bone marrow neutrophils. (D) Total femur bone marrow cellularity, (E) neutrophil abundance based on C, so that (F) total neutrophils could be quantified. (G) Expression of NLRP3 inflammasome protein components NLRP3 and ASC was assessed by Western blot in unstimulated neutrophils. Activation of NLRP3 inflammasome was assessed by IL-1β secretion from isolated adult and old neutrophils after (H) LPS+ATP or (I) LPS+ceramide (C6) stimulation in vitro. (D-F) Data are pooled from 2 independent experiments, each symbol represents an individual mouse. Statistical differences were calculated by t-test. (G-I) Data are representative of at least 3 independent experiments, each with n=3-5 pooled per sample.
PBMC
fra
ction
after
Ficoll
grad
ient
Adult
Neu
troph
ilsaft
er en
richm
ent
Old N
eutro
phils
after
enric
hmen
t
A B
C D
E F G H I

A B C
Figure S3. Ketogenic diet does not alter neutrophil infiltration into peritoneal cavity during peritonitis, related to Figure 3. Old mice were fed standard chow or ketogenic diet for 1 week prior to induction of peritonitis by i.p. injection of MSU crystals. (A) Total cells recovered from peritoneal cavity. (B) Neutrophil frequency within peritoneal cells was assessed by flow cytometry. (C) Total neutrophil infiltration into peritoneal cavity was calculated. Data are pooled from 3 independent experiments. Student’s t-tests were used to test for significant differences between old mice fed chow vs KD. Sham mice are included for illustrative purposes to demonstrate induction of inflammatory response after injection of MSU.

Caspase-11
Gasdermin d
β-actin
IL-1β
p17
Cells
Sup
Untre
ated
LPS
LPS+
ATP
1mM
BHB
5mM
BHB
10m
M B
HB
LPS+ATP
Figure S4. BHB does not alter neutrophil pyroptosis or viability, related to Figure 4.Impact of BHB on neutrophil cell death was assessed. (A) Markers of pyroptosis (caspase-11 and Gasdermin d) were assessed in neutrophils after activation in the presence of BHB at the indicated concentrations. BHB efficacy was confirmed by measuring secretion of active IL-1β in the culture supernatants. (B) Cells were stained with Sytox Green after in vitro stimulation to measure extracellular DNA as an indicator of cell death. Lysing cells (lysed) with TritonX-100 was also performed as a positive control. Sytox Green Fluorescence (arbitrary units, A.U.) was measured using a fluorescent plate reader. Graph shows raw fluorescence values, expressed as mean ± SEM, and are pooled from 2 independent experiments.
A B

- - - + + + + + + + + +- - - - - - - - - - - -- + - - 5 25 50 100 - - - -- - + - - - - - 5 25 50 100
- - - - - - - - - +- + + + + + + + + +- 5 25 50 100 - - - - -- - - - - 5 25 50 100 -
LPSATP
S100A9 fibril (μg)S100A9 oligo (μg)
pro-IL-1β
p17
E
0 20 40 60 80 100Vector Length (nm)
Hei
ght (
nm)
0.00.51.01.52.02.5
0 500 1000 1500Time (min)
Nor
m. f
luor
esce
nce
0.0
0.5
1.0B
Vector Length (nm)
Hei
ght (
nm)
1
2
3
0 10 20 30 400
C DA
F
Figure S1. Role of S100A9 in NLRP3 inflammasome activation and KD during bacterial infection, related to Figure 1. (A) AFM height image of S100A9 amyloid oligomers; scale bars equal to 120 nm. (B, C) AFM cross-sectional analysis of representative amyloid (B) oligomer and (C) fibril. (D) Kinetics of S100A9 amyloid monitored by thioflavin T fluorescence intensity. (E) BMDM were primed with S100A9 in lieu of LPS. (F) LPS-primed BMDM were treated with S100A9 as a DAMP. (E, F) IL-1β secretion was measured in culture supernates by Western blot. (G-I) Mice were infected with 106 or 108 cfu S. aureus. (G) Blood BHB levels were monitored through the course of infection. 24 hours post-infection, (H) blood BHB and (I) total cells in BALF were measured. Data are pooled from 2 independent experiments. Statistical differences were calculated by 2-way ANOVA (G) and 1-way ANOVA (H, I) with Bonferroni’s correction for multiple comparisons.
GH I

NLRP3
ASC
β-Actin
A O A O A O
LPSLPS+ATP
IB: anti-IL-1β
p17
A O A O
LPSLPS+
C6
Figure S2. NLRP3 inflammasome activation is maintained during aging, related to Figure 2.(A) Neutrophil isolation purity from adult and old mice. (B) Human neutrophil isolation purity. Top panel shows staining in un-enriched PBMC fraction after Ficoll density gradient. (C) Representative FCM gating strategy to identify bone marrow neutrophils. (D) Total femur bone marrow cellularity, (E) neutrophil abundance based on C, so that (F) total neutrophils could be quantified. (G) Expression of NLRP3 inflammasome protein components NLRP3 and ASC was assessed by Western blot in unstimulated neutrophils. Activation of NLRP3 inflammasome was assessed by IL-1β secretion from isolated adult and old neutrophils after (H) LPS+ATP or (I) LPS+ceramide (C6) stimulation in vitro. (D-F) Data are pooled from 2 independent experiments, each symbol represents an individual mouse. Statistical differences were calculated by t-test. (D-F) Data are representative of at least 3 independent experiments, each with n=3-5 pooled per sample.
PBMC
fra
ction
after
Ficoll
grad
ient
Adult
Neu
troph
ilsaft
er en
richm
ent
Old N
eutro
phils
after
enric
hmen
t
A B
C D
E F G H I

A B C
Figure S3. Ketogenic diet does not alter neutrophil infiltration into peritoneal cavity during peritonitis, related to Figure 3. Old mice were fed standard chow or ketogenic diet for 1 week prior to induction of peritonitis by i.p. injection of MSU crystals. (A) Total cells recovered from peritoneal cavity. (B) Neutrophil frequency within peritoneal cells was assessed by flow cytometry. (C) Total neutrophil infiltration into peritoneal cavity was calculated. Data are pooled from 3 independent experiments. Student’s t-tests were used to test for significant differences between old mice fed chow vs KD. Sham mice are included for illustrative purposes to demonstrate induction of inflammatory response after injection of MSU.

Caspase-11
Gasdermin d
β-actin
IL-1β
p17
Cells
Sup
Untre
ated
LPS
LPS+
ATP
1mM
BHB
5mM
BHB
10m
M B
HB
LPS+ATP
Figure S4. BHB does not alter neutrophil pyroptosis or viability, related to Figure 4.Impact of BHB on neutrophil cell death was assessed. (A) Markers of pyroptosis (caspase-11 and Gasdermin d) were assessed in neutrophils after activation in the presence of BHB at the indicated concentrations. BHB efficacy was confirmed by measuring secretion of active IL-1β in the culture supernatants. (B) Cells were stained with Sytox Green after in vitro stimulation to measure extracellular DNA as an indicator of cell death. Lysing cells (lysed) with TritonX-100 was also performed as a positive control. Sytox Green Fluorescence (arbitrary units, A.U.) was measured using a fluorescent plate reader. Graph shows raw fluorescence values, expressed as mean ± SEM, and are pooled from 2 independent experiments.
A B

Supplemental Experimental Procedures
Animals
Male and female adult (8-16 weeks) wild type mice were obtained from the Jackson Laboratory or bred within our
animal colony. Aged NLRP3 and ASC-deficient mice (Youm et al., 2013), FCAS mice (Brydges et al., 2009), and
Caspase1/11-/- mice (Kuida et al., 1995) have all been previously described. Old C57BL/6 mice (male, 18-24
months) were obtained from the National Institute on Aging Aged Rodent Colony. Outbred Sprague Dawley rats
(male, 8 weeks) were purchased from Envigo.
In vivo hypoglycemia
6 human subjects had bilateral antecubital i.v. catheters placed and underwent a hyperinsulinemic – hypoglycemic
clamp with 2mU/kg/min regular human insulin and a variable infusion of 20% dextrose (depicted in Figure 2F).
Throughout the procedure subjects underwent q5min plasma glucose measurements by glucose analyzer (YSI,
Yellow Springs, OH) to monitor the glycemic level. Once stable hypoglycemia with target glucose of 55mg/dl was
reached, whole blood samples were collected and neutrophils isolated and assayed as described below.
NLRP3 activation
For inflammasome activation assays, cells were plated 106 cells/ml in RPMI (10%FBS+1% antibiotic). Cells were
primed with LPS (1µg/mL, 4hr) or S100A9 fibrils (50µg/mL, 4hr, see Supplemental Information and Figure S1A-D)
followed by treatment with ATP (5mM, 1hr), ceramide (80µg/mL, 6hr), MSU (250µg, 5hr) BHB (1-10mM as
indicated, 1hr), niacin (5mM, 1hr), 3-MA (10mM, 1hr), and/or AOA (1mM, 1hr) as indicated. For S100A8
secretion, cells were stimulated with LPS+BHB for 18-20 hours.
Western blot
For Western Blot analyses, total protein content was measured by DC Protein Assay (Bio-RAD), separated by SDS-
PAGE, and visualized by chemiluminescence (Bio-RAD). The following antibodies (and source) were used to
measure protein expression: IL-1β (Genetex), NLRP3 (Adipogen), ASC (Enzo), β-Actin (Cell Signaling), SCOT
(Proteintech), HMGCL (Novus Bio), Caspase-11 (Novus), Gasdermin d (Sigma), Caspase-1 (generously provided
by Genentech), Tubulin (Sigma), pNFκB (p65, Cell Signaling), total NFκB (p65, Cell Signaling), followed by
incubation with appropriate HRP-conjugated secondary antibodies (Thermo).
Flow cytometry
Cells were pre-incubated with Fc-block (eBioscience) followed by surface staining, and then fixed with 2%
paraformaldehyde. Cells were gated based on viability (Life Technologies live/dead). Mouse antibodies used: Gr-1
(RB6-8C5), CD11b (M1/70), CD19 (1D3), CD3 (2C11), Ly6G (1A8), and F4/80 (BM8). Human antibodies used:
CD3 (OKT3), CD19 (HIB19), CD66b (G10F5), CD16 (eBioCB16). All antibodies were purchased from
eBioscience or Biolegend. Samples were collected on an LSRII flow cytometer (BD Bioscience) and analyzed with
FlowJo (TreeStar).
RNA Analysis
Cells were lysed in Trizol and RNA was extracted using the Qiagen RNeasy micro kit. cDNA was synthesized using
the Bio-RAD iScript cDNA synthesis kit according to manufacturer’s protocol. Gene expression was measured by
RT-PCR using the ΔΔCt method and normalized to Gapdh.
Production and verification of S100A9 amyloid species
S100A9 was expressed in E coli and purified as described previously (Vogl et al., 2006). Its concentration was
determined by using ε280 = 0.53 (mg/ml)−1 cm−1. In order to avoid the presence in S100A9 solution of the preformed
aggregates, protein was initially dissolved in 10 mM PBS buffer, pH 7.4, subjected to 15 min sonication, then to 15
min centrifugation at 14 000 rpm in a minicentrifuge (Eppendorf Centrifuge 5417R) and the supernatant collected
from the upper layer was filtered through the 0.22 um filter (Millex). The final solution was subjected to incubation
at 1 mg/ml concentration in 10 mM PBS buffer, pH 7.4, 37oC, using continuous agitation at 300 rpm (Eppendorf
Thermomixer Compact). Amyloid oligomers and fibrils of S100A9 were produced after 2 h and 24 h of incubation,
respectively. The samples were lyophilized for storage and reconstituted in PBS buffer prior applications. The

morphological parameters of reconstituted oligomers were compared with freshly produced structures by using
AFM imaging and they were confirmed to be essentially unchanged. The oligomers of S100A9 displayed a round-
shaped morphology (Figure S1A) assessed by AFM imaging with heights measured by AFM cross-section analysis
of 1-2.0 nm (Figure S1B). The oligomeric nature of these species was verified by interaction with generic A11
antibodies reactive towards amyloid oligomers (gift from Dr Rakez Kayed, Texas University, US). Fibrillar
structures (Figure 1A) displayed typical fibrillar morphology characterized by ca. 3 nm height as determined in the
AFM cross-sections and were coiled and flexible according to their appearance. (Figure S1C). They were
characterized by ca. 6 fold increase in thioflavin-T fluorescence intensity (Figure S1D).
Fluorescence assay
The thioflavin T binding assay was performed using a modification of LeVine’s method (LeVine, 1993). Thioflavin
T fluorescence was measured by a Jasco FP-6500 spectrofluorometer (Jasco, Japan), using excitation at 440 nm and
collecting emission between 450–550 nm, with excitation and emission slits set at 5 nm width.
AFM
Atomic force microscopy (AFM) imaging was carried out by a BioScope Catalyst AFM (Bruker) in peak force
mode in air at a resonance frequency of ca. 70 kHz and a resolution of 256 256 pixels; scan sizes were from 0.5 to
10 μm. S100A9 amyloid samples were deposited on the surface of freshly cleaved mica (Ted Pella) for 15 min,
washed 3 times with 100 μl deionized water and dried at room temperature.

Supplemental References
Brydges, S.D., Mueller, J.L., McGeough, M.D., Pena, C.A., Misaghi, A., Gandhi, C., Putnam, C.D., Boyle, D.L.,
Firestein, G.S., Horner, A.A., et al. (2009). Inflammasome-mediated disease animal models reveal roles for innate
but not adaptive immunity. Immunity 30, 875-887.
Kuida, K., Lippke, J.A., Ku, G., Harding, M.W., Livingston, D.J., Su, M.S., and Flavell, R.A. (1995). Altered
cytokine export and apoptosis in mice deficient in interleukin-1 beta converting enzyme. Science 267, 2000-2003.
LeVine, H., 3rd (1993). Thioflavine T interaction with synthetic Alzheimer's disease beta-amyloid peptides:
detection of amyloid aggregation in solution. Protein science : a publication of the Protein Society 2, 404-410.
Vogl, T., Leukert, N., Barczyk, K., Strupat, K., and Roth, J. (2006). Biophysical characterization of S100A8 and
S100A9 in the absence and presence of bivalent cations. Biochimica et biophysica acta 1763, 1298-1306.
Youm, Y.H., Grant, R.W., McCabe, L.R., Albarado, D.C., Nguyen, K.Y., Ravussin, A., Pistell, P., Newman, S.,
Carter, R., Laque, A., et al. (2013). Canonical Nlrp3 inflammasome links systemic low-grade inflammation to
functional decline in aging. Cell metabolism 18, 519-532.
Related Documents